LORATADINE
-
loratadine tablet
Rebel Distributors Corp
----------
Loratadine TabletsActive ingredient
Loratadine USP, 10 mg
Purpose
Antihistamine
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies: · itching of the nose or throat · runny nose · itchy, watery eyes · sneezing
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before use if you have
liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
do not take more than directed. Taking more than directed may cause drowsiness.
Stop use and ask a doctor if
an allergic reaction to thsi product occurs. Seek medical help right away.
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
| adults and children 6 years and over | 1 tablet daily; not more than 1 tablet in 24 hours |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information
store between 20 and 25°C (68 and 77°F) · protect from excessive moisture.
Inactive ingredients
corn starch, lactose monohydrate, magnesium stearate, pregelatinized starch
Questions?
call 1-800-406-7984
†This product is not manufactured or distributed by Schering-Plough Healthcare Products Inc., owner of the registered trademark Claritin®.
Manufactured by
Ohm Laboratories, Inc.
North Brunswick, NJ 08902
Repackaged by
Rebel Distributors Corp
Thousand Oaks, CA 91320
Principal Display Panel
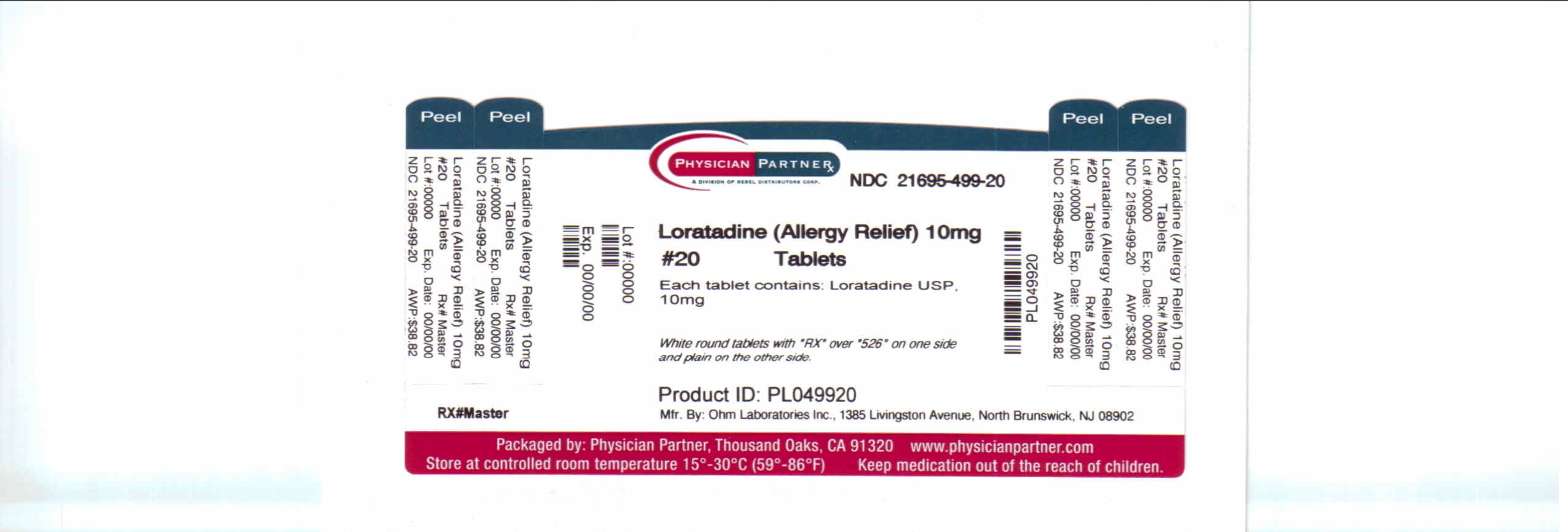
| LORATADINE
loratadine tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| Unapproved drug other | 01/21/2010 | ||
| Labeler - Rebel Distributors Corp (118802834) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Rebel Distributors Corp | 118802834 | RELABEL, REPACK | |