KERAFOAM
-
urea aerosol, foam
Onset Therapeutics, LLC
----------
Kerafoam
For softening, smoothing and removing rough scaling hyperkeratotic skin
DESCRIPTION:
KERAFOAM® Emollient Foam is a keratolytic
emollient foam which is a tissue softener for skin and/or nails. KERAFOAM Emollient
Foam contains 30% urea USP in an aqueous based emollient foam vehicle. Each
gram of KERAFOAM Emollient Foam contains 30% urea USP, ammonium lactate, cetyl
alcohol NF, emulsifying wax NF, methylparaben NF, propylene glycol USP, propylparaben
NF, purified water USP, steareth-10. Also contains: Propellant HFA-134a
(1,1,1,2-tetrafluoroethane).
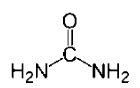
Urea USP is a diamide of carbonic acid with the
following chemical structure:
CLINICAL PHARMACOLOGY
Urea gently lyses/dissolves the intercellular matrix of surface skin cells loosening and allowing a shedding of rough, thickened and scaly hyperkeratotic skin. Urea also moisturizes and softens skin.
INDICATIONS
For softening, smoothing and removing rough scaling hyperkeratotic skin in conditions such as xerosis, ichthyosis, skin cracks and fissures, dermatitis, eczema, psoriasis, keratoses and calluses.
CONTRAINDICATIONS
Known hypersensitivity to any of the listed ingredients. Discontinue use if hypersensitivity is observed.
WARNINGS
FOR EXTERNAL USE ONLY. Avoid contact with eyes, lips or mucous membranes. Keep out of the reach of children. Contents under pressure. Do not puncture or incinerate container. Do not expose to temperatures above 120ºF (49ºC).
PRECAUTIONS
Information for patients:
This medication is to be used as directed by a physician and should not be used to treat any condition other than that for which it was prescribed. If redness or irritation occurs, discontinue use.
Pregnancy
Pregnancy Category C. Animal reproduction studies have not been conducted with KERAFOAM Emollient Foam. It is not known whether KERAFOAM Emollient Foam can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. KERAFOAM Emollient Foam should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether or not this drug is secreted in human milk. Because many drugs are secreted in human milk, caution should be exercised when KERAFOAM Emollient Foam is administered to a nursing woman.
ADVERSE REACTIONS
Transient stinging, burning, itching or irritation may occur and normally disappear on discontinuing the medication.
DOSAGE AND ADMINISTRATION
Shake Vigorously, Tap Bottom of Can, and Prime Before
Initial Use. Shake Vigorously and Tap Before Each Use.
To Prime: After shaking, gently tap bottom of can onto palm of other hand or a solid
surface at least 3 times. Hold the can upright, direct away from the patient, and
firmly depress the actuator for 1 to 3 seconds or until foam begins to dispense. (If
foam does not dispense within 3 seconds: re-shake can, gently tap bottom of can onto a
solid surface at least 3 times, and depress the actuator again until foam begins to
dispense.)
Before Each Use: Shake vigorously and gently tap bottom of can onto palm of other hand or a
solid surface at least 3 times.
During Use: Holding can upright, dispense KERAFOAM into palm of hand and apply
to affected area twice per day, or as directed by a physician. Rub in until completely
absorbed. Wipe off any excess foam from actuator after use.
HOW SUPPLIED
KERAFOAM Emollient Foam is supplied in a 60 g (NDC# 16781-157-60) aluminum can.
Store at room temperature: 59º - 77ºF (15º - 25ºC). Protect from freezing. Store upright.
Patent Pending
P/N 2604 Rev. 3
Manufactured For:

Onset Therapeutics
Cumberland, RI 02864
(888) 713-8154
www.onsettx.com
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - Kerafoam Outer Carton - 5g
NDC 16781-157-06
Rx Only
Kerafoam®
Emollient Foam
Urea (30%)
For softening, smoothing, and removing rough scaling hyperkeratotic skin
See prescribing information enclosed
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - Kerafoam Inner Label - 5g
NDC 16781-157-06
Rx Only
Professional Sample
Not for Sale
Net Weight
5g
Kerafoam®
Emollient Foam
Urea (30%)
Sample will not dispense entire
contents.
| KERAFOAM
urea aerosol, foam |
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| Unapproved drug other | 12/01/2006 | ||
| Labeler - Onset Therapeutics, LLC (793223707) |
| Registrant - Onset Therapeutics, LLC (964275155) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Onset Therapeutics, LLC | 793223707 | Manufacture | |