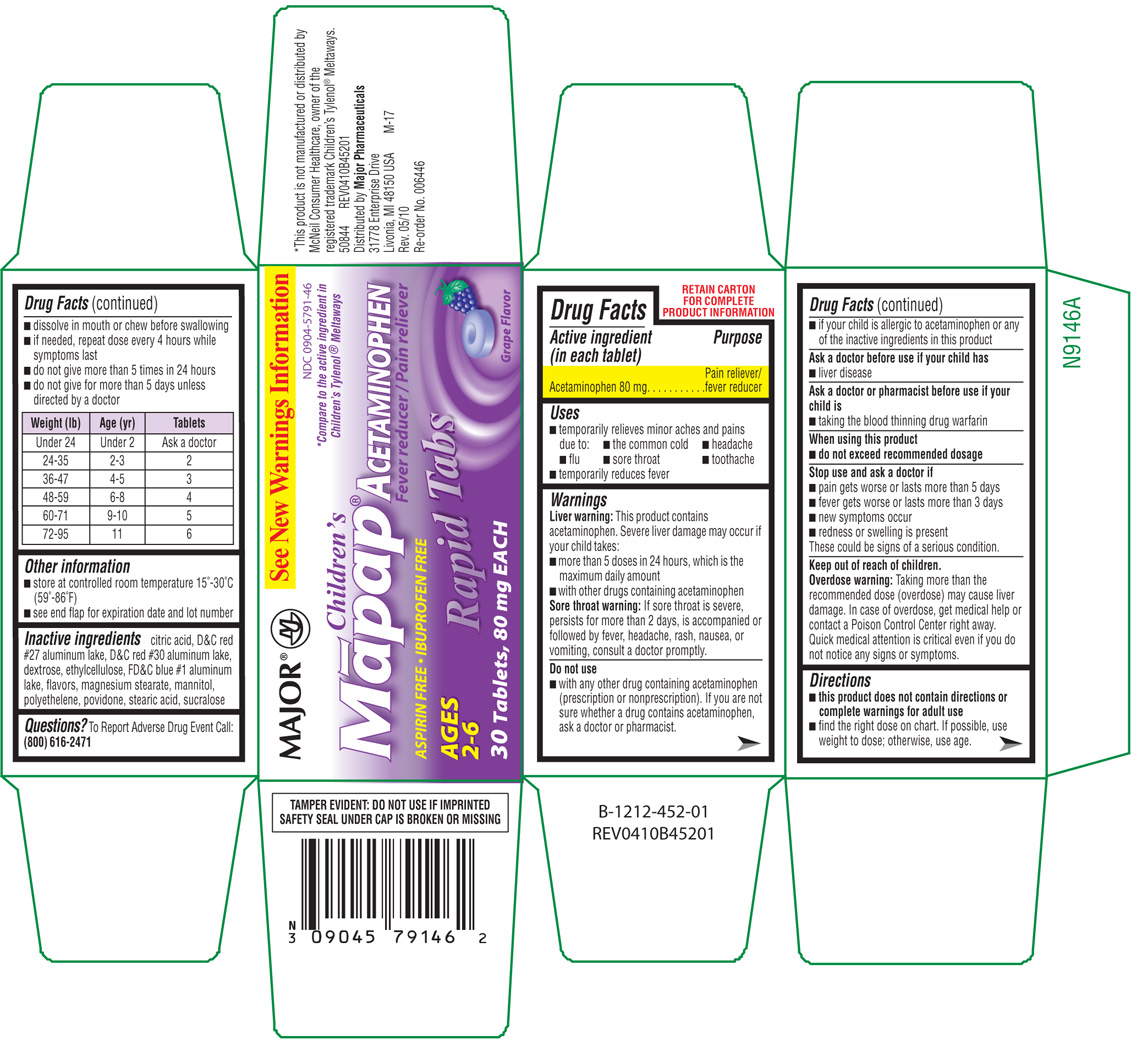
CHILDRENS MAPAP
-
acetaminophen tablet, chewable
Major Pharmaceuticals
----------
Major 44-452Active ingredient
(in each tablet)
Acetaminophen 80 mg
Purpose
Pain reliever/fever reducer
Uses
- temporaily relieves minor aches and pains due to:
- the common cold
- headache
- flu
- sorethroat
- toothache
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if your child takes:
- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if your child is allergic to acetaminophen or any of the inactive ingredients in this product
Ask a doctor before use if your child has
- liver disease
Ask a doctor or pharmacist before use if your child is
- taking the blood thinning drug warfarin
When using this product
- do not exceed recommended dosage
Stop use and ask a doctor if
- pain gets worse or lasts more than 5 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
These could be signs of a serious condition.
Keep out of reach of children.
Overdose warning: Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical even if you do not notice any signs or symptoms.
Directions
- this product does not contain directions or complete warnings for adult use
- find the right dose on chart. If possible, use weight to dose; otherwise, use age.
- dissolve in mouth or chew before swallowing
- if needed, repeat dose every 4 hours while symptoms last
- do not give more than 5 times in 24 hours
- do not give for more than 5 days unless directed by a doctor
| Weight (lb) | Age (yr) | Tablets |
| Under 24 | Under 2 | Ask a doctor |
| 24-35 | 2-3 | 2 |
| 36-47 | 4-5 | 3 |
| 48-59 | 6-8 | 4 |
| 60-71 | 9-10 | 5 |
| 72-95 | 11 | 6 |
Other information
- store at controlled room temperature 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients
citric acid, D&C red #27 aluminum lake, D&C red #30 aluminum lake, dextrose, ethylcellulose, FD&C blue #1 aluminum lake, flavors, magnesium stearate, mannitol, polyethelene, povidone, stearic acid, sucralose
Questions or comments?
1-800-426-9391
Product Packaging
The product packaging shown below represents a sample of that currently in use. Additional packaging may also be available.
MAJOR®
See New Warnings Information
NDC 0904-5791-46
*Compare to the active ingredient in
Children's Tylenol® Meltaways
Children's
Mapap®
Acetaminophen
Fever reducer / Pain reliever
ASPIRIN FREE • IBUPROFEN FREE
AGES 2-6
Rapid Tabs
30 Tablets, 80 mg EACH
Grape Flavor
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
50844 REV0410B45201
Distributed by Major Pharmaceuticals
31778 Enterprise Drive
Livonia, MI 48150 USA M-17
Rev. 05/10
Re-order No. 006446
Product Packaging
| CHILDRENS MAPAP
acetaminophen tablet, chewable |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH NOT FINAL | part343 | 12/06/2004 | |
| Labeler - Major Pharmaceuticals (191427277) |