aggrenox (aspirin and dipyridamole) capsule
[Boehringer Ingelheim Pharmaceuticals Inc.]
Note to Pharmacist: Dispense in this unit-of-use container. If repackaging is requested, use USP tight containers with not more than 60 capsules per container.
Prescribing Information
DESCRIPTION
AGGRENOX capsules is a combination antiplatelet agent intended for oral administration. Each hard gelatin capsule contains 200 mg dipyridamole in an extended-release form and 25 mg aspirin, as an immediate-release sugar-coated tablet. In addition, each capsule contains the following inactive ingredients: acacia, aluminum stearate, colloidal silicon dioxide, corn starch, dimethicone, hypromellose, hypromellose phthalate, lactose monohydrate, methacrylic acid copolymer, microcrystalline cellulose, povidone, stearic acid, sucrose, talc, tartaric acid, titanium dioxide and triacetin.
Each capsule shell contains gelatin, red iron oxide and yellow iron oxide, titanium dioxide and water.
Dipyridamole
Dipyridamole is an antiplatelet agent chemically described as 2,2',2'',2'''-[(4,8-Dipiperidinopyrimido[5,4-d]pyrimidine-2,6-diyl)dinitrilo]-tetraethanol. It has the following structural formula:
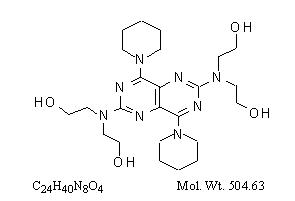
Dipyridamole is an odorless yellow crystalline substance, having a bitter taste. It is soluble in dilute acids, methanol and chloroform, and is practically insoluble in water.
Aspirin
The antiplatelet agent aspirin (acetylsalicylic acid) is chemically known as benzoic acid, 2-(acetyloxy)-, and has the following structural formula:
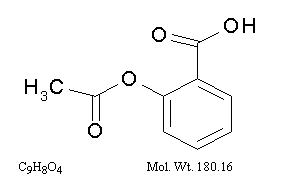
Aspirin is an odorless white needle-like crystalline or powdery substance. When exposed to moisture, aspirin hydrolyzes into salicylic and acetic acids, and gives off a vinegary odor. It is highly lipid soluble and slightly soluble in water.
CLINICAL PHARMACOLOGY
Mechanism of Action
The antithrombotic action of Aggrenox® (aspirin/extended-release dipyridamole) capsules is the result of the additive antiplatelet effects of dipyridamole and aspirin.
Dipyridamole
Dipyridamole inhibits the uptake of adenosine into platelets, endothelial cells and erythrocytes in vitro and in vivo; the inhibition occurs in a dose-dependent manner at therapeutic concentrations (0.5–1.9 μg/mL). This inhibition results in an increase in local concentrations of adenosine which acts on the platelet A2-receptor thereby stimulating platelet adenylate cyclase and increasing platelet cyclic-3',5'-adenosine monophosphate (cAMP) levels. Via this mechanism, platelet aggregation is inhibited in response to various stimuli such as platelet activating factor (PAF), collagen and adenosine diphosphate (ADP).
Dipyridamole inhibits phosphodiesterase (PDE) in various tissues. While the inhibition of cAMP-PDE is weak, therapeutic levels of dipyridamole inhibit cyclic-3',5'-guanosine monophosphate-PDE (cGMP-PDE), thereby augmenting the increase in cGMP produced by EDRF (endothelium-derived relaxing factor, now identified as nitric oxide).
Aspirin
Aspirin inhibits platelet aggregation by irreversible inhibition of platelet cyclooxygenase and thus inhibits the generation of thromboxane A2, a powerful inducer of platelet aggregation and vasoconstriction.
Pharmacokinetics
There are no significant interactions between aspirin and dipyridamole. The kinetics of the components are unchanged by their co-administration as AGGRENOX.
Dipyridamole
Absorption
Peak plasma levels of dipyridamole are achieved 2 hours (range 1–6 hours) after administration of a daily dose of 400 mg AGGRENOX (given as 200 mg BID). The peak plasma concentration at steady-state is 1.98 μg/mL (1.01–3.99 μg/mL) and the steady-state trough concentration is 0.53 μg/mL (0.18–1.01 μg/mL).
Effect of Food
When Aggrenox® (aspirin/extended-release dipyridamole) capsules were taken with a high fat meal, dipyridamole peak plasma levels (Cmax) and total absorption (AUC) were decreased at steady-state by 20-30% compared to fasting. Due to the similar degree of inhibition of adenosine uptake at these plasma concentrations, this food effect is not considered clinically relevant.
Distribution
Dipyridamole is highly lipophilic (log P=3.71, pH=7); however, it has been shown that the drug does not cross the blood-brain barrier to any significant extent in animals. The steady-state volume of distribution of dipyridamole is about 92 L. Approximately 99% of dipyridamole is bound to plasma proteins, predominantly to alpha 1-acid glycoprotein and albumin.
Metabolism and Elimination
Dipyridamole is metabolized in the liver, primarily by conjugation with glucuronic acid, of which monoglucuronide which has low pharmacodynamic activity is the primary metabolite. In plasma, about 80% of the total amount is present as parent compound and 20% as monoglucuronide. Most of the glucuronide metabolite (about 95%) is excreted via bile into the feces, with some evidence of enterohepatic circulation. Renal excretion of parent compound is negligible and urinary excretion of the glucuronide metabolite is low (about 5%). With intravenous (i.v.) treatment of dipyridamole, a triphasic profile is obtained: a rapid alpha phase, with a half-life of about 3.4 minutes, a beta phase, with a half-life of about 39 minutes, (which, together with the alpha phase accounts for about 70% of the total area under the curve, AUC) and a prolonged elimination phase λz with a half-life of about 15.5 hours. Due to the extended absorption phase of the dipyridamole component, only the terminal phase is apparent from oral treatment with AGGRENOX which, in Trial 9.123 was 13.6 hours.
Special Populations
Geriatric Patients
In ESPS2 (see CLINICAL PHARMACOLOGY, Clinical Trials), plasma concentrations (determined as AUC) of dipyridamole in healthy elderly subjects (>65 years) were about 40% higher than in subjects younger than 55 years receiving treatment with AGGRENOX.
Hepatic Dysfunction
No study has been conducted with the AGGRENOX formulation in patients with hepatic dysfunction.
In a study conducted with an intravenous formulation of dipyridamole, patients with mild to severe hepatic insufficiency showed no change in plasma concentrations of dipyridamole but showed an increase in the pharmacologically inactive monoglucuronide metabolite. Dipyridamole can be dosed without restriction as long as there is no evidence of hepatic failure.
Renal Dysfunction
No study has been conducted with the AGGRENOX formulation in patients with renal dysfunction.
In ESPS2 patients (see CLINICAL PHARMACOLOGY, Clinical Trials), with creatinine clearances ranging from about 15 mL/min to >100 mL/min, no changes were observed in the pharmacokinetics of dipyridamole or its glucuronide metabolite if data were corrected for differences in age.
Aspirin
Absorption
Peak plasma levels of aspirin are achieved 0.63 hours (0.5–1 hour) after administration of a 50 mg aspirin daily dose from AGGRENOX (given as 25 mg BID). The peak plasma concentration at steady-state is 319 ng/mL (175–463 ng/mL). Aspirin undergoes moderate hydrolysis to salicylic acid in the liver and the gastrointestinal wall, with 50%–75% of an administered dose reaching the systemic circulation as intact aspirin.
Effect of Food
When Aggrenox® (aspirin/extended-release dipyridamole) capsules were taken with a high fat meal, there was no difference for aspirin in AUC at steady-state, and the approximately 50% decrease in Cmax was not considered clinically relevant based on a similar degree of cyclooxygenase inhibition comparing the fed and fasted state.
Distribution
Aspirin is poorly bound to plasma proteins and its apparent volume of distribution is low (10 L). Its metabolite, salicylic acid, is highly bound to plasma proteins, but its binding is concentration-dependent (nonlinear). At low concentrations (<100 μg/mL), approximately 90% of salicylic acid is bound to albumin. Salicylic acid is widely distributed to all tissues and fluids in the body, including the central nervous system, breast milk, and fetal tissues. Early signs of salicylate overdose (salicylism), including tinnitus (ringing in the ears), occur at plasma concentrations approximating 200 μg/mL (see ADVERSE REACTIONS and OVERDOSAGE).
Metabolism and Elimination
Aspirin is rapidly hydrolyzed in plasma to salicylic acid, with a half-life of 20 minutes. Plasma levels of aspirin are essentially undetectable 2–2.5 hours after dosing and peak salicylic acid concentrations occur 1 hour (range: 0.5–2 hours) after administration of aspirin. Salicylic acid is primarily conjugated in the liver to form salicyluric acid, a phenolic glucuronide, an acyl glucuronide, and a number of minor metabolites. Salicylate metabolism is saturable and total body clearance decreases at higher serum concentrations due to the limited ability of the liver to form both salicyluric acid and phenolic glucuronide. Following toxic doses (10–20 g), the plasma half-life may be increased to over 20 hours.
The elimination of acetylsalicylic acid follows first-order kinetics with AGGRENOX and has a half-life of 0.33 hours. The half-life of salicylic acid is 1.71 hours. Both values correspond well with data from the literature at lower doses which state a resultant half-life of approximately 2–3 hours. At higher doses, the elimination of salicylic acid follows zero-order kinetics (i.e., the rate of elimination is constant in relation to plasma concentration), with an apparent half-life of 6 hours or higher. Renal excretion of unchanged drug depends upon urinary pH. As urinary pH rises above 6.5, the renal clearance of free salicylate increases from <5% to >80%. Alkalinization of the urine is a key concept in the management of salicylate overdose (see OVERDOSAGE). Following therapeutic doses, about 10% is excreted as salicylic acid and 75% as salicyluric acid, as the phenolic and acyl glucuronides, in urine.
Special Populations
Hepatic Dysfunction: Aspirin is to be avoided in patients with severe hepatic insufficiency.
Renal Dysfunction: Aspirin is to be avoided in patients with severe renal failure (glomerular filtration rate less than 10 mL/min).
Clinical Trials
AGGRENOX was studied in a double-blind, placebo-controlled, 24-month study (European Stroke Prevention Study 2, ESPS2) in which 6602 patients had an ischemic stroke (76%) or transient ischemic attack (TIA, 24%) within three months prior to entry. Patients were randomized to one of four treatment groups: AGGRENOX (aspirin/extended-release dipyridamole) 25 mg/200 mg; extended-release dipyridamole (ER-DP) 200 mg alone; aspirin (ASA) 25 mg alone; or placebo. Patients received one capsule twice daily (morning and evening). Efficacy assessments included analyses of stroke (fatal or nonfatal) and death (from all causes) as confirmed by a blinded morbidity and mortality assessment group.
Stroke Endpoint
AGGRENOX reduced the risk of stroke by 22.1% compared to aspirin 50 mg/day alone (p =0.008) and reduced the risk of stroke by 24.4% compared to extended-release dipyridamole 400 mg/day alone (p = 0.002) (Table 1). AGGRENOX reduced the risk of stroke by 36.8% compared to placebo (p <0.001).
| Total Number of Patients n | Number of Patients With Stroke Within 2 Years n (%) | Kaplan-Meier Estimate of Survival at 2 Years (95% C.I.) | Gehan-Wilcoxon Test P-value |
Risk Reduction at 2 Years |
Odds Ratio (95% C.I.) |
|
|---|---|---|---|---|---|---|
| *0.010 < p-value ≤0.050; **p-value ≤0.010. | ||||||
| Note: ER-DP = extended-release dipyridamole 200 mg; ASA = aspirin 25 mg. The dosage regimen for all treatment groups is BID | ||||||
| Individual Treatment Group |
||||||
| AGGRENOX | 1650 | 157 ( 9.5%) | 89.9% (88.4%, 91.4%) | - | - | - |
| ER-DP | 1654 | 211 (12.8%) | 86.7% (85.0%, 88.4%) | - | - | - |
| ASA | 1649 | 206 (12.5%) | 87.1% (85.4%, 88.7%) | - | - | - |
| Placebo | 1649 | 250 (15.2%) | 84.1% (82.2%, 85.9%) | - | - | - |
| Pairwise Treatment Group Comparisons | ||||||
| AGGRENOX vs. ER-DP | - | - | - | 0.002** | 24.4% | 0.72 (0.58, 0.90) |
| AGGRENOX vs. ASA | - | - | - | 0.008** | 22.1% | 0.74 (0.59, 0.92) |
| AGGRENOX vs. Placebo | - | - | - | <0.001** | 36.8% | 0.59 (0.48, 0.73) |
| ER-DP vs. Placebo | - | - | - | 0.036* | 16.5% | 0.82 (0.67, 1.00) |
| ASA vs. Placebo | - | - | - | 0.009** | 18.9% | 0.80 (0.66, 0.97) |
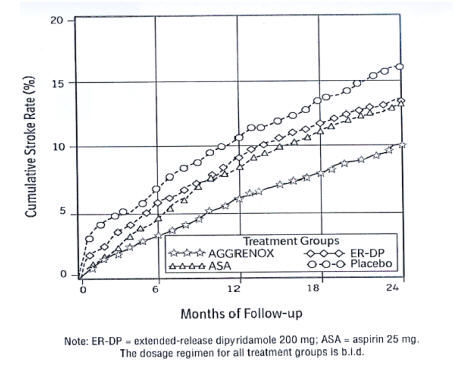
ESPS2: Cumulative Stroke Rate (Fatal or Nonfatal) Over 24 months of Follow-Up
Combined Stroke or Death Endpoint
In ESPS2, AGGRENOX reduced the risk of stroke or death by 12.1% compared to aspirin alone and by 10.3% compared to extended-release dipyridamole alone. These results were not statistically significant. AGGRENOX reduced the risk of stroke or death by 24.2% compared to placebo.
Death Endpoint
The incidence rate of all cause mortality was 11.3% for AGGRENOX, 11.0% for aspirin alone, 11.4% for extended-release dipyridamole alone and 12.3% for placebo alone. The differences between the AGGRENOX, aspirin alone and extended-release dipyridamole alone treatment groups were not statistically significant. These incidence rates for AGGRENOX and aspirin alone are consistent with previous aspirin studies in stroke and TIA patients.
INDICATIONS AND USAGE
Aggrenox® (aspirin/extended-release dipyridamole) capsules is indicated to reduce the risk of stroke in patients who have had transient ischemia of the brain or completed ischemic stroke due to thrombosis.
CONTRAINDICATIONS
AGGRENOX capsules is contraindicated in patients with hypersensitivity to dipyridamole, aspirin or any of the other product components.
Allergy
Aspirin is contraindicated in patients with known allergy to nonsteroidal anti-inflammatory drug products and in patients with the syndrome of asthma, rhinitis, and nasal polyps. Aspirin may cause severe urticaria, angioedema or bronchospasm (asthma).
Reye's Syndrome
Aspirin should not be used in children or teenagers for viral infections, with or without fever, because of the risk of Reye's syndrome with concomitant use of aspirin in certain viral illnesses.
WARNINGS
Alcohol Warning
Patients who consume three or more alcoholic drinks every day should be counseled about the bleeding risks involved with chronic, heavy alcohol use while taking aspirin.
Coagulation Abnormalities
Even low doses of aspirin can inhibit platelet function leading to an increase in bleeding time. This can adversely affect patients with inherited or acquired (liver disease or vitamin K deficiency) bleeding disorders.
Gastrointestinal (GI) Side Effects
GI side effects include stomach pain, heartburn, nausea, vomiting, and gross GI bleeding. Although minor upper GI symptoms, such as dyspepsia, are common and can occur anytime during therapy, physicians should remain alert for signs of ulceration and bleeding, even in the absence of previous GI symptoms. Physicians should inform patients about the signs and symptoms of GI side effects and what steps to take if they occur.
Peptic Ulcer Disease
Patients with a history of active peptic ulcer disease should avoid using aspirin, which can cause gastric mucosal irritation, and bleeding.
Pregnancy
Aggrenox® (aspirin/extended-release dipyridamole) capsules can cause fetal harm when administered to a pregnant woman. Maternal aspirin use during later stages of pregnancy may cause low birth weight, increased incidence for intracranial hemorrhage in premature infants, stillbirths and neonatal death. Because of the above and because of the known effects of nonsteroidal anti-inflammatory drugs (NSAIDs) on the fetal cardiovascular system (closure of the ductus arteriosus), AGGRENOX capsules should be avoided in the third trimester of pregnancy.
Aspirin has been shown to be teratogenic in rats (spina bifida, exencephaly, microphthalmia and coelosomia) and rabbits (congested fetuses, agenesis of skull and upper jaw, generalized edema with malformation of the head, and diaphanous skin) at oral doses of 330 mg/kg/day and 110 mg/kg/day, respectively. These doses, which also resulted in a high resorption rate in rats (63% of implantations versus 5% in controls), are, on a mg/m2 basis, about 66 and 44 times, respectively, the dose of aspirin contained in the maximum recommended daily human dose of Aggrenox® (aspirin/extended-release dipyridamole) capsules. Reproduction studies with dipyridamole have been performed in mice, rabbits and rats at oral doses of up to 125 mg/kg, 40 mg/kg and 1000 mg/kg, respectively (about 1½, 2 and 25 times the maximum recommended daily human oral dose, respectively, on a mg/m2 basis) and have revealed no evidence of harm to the fetus due to dipyridamole. When 330 mg aspirin/kg/day was combined with 75 mg dipyridamole/kg/day in the rat, the resorption rate approached 100%, indicating potentiation of aspirin-related fetal toxicity. There are no adequate and well-controlled studies in pregnant women. If AGGRENOX capsules is used during pregnancy, or if the patient becomes pregnant while taking AGGRENOX capsules, the patient should be apprised of the potential hazard to the fetus.
PRECAUTIONS
General
AGGRENOX capsules is not interchangeable with the individual components of aspirin and Persantine® Tablets.
Coronary Artery Disease
Dipyridamole has a vasodilatory effect and should be used with caution in patients with severe coronary artery disease (e.g., unstable angina or recently sustained myocardial infarction). Chest pain may be aggravated in patients with underlying coronary artery disease who are receiving dipyridamole.
For stroke or TIA patients for whom aspirin is indicated to prevent recurrent myocardial infarction (MI) or angina pectoris, the aspirin in this product may not provide adequate treatment for the cardiac indications.
Hepatic Insufficiency
Elevations of hepatic enzymes and hepatic failure have been reported in association with dipyridamole administration.
Hypotension
Dipyridamole should be used with caution in patients with hypotension since it can produce peripheral vasodilation.
Renal Failure
Avoid aspirin in patients with severe renal failure (glomerular filtration rate less than 10 mL/minute).
Risk of Bleeding
In ESPS2 the incidence of gastrointestinal bleeding was 68 patients (4.1%) in the AGGRENOX group, 36 patients (2.2%) in the extended-release dipyridamole group, 52 patients (3.2%) in the aspirin group, and 34 patients (2.1%) in the placebo groups.
The incidence of intracranial hemorrhage was 9 patients (0.6%) in the AGGRENOX group, 6 patients (0.5%) in the extended-release dipyridamole group, 6 patients (0.4%) in the aspirin group and 7 patients (0.4%) in the placebo groups.
Laboratory Tests
Aspirin has been associated with elevated hepatic enzymes, blood urea nitrogen and serum creatinine, hyperkalemia, proteinuria and prolonged bleeding time.
Dipyridamole has been associated with elevated hepatic enzymes.
Drug Interactions
No pharmacokinetic drug-drug interaction studies were conducted with the AGGRENOX formulation. The following information was obtained from the literature.
Adenosine: Dipyridamole has been reported to increase the plasma levels and cardiovascular effects of adenosine. Adjustment of adenosine dosage may be necessary.
Angiotensin Converting Enzyme (ACE) Inhibitors: Due to the indirect effect of aspirin on the renin-angiotensin conversion pathway, the hyponatremic and hypotensive effects of ACE inhibitors may be diminished by concomitant administration of aspirin.
Acetazolamide: Concurrent use of aspirin and acetazolamide can lead to high serum concentrations of acetazolamide (and toxicity) due to competition at the renal tubule for secretion.
Anticoagulant Therapy (heparin and warfarin): Patients on anticoagulation therapy are at increased risk for bleeding because of drug-drug interactions and effects on platelets. Aspirin can displace warfarin from protein binding sites, leading to prolongation of both the prothrombin time and the bleeding time. Aspirin can increase the anticoagulant activity of heparin, increasing bleeding risk.
Anticonvulsants: Salicylic acid can displace protein-bound phenytoin and valproic acid, leading to a decrease in the total concentration of phenytoin and an increase in serum valproic acid levels.
Beta Blockers: The hypotensive effects of beta blockers may be diminished by the concomitant administration of aspirin due to inhibition of renal prostaglandins, leading to decreased renal blood flow and salt and fluid retention.
Cholinesterase Inhibitors: Dipyridamole may counteract the anticholinesterase effect of cholinesterase inhibitors, thereby potentially aggravating myasthenia gravis.
Diuretics: The effectiveness of diuretics in patients with underlying renal or cardiovascular disease may be diminished by the concomitant administration of aspirin due to inhibition of renal prostaglandins, leading to decreased renal blood flow and salt and fluid retention.
Methotrexate: Salicylate can inhibit renal clearance of methotrexate, leading to bone marrow toxicity, especially in the elderly or renal impaired.
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs): The concurrent use of aspirin with other NSAIDs may increase bleeding or lead to decreased renal function.
Oral Hypoglycemics: Moderate doses of aspirin may increase the effectiveness of oral hypoglycemic drugs, leading to hypoglycemia.
Uricosuric Agents (probenecid and sulfinpyrazone): Salicylates antagonize the uricosuric action of uricosuric agents.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In studies in which dipyridamole was administered in the feed to mice (up to 111 weeks in males and females) and rats (up to 128 weeks in males and up to 142 weeks in females), there was no evidence of drug-related carcinogenesis. The highest dose administered in these studies (75 mg/kg/day) was, on a mg/m2 basis, about equivalent to the maximum recommended daily human oral dose (MRHD) in mice and about twice the MRHD in rats.
Combinations of dipyridamole and aspirin (1:5 ratio) tested negative in the Ames test, in vivo chromosome aberration tests (in mice and hamsters), oral micronucleus tests (in mice and hamsters) and oral dominant lethal test (in mice). Aspirin, alone, induced chromosome aberrations in cultured human fibroblasts. Mutagenicity tests of dipyridamole alone with bacterial and mammalian cell systems were negative.
Combinations of dipyridamole and aspirin have not been evaluated for effects on fertility and reproductive performance. There was no evidence of impaired fertility when dipyridamole was administered to male and female rats at oral doses up to 500 mg/kg/day (about 12 times the MRHD on a mg/m2 basis). A significant reduction in number of corpora lutea with consequent reduction in implantations and live fetuses was, however, observed at 1250 mg/kg (more than 30 times the MRHD on a mg/m2 basis). Aspirin inhibits ovulation in rats.
Pregnancy
Teratogenic Effects: Pregnancy Category D. (see WARNINGS)
Labor and Delivery
Aspirin can result in excessive blood loss at delivery as well as prolonged gestation and prolonged labor. Because of these effects on the mother and because of adverse fetal effects seen with aspirin during the later stages of pregnancy (see WARNINGS, Pregnancy), Aggrenox® (aspirin/extended-release dipyridamole) capsules should be avoided in the third trimester of pregnancy and during labor and delivery.
Nursing Mothers
Both dipyridamole and aspirin are excreted in human milk. Caution should be exercised when AGGRENOX capsules is administered to a nursing woman.
Pediatric Use
Safety and effectiveness of AGGRENOX capsules in pediatric patients have not been studied. Due to the aspirin component, use of this product in the pediatric population is not recommended (see CONTRAINDICATIONS).
ADVERSE REACTIONS
A 24-month, multicenter, double-blind, randomized study (ESPS2) was conducted to compare the efficacy and safety of AGGRENOX capsules with placebo, extended-release dipyridamole alone and aspirin alone. The study was conducted in a total of 6602 male and female patients who had experienced a previous ischemic stroke or transient ischemia of the brain within three months prior to randomization.
Table 2 presents the incidence of adverse events that occurred in 1% or more of patients treated with AGGRENOX capsules where the incidence was also greater than in those patients treated with placebo. There is no clear benefit of the dipyridamole/aspirin combination over aspirin with respect to safety.
| Individual Treatment Group | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Body System/Preferred Term | AGGRENOX | ER-DP Alone | ASA Alone | Placebo | |||||
| * Reported by ≥1% of patients during AGGRENOX treatment where the incidence was greater than in those treated with placebo. | |||||||||
| Note: ER-DP = extended-release dipyridamole 200 mg; ASA = aspirin 25 mg. The dosage regimen for all treatment groups is BID NOS = not otherwise specified. | |||||||||
| Total Number of Patients | 1650 | 1654 | 1649 | 1649 | |||||
| Total Number (%) of Patients With at Least One On-Treatment Adverse Event |
1319 |
(79.9%) |
1305 |
(78.9%) |
1323 |
(80.2%) |
1304 |
(79.1%) |
|
| Central & Peripheral Nervous System Disorders | |||||||||
| Headache | 647 | (39.2%) | 634 | (38.3%) | 558 | (33.8%) | 543 | (32.9%) | |
| Convulsions | 28 | (1.7%) | 15 | ( 0.9%) | 28 | ( 1.7%) | 26 | (1.6%) | |
| Gastro-Intestinal System Disorders | |||||||||
| Dyspepsia | 303 | (18.4%) | 288 | (17.4%) | 299 | (18.1%) | 275 | (16.7%) | |
| Abdominal Pain | 289 | (17.5%) | 255 | (15.4%) | 262 | (15.9%) | 239 | (14.5%) | |
| Nausea | 264 | (16.0%) | 254 | (15.4%) | 210 | (12.7%) | 232 | (14.1%) | |
| Diarrhea | 210 | (12.7%) | 257 | (15.5%) | 112 | ( 6.8%) | 161 | (9.8%) | |
| Vomiting | 138 | ( 8.4%) | 129 | ( 7.8%) | 101 | ( 6.1%) | 118 | (7.2%) | |
| Hemorrhage Rectum | 26 | (1.6%) | 22 | (1.3%) | 16 | (1.0%) | 13 | (0.8%) | |
| Melena | 31 | (1.9%) | 10 | (0.6%) | 20 | (1.2%) | 13 | (0.8%) | |
| Hemorrhoids | 16 | (1.0%) | 13 | (0.8%) | 10 | (0.6%) | 10 | (0.6%) | |
| GI Hemorrhage | 20 | (1.2%) | 5 | (0.3%) | 15 | (0.9%) | 7 | (0.4%) | |
| Body as a Whole - General Disorders | |||||||||
| Pain | 105 | ( 6.4%) | 88 | ( 5.3%) | 103 | ( 6.2%) | 99 | (6.0%) | |
| Fatigue | 95 | ( 5.8%) | 93 | ( 5.6%) | 97 | ( 5.9%) | 90 | (5.5%) | |
| Back Pain | 76 | ( 4.6%) | 77 | ( 4.7%) | 74 | ( 4.5%) | 65 | (3.9%) | |
| Accidental Injury | 42 | ( 2.5%) | 24 | ( 1.5%) | 51 | ( 3.1%) | 37 | (2.2%) | |
| Malaise | 27 | ( 1.6%) | 23 | (1.4%) | 26 | (1.6%) | 22 | (1.3%) | |
| Asthenia | 29 | ( 1.8%) | 19 | ( 1.1%) | 17 | ( 1.0%) | 18 | (1.1%) | |
| Syncope | 17 | ( 1.0%) | 13 | ( 0.8%) | 16 | ( 1.0%) | 8 | (0.5%) | |
| Psychiatric Disorders | |||||||||
| Amnesia | 39 | ( 2.4%) | 40 | ( 2.4%) | 57 | (3.5%) | 34 | (2.1%) | |
| Confusion | 18 | ( 1.1%) | 9 | (0.5%) | 22 | (1.3%) | 15 | (0.9%) | |
| Anorexia | 19 | ( 1.2%) | 17 | (1.0%) | 10 | (0.6%) | 15 | (0.9%) | |
| Somnolence | 20 | ( 1.2%) | 13 | (0.8%) | 18 | ( 1.1%) | 9 | (0.5%) | |
| Musculoskeletal System Disorders | |||||||||
| Arthralgia | 91 | ( 5.5%) | 75 | ( 4.5%) | 91 | (5.5%) | 76 | (4.6%) | |
| Arthritis | 34 | ( 2.1%) | 25 | (1.5%) | 17 | ( 1.0%) | 19 | (1.2%) | |
| Arthrosis | 18 | ( 1.1%) | 22 | ( 1.3%) | 13 | (0.8%) | 14 | (0.8%) | |
| Myalgia | 20 | ( 1.2%) | 16 | (1.0%) | 11 | (0.7%) | 11 | (0.7%) | |
| Respiratory System Disorders | |||||||||
| Coughing | 25 | ( 1.5%) | 18 | (1.1%) | 32 | (1.9%) | 21 | (1.3%) | |
| Upper Respiratory Tract Infection | 16 | ( 1.0%) | 9 | (0.5%) | 16 | (1.0%) | 14 | (0.8%) | |
| Cardiovascular Disorders, General | |||||||||
| Cardiac Failure | 26 | ( 1.6%) | 17 | (1.0%) | 30 | ( 1.8%) | 25 | (1.5%) | |
| Platelet, Bleeding & Clotting Disorders | |||||||||
| Hemorrhage NOS | 52 | (3.2%) | 24 | (1.5%) | 46 | (2.8%) | 24 | (1.5%) | |
| Epistaxis | 39 | ( 2.4%) | 16 | (1.0%) | 45 | (2.7%) | 25 | (1.5%) | |
| Purpura | 23 | (1.4%) | 8 | (0.5%) | 9 | (0.5%) | 7 | (0.4%) | |
| Neoplasm | |||||||||
| Neoplasm NOS | 28 | (1.7%) | 16 | (1.0%) | 23 | (1.4%) | 20 | (1.2%) | |
| Red Blood Cell Disorders | |||||||||
| Anemia | 27 | (1.6%) | 16 | ( 1.0%) | 19 | ( 1.2%) | 9 | (0.5%) | |
Discontinuation due to adverse events in ESPS2 was 25% for AGGRENOX, 25% for extended-release dipyridamole, 19% for aspirin, and 21% for placebo (refer to Table 3).
| Treatment Groups | ||||||||
|---|---|---|---|---|---|---|---|---|
| AGGRENOX | ER-DP | ASA | Placebo | |||||
| Note: ER-DP = extended-release dipyridamole 200 mg; ASA = aspirin 25 mg. The dosage regimen for all treatment groups is BID | ||||||||
| Total Number of Patients | 1650 | 1654 | 1649 | 1649 | ||||
| Patients with at least one Adverse Event that led to treatment discontinuation |
417 |
(25%) |
419 |
(25%) |
318 |
(19%) |
352 |
(21%) |
| Headache | 165 | (10%) | 166 | (10%) | 57 | (3%) | 69 | (4%) |
| Dizziness | 85 | (5%) | 97 | (6%) | 69 | (4%) | 68 | (4%) |
| Nausea | 91 | (6%) | 95 | (6%) | 51 | (3%) | 53 | (3%) |
| Abdominal Pain | 74 | (4%) | 64 | (4%) | 56 | (3%) | 52 | (3%) |
| Dyspepsia | 59 | (4%) | 61 | (4%) | 49 | (3%) | 46 | (3%) |
| Vomiting | 53 | (3%) | 52 | (3%) | 28 | (2%) | 24 | (1%) |
| Diarrhea | 35 | (2%) | 41 | (2%) | 9 | (<1%) | 16 | (<1%) |
| Stroke | 39 | (2%) | 48 | (3%) | 57 | (3%) | 73 | (4%) |
| Transient Ischemic Attack | 35 | (2%) | 40 | (2%) | 26 | (2%) | 48 | (3%) |
| Angina Pectoris | 23 | (1%) | 20 | (1%) | 16 | (<1%) | 26 | (2%) |
Headache was most notable in the first month of treatment.
Other Adverse Events
Adverse reactions that occurred in less than 1% of patients treated with Aggrenox® (aspirin/extended-release dipyridamole) capsules in the ESPS2 study and that were medically judged to be possibly related to either dipyridamole or aspirin are listed below (see WARNINGS).
Body as a Whole: Allergic reaction, fever
Cardiovascular: Hypotension
Central Nervous System: Coma, dizziness, paresthesia, cerebral hemorrhage, intracranial hemorrhage, subarachnoid hemorrhage
Gastrointestinal: Gastritis, ulceration and perforation
Hearing and Vestibular Disorders: Tinnitus, and deafness. Patients with high frequency hearing loss may have difficulty perceiving tinnitus. In these patients, tinnitus cannot be used as a clinical indicator of salicylism
Heart Rate and Rhythm Disorders: Tachycardia, palpitation, arrhythmia, supraventricular tachycardia
Liver and Biliary System Disorders: Cholelithiasis, jaundice, hepatic function abnormal
Metabolic and Nutritional Disorders: Hyperglycemia, thirst
Platelet, Bleeding and Clotting Disorders: Hematoma, gingival bleeding
Psychiatric Disorders: Agitation
Reproductive: Uterine hemorrhage
Respiratory: Hyperpnea, asthma, bronchospasm, hemoptysis, pulmonary edema
Special Senses Other Disorders: Taste loss
Skin and Appendages Disorders: Pruritus, urticaria
Urogenital: Renal insufficiency and failure, hematuria
Vascular (Extracardiac) Disorders: Flushing
The following is a list of additional adverse reactions that have been reported either in the literature or are from postmarketing spontaneous reports for either dipyridamole or aspirin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure.
Body as a Whole: Hypothermia, chest pain
Cardiovascular: Angina pectoris
Central Nervous System: Cerebral edema
Fluid and Electrolyte: Hyperkalemia, metabolic acidosis, respiratory alkalosis, hypokalemia
Gastrointestinal: Pancreatitis, Reye's syndrome, hematemesis
Hearing and Vestibular Disorders: Hearing loss
Hypersensitivity: Acute anaphylaxis, laryngeal edema
Liver and Biliary System Disorders: Hepatitis, hepatic failure
Musculoskeletal: Rhabdomyolysis
Metabolic and Nutritional Disorders: Hypoglycemia, dehydration
Platelet, Bleeding and Clotting Disorders: Prolongation of the prothrombin time, disseminated intravascular coagulation, coagulopathy, thrombocytopenia
Reproductive: Prolonged pregnancy and labor, stillbirths, lower birth weight infants, antepartum and postpartum bleeding
Respiratory: Tachypnea, dyspnea
Skin and Appendages Disorders: Rash, alopecia, angioedema, Stevens-Johnson syndrome, skin hemorrhages such as bruising, ecchymosis, and hematoma
Urogenital: Interstitial nephritis, papillary necrosis, proteinuria
Vascular (Extracardiac Disorders): Allergic vasculitis
Other adverse events: anorexia, aplastic anemia, migraine, pancytopenia, thrombocytosis.
Laboratory Changes
Over the course of the 24-month study (ESPS2), patients treated with AGGRENOX showed a decline (mean change from baseline) in hemoglobin of 0.25 g/dL, hematocrit of 0.75%, and erythrocyte count of 0.13x106/mm3.
OVERDOSAGE
Because of the dose ratio of dipyridamole to aspirin, overdosage of Aggrenox® (aspirin/extended-release dipyridamole) capsules is likely to be dominated by signs and symptoms of dipyridamole overdose. In case of real or suspected overdose, seek medical attention or contact a Poison Control Center immediately. Careful medical management is essential.
Dipyridamole
Based upon the known hemodynamic effects of dipyridamole, symptoms such as warm feeling, flushes, sweating, restlessness, feeling of weakness and dizziness may occur. A drop in blood pressure and tachycardia might also be observed.
Symptomatic treatment is recommended, possibly including a vasopressor drug. Gastric lavage should be considered. Administration of xanthine derivatives (e.g., aminophylline) may reverse the hemodynamic effects of dipyridamole overdose. Since dipyridamole is highly protein bound, dialysis is not likely to be of benefit.
Aspirin
Salicylate toxicity may result from acute ingestion (overdose) or chronic intoxication. The early signs of salicylic overdose (salicylism), including tinnitus (ringing in the ears), occur at plasma concentrations approaching 200 μg/mL. Plasma concentrations of aspirin above 300 μg/mL are clearly toxic. Severe toxic effects are associated with levels above 400 μg/mL. A single lethal dose of aspirin in adults is not known with certainty but death may be expected at 30 g.
Treatment consists primarily of supporting vital functions, increasing salicylate elimination, and correcting the acid-base disturbance. Gastric emptying and/or lavage are recommended as soon as possible after ingestion, even if the patient has vomited spontaneously. After lavage and/or emesis, administration of activated charcoal, as a slurry, is beneficial, if less than 3 hours have passed since ingestion. Charcoal absorption should not be employed prior to emesis and lavage.
Severity of aspirin intoxication is determined by measuring the blood salicylate level. Acid-base status should be closely followed with serial blood gas and serum pH measurements. Fluid and electrolyte balance should also be maintained.
In severe cases, hyperthermia and hypovolemia are the major immediate threats to life. Children should be sponged with tepid water. Replacement fluid should be administered intravenously and augmented with correction of acidosis. Plasma electrolytes and pH should be monitored to promote alkaline diuresis of salicylate if renal function is normal. Infusion of glucose may be required to control hypoglycemia.
Hemodialysis and peritoneal dialysis can be performed to reduce the body drug content. In patients with renal insufficiency or in cases of life-threatening intoxication, dialysis is usually required. Exchange transfusion may be indicated in infants and young children.
DOSAGE AND ADMINISTRATION
The recommended dose of Aggrenox® (aspirin/extended-release dipyridamole) capsules is one capsule given orally twice daily, one in the morning and one in the evening. The capsules should be swallowed whole without chewing. AGGRENOX capsules may be administered with or without food.
Alternative Regimen in Case of Intolerable Headaches
In the event of intolerable headaches during initial treatment, switch to one capsule at bedtime and low-dose aspirin in the morning. Because there are no outcome data with this regimen and headaches become less of a problem as treatment continues, patients should return to the usual regimen as soon as possible, usually within one week.
AGGRENOX capsules is not interchangeable with the individual components of aspirin and Persantine® Tablets.
HOW SUPPLIED
AGGRENOX capsules is available as a hard gelatin capsule, with a red cap and an ivory-colored body, 24.0 mm in length, containing yellow extended-release pellets incorporating dipyridamole and a round white tablet incorporating immediate-release aspirin. The capsule body is imprinted in red with the Boehringer Ingelheim logo and with “01A”.
AGGRENOX capsules is supplied in unit-of-use bottles of 60 capsules (NDC 0597-0001-60).
Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature]. Protect from excessive moisture.
Marketed by:
Boehringer Ingelheim Pharmaceuticals Inc., Ridgefield, CT 06877 USA
Manufactured by:
Boehringer Ingelheim Pharma GmbH & Co. KG, Biberach, Germany
Licensed from:
Boehringer Ingelheim International GmbH
©Copyright Boehringer Ingelheim International GmbH
2007, ALL RIGHTS RESERVED
Patent No. 6,015,577
January 31, 2007
OT1000D
42633/US/7
| Aggrenox (aspirin and dipyridamole) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Revised: 06/2007