GUAIFENESIN DM
-
guaifenesin and
dextromethorphan hydrobromide tablet
Prasco Laboratories
----------
Guaifenesin DMTablets
11 DESCRIPTION
Each extended-release tablet contains:
Guaifenesin. . . . . . . 600 mg
Dextromethorphan Hydrobromide. . . . 30 mg
Inactive ingredients: calcium phosphate, D&C Yellow #10, FD&C Blue #1, magnesium stearate, methylcellulose, povidone, stearic acid.
Chemically, guaifenesin is 1,2-propanediol, 3-(2-methoxyphenoxy)-,(±)-. It has the following structural formula:
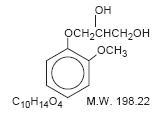
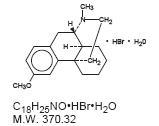
Dextromethorphan Hydrobromide is a salt of the methyl ether of the dextrorotatory isomer of levorphanol, a narcotic analgesic. Chemically, it is Morphinan 3-methoxy-17-methyl-,(9α, 13α, 14α,)-, hydrobromide monohydrate and has the following structural formula:
12 CLINICAL PHARMACOLOGY
Guaifenesin is an expectorant that increases respiratory tract fluid secretions and helps loosen phlegm and bronchial secretions. By reducing the viscosity of secretions, guaifenesin increases the efficiency of the mucocilary mechanism in removing accumulated secretions from the upper and lower airways. Guaifenesin is readily absorbed from the gastrointestinal tract and is rapidly metabolized and excreted in the urine. Guaifenesin has a plasma half-life of 1 hour. The major urinary metabolite is β-(2-methoxyphenoxy) lactic acid.
Dextromethorphan is an antitussive agent which, unlike the isometric levorphanol, has no analgesic properties or addictive properties. The drug acts centrally and elevates the threshold for coughing. It is about equal to codeine in depressing the cough reflex. In therapeutic dosage, dextromethorphan does not inhibit ciliary activity. Dextromethorphan is rapidly absorbed from the gastrointestinal tract, metabolized by the liver, and is excreted primarily in the urine.
1 INDICATIONS & USAGE
This product is indicated for the temporary relief of coughs associated with upper and lower respiratory tract infections and related conditions such as sinusitis, pharyngitis, and bronchitis, particularly when these conditions are complicated by tenacious mucus and/or mucus plugs and congestion. The product is effective in a productive as well as a non-productive cough that tends to injure the mucus membrane of the air passages.
4 CONTRAINDICATIONS
This drug is contraindicated in patients with hypersensitivity to guaifenesin or dextromethorphan and in patients receiving monoamine oxidase inhibitor (MAOI) therapy and for 14 days after stopping MAOI therapy. (See drug interactions section).
Risk-benefit should be considered when the following medical problem exists : asthma (may increase airway resistance) and hepatic function impairment (metabolism of dextromethorphan may be impaired).
WARNINGS
Before prescribing medication to suppress or modify a cough, it is important that the underlying cause of the cough is identified, that modification of the cough does not increase the risk of clinical or physiological complications, and that appropriate therapy for the primary disease is instituted.
PRECAUTIONS
GENERAL PRECAUTIONS
Dextromethorphan should be used with caution in sedated or debilitated patients, and in patients confined to the supine position. Administration of dextromethorphan may be accompanied by histamine release and should be used with caution in atopic children.
17 INFORMATION FOR PATIENTS
If cough has not improved after 7 days, or is accompanied by high fever, skin rash, or continuing headache, check with your doctor. These signs may indicate other medical problems.
Patient consultation should include the following information regarding the proper use of this medication:
•Do not take more medication than the amount recommended.
•If a dose is missed, the medication should be taken as soon as possible unless it is almost time for the next dose, not doubling doses.
•Swallow sustained release tablets whole. Do not crush or chew tablets.
•To help loosen mucus in the lungs, patient should drink a glass of water after each dose.
7 DRUG INTERACTIONS
patients should be warned not to use this product if they are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug.
DRUG & OR LABORATORY TEST INTERACTIONS
Guaifenesin may increase renal clearance for urate and thereby lower serum uric acid levels. Guaifenesin may produce an increase in 5-hydroxyindoleacetic acid and may therefore interfere with the interpretation of this test for the diagnosis of carcinoid syndrome. It may also falsely elevate the VMA test for catechols. Administration of this product should be discontinued 48 hours prior to the collection of urine specimens for such tests.
1 CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
No long term studies have been conducted to evaluate the carcinogenic or mutagenic potential of this product.
1 PREGNANCY
Category C: Animal reproduction studies have not been conducted with this product. It is also not known whether this product can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Therefore, this product should be given to a pregnant woman only if clearly needed.
3 NURSING MOTHERS
It is not known whether this drug is excreted in human milk.
Caution should be exercised when this product is administered to a nursing woman and a decision should be made whether to discontinue nursing or discontinue taking the drug, taking into account the importance of the drug to the mother.
4 PEDIATRIC USE
Appropriate studies on the relationship of age to the effects of guaifenesin and dextromethorphan have not been conducted in the pediatric population. However, no pediatric specific problems have been documented to date.
5 GERIATRIC USE
Appropriate studies on the relationship of age to the effects of guaifenesin have not been conducted in the geriatric population. However, no geriatric-specific problems have been documented to date. No information is available on the relationship of age to the effects of dextromethorphan in geriatric patients.
6 ADVERSE REACTIONS
Signs of potential side effects that require medical attention only if persistant include, but are not limited to: mild dizziness, mild drowsiness, nausea or vomiting, stomach pain.
Those indicating immediate medical attention include confusion, drowsiness or dizziness (severe), nausea or vomiting (severe), unusual excitement, nervousness, restlessness, or irritability (severe).
10 OVERDOSAGE
Signs and Symptoms:
Overdosage with guaifenesin is unlikely to produce toxic effects since its toxicity is low. Guaifenesin, when administered by stomach tube to test animals in doses up to 5 gm/kg, produced no signs of toxicity. Overdosage with dextromethorphan may produce central nervous system excitement and mental confusion. Very high doses may produce respiratory depression. One case of toxic psychosis (hyperactivity, marked visual and auditory hallucinations) after an ingestion of a single 300 mg dose of dextromethorphan has been reported.
Recommended Treatment:
In severe overdosage treatment should be aimed at reducing further absorption of the drug. Gastric emptying (Syrup of Ipecac) and/or lavage is recommended as soon as possible after ingestion.
2 DOSAGE & ADMINISTRATION
Adults and adolescents: 1 or 2 tablets every 12 hours not to exceed 4 tablets in
24 hours.
Children 6 to 12 years: 1 tablet every 12 hours not to exceed 2 tablets in 24 hours. Children 2 to 6 years: 1/2 tablet every 12 hours not to exceed 1 tablet in 24 hours.
16 HOW SUPPLIED
supplied as green, capsule-shaped tablets scored and debossed “310” on top, Prasco Laboratories logo on bottom. Bottles of 100 tablets, NDC 66993-310-02.
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN. IN
CASE OF ACCIDENTAL OVERDOSE, SEEK PROFESSIONAL ASSISTANCE OR CONTACT A POISON CONTROL CENTER IMMEDIATELY.
Dispense in tight, light-resistant containers as defined in the USP-NF.
Store at controlled room temperature 15°- 30°C (59° - 86°F).
Manufactured For:
PRASCO LABORATORIES
Cincinnati, OH 45249
by:
Sovereign Pharmaceuticals, Ltd.
Fort Worth, TX 76118
PIPRAS-10
Iss. 3/2002
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL

| GUAIFENESIN
DM
guaifenesin, dextromethorphan hydrobromide tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| Unapproved drug other | 12/01/2010 | 01/20/2011 | |
| Labeler - Prasco Laboratories (065969375) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Sovereign Pharmaceuticals, Ltd. | 623168267 | MANUFACTURE | |