DRIHIST SR
-
phenylephrine hydrochloride,
chlorpheniramine maleate and
methscopolamine nitrate tablet
Prasco Laboratories
----------
DriHist SR Tablets11 DESCRIPTION
Each long-acting tablet contains:
Phenylephrine HCl……………20 mg
Chlorpheniramine Maleate……..8 mg
Methscopolaimine Nitrate……2.5 mg
Inactive ingredients: calcium phosphate, magnesium stearate, methylcellulose, povidone, stearic acid.
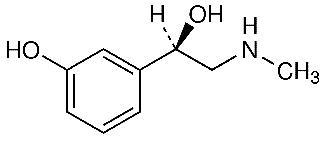
Phenylephrine HCl is a sympathomimetic amine with the chemical structure: Benzenemethanol, 3-hydroxy-α{(methylamino)methyl}, hydrochloride.
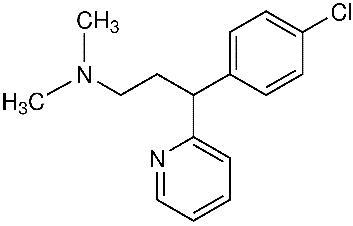
Chlorpheniramine Maleate is an antihistamine with the chemical structure 2-Pyridinepropanamine, γ-(4-chlorphenyl)-N,N-dimethyl-, (Z)-2-butenedioate (1:1).
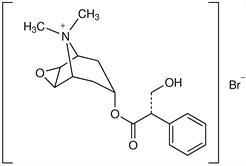
Methscopolamine Nitrate is an anticholinergic belladonna alkaloid derivative with the chemical structure: 3-Oxa-9-azoniatricyclo(3.3.1.02,4)nonane, 7-(3-hydroxy-1-oxo-2-phenylpropoxy)-9,9-dimethyl-, nitrate. [7(s)-1α,2β-5α-7β].
12 CLINICAL PHARMACOLOGY
Phenylephrine, a sympathomimetic amine acts directly on α-adrenergic receptors in the mucosa of the respiratory tract to produce vasoconstriction that increases peripheral resistance, resulting in an increase in both systolic and diastolic blood pressure. Accompanying the pressor response is a marked reflex bradycardia due to increased vagal activity. It produces vasoconstriction that lasts longer than that produced by ephedrine and epinephrine, and in therapeutic doses, produces little or no central nervous system (CNS) stimulation. Phenylephrine has reduced bioavailability from the gastrointestinal tract because of first pass metabolism by the liver.
Chlorpheniramine maleate competitively antagonizes most of the smooth muscle stimulating actions of histamine on the H1 receptors of the GI tract, uterus, large blood vessels, and bronchial muscle. It also antagonizes the action of histamine that results in increased capillary permeability and the formation of edema. Chlorpheniramine maleate is an alkylamine-type antihistamine. This group of antihistamines are among the most active histamine antagonists and are generally effective in relatively low doses. They thereby prevent, but do not reverse, responses mediated by histamine alone. The anticholinergic actions of most antihistamines provide a drying effect on the nasal mucosa. These drugs are not so prone to produce drowsiness and are among the most suitable agents for daytime use, but a significant proportion of patients do experience this effect.
Methscopolamine Nitrate is one of the principal anticholinergic/antispasmodic components of belladonna alkaloids that exhibits antisecretory activity. Methscopolamine inhibits the muscarinic actions of acetylcholine on structures innervated by postganglionic cholinergic nerves: smooth muscle, cardiac muscle, sinoatrial and atrioventricular nodes, and exocrine glands. In general, the smaller doses of anticholinergics inhibit salivary and bronchial secretions, sweating, and accommodation; cause dilation of the pupil; and may affect the heart rate.
1 INDICATIONS & USAGE
This product provides relief of the symptoms resulting from irritation of sinus, nasal, and upper respiratory tract tissue.
Phenylephrine exerts a vasoconstrictive and decongestive action while chlorpheniramine maleate decreases the symptoms of watering eyes, post-nasal drip, and sneezing. Methscopolamine nitrate further augments the antisecretory activity of this product.
4 CONTRAINDICATIONS
This product is contraindicated in women who are pregnant or nursing. This product is contraindicated in children under six years of age, because this age group is sensitive to the effects of sympathomimetic amines. It is also contraindicated in newborn or premature infants, because this age group has an increased susceptibility to the anticholinergic side effects of chlorpheniramine maleate. Geriatric patients may be more sensitive to the effects of this medication.
Risk-benefit should be considered when the following conditions exist: Sensitivity to phenylephrine, chlorpheniramine or methscopolamine; Acute asthma; Bladder neck obstruction; Brain damage in children; Cardiac disease, especially cardiac arrhythmias, congestive heart failure, coronary artery disease, and mitral stenosis; Cardiovascular disease; Diabetes Mellitus; Down's Syndrome; Esophagitis, reflux; Glaucoma; Acute hemorrhage with unstable cardiovascular status; Hepatic function impairment; Hernia; Hypertension; Hyperthyroidism; Intestinal atony in the elderly or debilitated patient; Chronic lung disease; Myasthenia gravis; Autonomic neuropathy; Paralytic ileus; Prostatic hypertrophy; Psychiatric disorders; Pyloric obstruction; Renal function impairment; Spastic paralysis, in children; Tachycardia; Toxemia of pregnancy; Ulcerative colitis; Urinary retention, or predisposition to; Urothopy; Xerostomia.
WARNINGS
This product may cause drowsiness or blurred vision. Patients taking this product should be warned not to engage in activities requiring mental alertness such as operating a motor vehicle or other machinery or to perform hazardous tasks while taking this drug.
Sympathomimetic amines should be used with caution in patients with hypertension, ischemic heart disease, diabetes mellitus, increased intraocular pressure, hyperthyroidism, or prostatic hypertrophy. The overdosage of sympathomimetic amines may produce CNS stimulation with convulsions or cardiovascular collapse with accompanying hypotension. Do not exceed recommended dosage.
Heat prostration can occur with the use of methscopolamine when the environmental temperature is high. Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy; in this instance, use of methscopolamine would be inappropriate and possibly harmful.
PRECAUTIONS
General: Use phenylephrine with caution in patients with hypoxia, acidosis, or a history of arteriosclerosis, bradycardia, partial heart block, hypertension, myocardial disease, thrombosis, or ventricular tachycardia. Antihistamines have an atropine-like action and should be used with caution in patients with a history of bronchial asthma, emphysema, increased intraocular pressure, hyperthyroidism, cardiovascular disease and hypertension.
Use methscopolamine with caution in patients with hiatal hernia associated with reflux esophagitis. Use extreme caution and only when needed in patients with autonomic neuropathy, hyperthyroidism, coronary heart disease, congestive heart failure, and cardiac arrhythmia.
17 INFORMATION FOR PATIENTS
Patient consultation should include the following information regarding proper use of this medication:
• Do not take more medication than the amount recommended.
• This medication should be used with caution during exercise or hot weather, overheating may result in heat stroke.
• Do not drive or operate machinery if drowsiness or dizziness occurs.
• Do not ingest alcoholic beverages, monoamine oxidase (MAO) inhibitors, or CNS depression-producing medications (hypnotics, sedatives, tranquilizers) while taking this medication.
• This medication possibly increases sensitivity of eyes to light.
• Methscopolamine nitrate may cause blurred vision.
• If a dose is missed, the medication should be taken as soon as possible unless it is almost time for the next dose. Do not double doses.
• This medication should be stored in a tight, light-resistant container at temperatures between 15°-30°C (59°-86°F).
• Keep all medications out of reach of children. In case of accidental overdose, seek professional assistance or contact a poison control center immediately. Caution patients about the signs of potential side effects, especially:
• Anticholinergic effects – clumsiness or unsteadiness; severe drowsiness; severe dryness of mouth, nose, or throat; flushing or redness of face; shortness of breath or trouble breathing.
• Blood dyscrasias-sore throat and fever; unusual bleeding or bruising; unusual tiredness or weakness.
• Fast or irregular heartbeat.
• Psychotic episodes.
• Tightness in chest.
Note: When anticholinergics are given to patients, especially children, where the environmental temperature is high there is a risk of a rapid increase in body temperature because of suppression of sweat gland activity. Infants, patients with Down’s syndrome, and children with spastic paralysis or brain damage may show an increased response to anticholinergics, thus increasing the potential for side effects.
Geriatric or debilitated patients may respond to usual doses of anticholinergics with excitement, agitation, drowsiness, or confusion.
LABORATORY TESTS
The following may be especially important in patient monitoring (other tests may be warranted in some patients, depending on conditions): Blood pressure determination – recommended at frequent intervals during therapy: Electrocardiogram (ECG) – monitoring may be required: Intraocular pressure determination – recommended at periodic intervals, as these medications may increase the intraocular pressure.
7 DRUG INTERACTIONS
Do not take this product if you are presently taking, or have taken within the preceding two weeks, a prescription drug for high blood pressure or depression without first consulting your physician. Absorption of other oral medications may be decreased during concurrent use with anticholinergics due to decreased gastrointestinal motility and delayed gastric emptying.
Combinations containing any or the following medications, depending on the amount present, may also interact with this product:
- •
- Alkalizers, such as: calcium and/or magnesium-containing antacids; Carbonic anhydrase inhibitors; citrates; sodium bicaronate-urinary excretion of anticholinergics may be delayed by alkalization of the urine, thus potentiating methscopolamine's therapeutic and/or side effects.
- •
- α-adrenergic blocking agents or other medications with α-adrenergic blocking action - prior to administration of α-adrenergics may block the pressor response to phenylephrine, possibly resulting in severe hypotension; medications with α-adrenergic blocking action may decrease the pressor effect and shorten the duration of action of phenylephrine.
- •
- Antacids or absorbent antidiarrheals-simultaneous use of these medications may reduce absorption of methscopolamine, resulting in decreased therapeutic effectiveness; doses of these medications should be spaced 2 or 3 hours apart from doses of methscopolamine.
- •
- Anesthetics, hydrocarbon inhalation - Concurrent use of chloroform, cyclopropane, halothane, or trichloroethylene with phenylephrine may increase the risk of severe ventricular arrhythmias because these anesthetics greatly sensitize the myocardium to the effects of sympathomimetic amines; phenylephrine should be used with caution and in substantially reduced dosage in patients receiving these anesthetics. Enflurane, isoflurane, or methoxyflurane may also cause some sensitization of the myocardium to the effects of sympathomimetic amines.
- •
- Anesthetics, parenteral-local - Phenylephrine should be used cautiously and in carefully circumscribed quantities, if at all, with local anesthetics for anesthetizing areas with end artieries (such as the finger, toes, or penis) or otherwise compromised blood supply; ischemia leading to gangrene may result.
- •
- Anticholinergics - Concurrent use with anticholinergic effects; patients should be advised to report occurrence of gastrointestinal problems promptly since paralytic ileus may occur with concurrent therapy.
- •
- Antidepressants, tricyclic or maprotiline - Concurrent use may potentiate the cardiovascular effects of phenylephrine, possible resulting in arrhythmias, tachycardia, or severe hypertension or hyperpyrexia.
- •
- Antihypertensives, or diuretics used as - Antihypertensive effects may be reduced when these medications are used concurrently with phenylephrine; the patient should be carefully monitored to confirm that the desired effect is being obtained.
- •
- β-adrenergic blocking agents - Therapeutic effects may be inhibited when these medications are used concurrently with phenylephrine, especially larger doses; also, β-adrenergic blockage may result in unopposed α-adrenergic activity with a risk of hypertension and excessive bradycardia with possible heart block.
- •
- CNS Depressants - Concurrent use of alcohol, antihistamines with alcohol, tricyclic antidepressants, barbiturates and other CNS depressants may have an additive effect.
- •
- Cocaine, mucosal-local - Concurrent use with phenylephrine may increase the cardiovascular effects of either or both medications and the risk of adverse side effects.
- •
- Digitalis glycosides - Concurrent use with phenylephrine may increase the risk of cardiac arrhythmias; caution and ECG monitoring are necessary if concurrent use is required.
- •
- Ergoloid mesylates or Ergotamine with phenylephrine may produce peripheral vascular ischemia and gangrene and is not recommended. Concurrent use of ergotamine with phenylephrine, resulting in possible severe hypertension and rupture of cerebral blood vessels.
- •
- Doxapram - Concurrent use may increase the pressor effects of either doxapram or phenylephrine.
- •
- Ketoconazole - Anticholinergics may increase gastrointestinal pH, possibly resulting in a marked reduction in ketoconazole absorption during concurrent use with anticholinergics; patients should be advised to take these medications at least 2 hours after ketoconazole.
- •
- Methyldopa - In addition to possibly decreasing the hypotensive effects of these medications, concurrent use may enhance the pressor response to phenylephrine; caution is required with very small initial doses of methyldopa being administered.
- •
- MAO inhibitors - Concurrent use may prolong and intensify cardiac stimulate and vasopressor effects of phenylephrine and chlorpheniramine, resulting in headache, cardiac arrhythmias, vomiting or sudden and severe hypertensive and/or hyperpyretic crisis. These medications should not be administered during or within 14 days following the administration of MAO inhibitor therapy.
- •
- Metoclopramide - Concurrent use of metoclopramide with anticholinergics may antagonize metoclopramide's effects on gastrointestinal motility.
- •
- Potassium chloride - Concurrent use with anticholinergics may increase the severity of potassium chloride-induced gastrointestinal lesions.
- •
- Rauwolfia alkaloids - Concurrent use may prolong the direct-acting sympathomimetic amines by preventing the uptake into storage granules.
DRUG & OR LABORATORY TEST INTERACTIONS
Antihistamines may interfere with diagnostic test results for skin tests using allergen extracts. Anticholinergics may interfere with diagnostic test results for gastric acid secretion by antagonizing the effect of pantagastrin and histamine, and for radionucleotide gastric emptying studies by delaying gastric emptying.
1 CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
No data is available on the long-term potential of the components of this product for Carcinogenesis, Mutagenesis or Impairment of Fertility in animals or humans.
1 PREGNANCY
Category C: Reproduction studies have been performed with chlorpheniramine maleate. Studies in rabbits and rats at doses up to 50 times and 85 times the human dose revealed no evidence of harm to the fetus. There are, however, no adequate and well-controlled studies in pregnant women. Therefore, it is not known whether these drugs can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Animal reproduction studies have not been conducted with phenylephrine or methscopolamine. This product should be given to a pregnant woman only if clearly needed.
2 LABOR & DELIVERY
Use of phenylephrine during labor may cause fetal anoxia and
bradycardia by increasing contractility of the uterus and decreasing uterine blood flow.
3 NURSING MOTHERS
Small amounts of sympathomimetic amines and antihistamines are excreted in breast milk; use is not recommended because of the risk of adverse effects, such as unusual excitement or irritability in infants. Anticholinergics and antihistamines may inhibit lactation.
4 PEDIATRIC USE
Use of antihistamines is not recommended in newborn or premature infants because this age group has an increased susceptibility to anticholinergic side effects, such as CNS excitation, and an increased tendency toward convulsion.
In infants and children, overdosage may cause hallucinations, convulsions, and death.
A paradoxical reaction characterized by hyperexcitability may occur in older children taking antihistamines.
Use is not recommended for children under six years of age. Infants and young children with spastic paralysis or brain damage since an increased response to anticholinergics are given to children where the environmental temperature because of the suppression of sweat gland activity. A paradoxical reaction characterized by hyperexcitability may occur in children taking large doses of anticholinergics.
Appropriate studies with phenylephrine have not been performed in the pediatric population.
5 GERIATRIC USE
Confusion, hallucinations, seizures, and CNS depression may be more likely to occur in geriatric patients taking sympathomimetic amines. Geriatric patients may also be more sensitive to the effects, especially the vasopressor effects, of
sympathomimetic amines. Confusion, dizziness, sedation, hypotension, hyperexcitability, and anticholinergic side effects, such as dryness of mouth and urinary retention (especially in males), may be more likely to occur in geriatric patients taking antihistamines.
Geriatric patients may respond to usual doses of anticholinergics with excitement, agitation, drowsiness, or confusion.
Geriatric patients are especially susceptible to the anticholinergic side effects, such as constipation, dryness of the mouth, and urinary retention (especially in males). If these side effects occur and continue or are severe, medication should probably be discontinued.
Caution is also recommended when anticholinergics are given to geriatric patients, because of the danger of precipitating undiagnosed glaucoma. Memory may become severely impaired in geriatric patients, with the continued use of anticholinergics, since these drugs block the action of acetylcholine, which is responsible for many functions of the brain, including memory function.
6 ADVERSE REACTIONS
The following adverse reactions have been observed with the use of phenylephrine, chlorpheniramine and methscopolamine; Arrhytmias, blood
dyscrasias, CNS depression, CNS stimulation, dizziness, drowsiness, dryness of mouth, hallucinations, hypotension, hypertension, increased sweating, loss of appetite, paradoxical reaction, restlessness, skin rash, stomach upset or pain, thickening of mucus, tingling in hands or feet, trembling, troubled breathing, unusual tiredness or weakness, vomiting.
Note: Agitation; confusion; difficult or painful urination; drowsiness; dizziness; and dryness of mouth, nose and throat are more likely to occur in the elderly. Nightmares, unusual excitement, nervousness, restlessness, or irritability are more likely to occur in children and the elderly. When anticholinergics are given to patients, especially children, where the environmental temperature is high, there is risk of a rapid increase in body temperature.
9 DRUG ABUSE AND DEPENDENCE
Central nervous system stimulants such as phenylephrine have been abused. At high doses, subjects commonly experience an elevation of mood, a sense of increased energy and alertness, and decreased appetite. Some individuals become anxious, irritable and loquacious. In addition to the marked euphoria, the user experiences a sense of markedly enhanced physical strength and mental capacity. With continued use, tolerance develops, the user increases the dose, and toxic signs and symptoms appear. Depression may follow rapid withdrawal.
Stimulants, such as phenylephrine, are banned and tested for by the U.S. Olympic Committee (USOC) and the National Collegiate Athletic Association (NCAA).
10 OVERDOSAGE
This product is comprised of pharmacologically different components (sympathomimetic amine, antihistamine, anticholinergic). Therefore, it is difficult to predict the exact manifestation of symptoms in a given individual. Reaction to an overdose of this product may vary from CNS depression to stimulant. A description of symptoms which are likely to appear after ingestion of an excess of the individual components follows:
• Overdosage with sympathomimetic amines can cause cardiac arrhythmias, cerebral hemorrhage and pulmonary edema.
It can also cause palpitations, tremor, dizziness,vomiting, fear, labored breathing, headache, dryness of mouth, pallor,weakness, panic, anxiety, confusion,and hallucinations.
• Manifestation of antihistamine overdosage may vary from CNS depression to stimulation. Other signs and symptoms may be dizziness, tinnitus, ataxia, blurred vision, and hypotension. Stimulation is particularly likely in children as are atropine-like signs and symptoms (dry mouth, fixed, dilated pupils, flushing, hypothermia, and gastrointestinal symptoms). In infants and children particularly, antihistamines, in overdosage may produce convulsion and/or death.
• The signs and symptoms of overdosage of anticholinergics are headache, nausea, vomiting, blurred vision, fixed and dilated pupils, hot dry skin, dizziness, dryness of mouth, difficulty in swallowing and CNS stimulation.
Treatment of acute overdosage would probably be based upon treating the patient for phenylephrine toxicity which may manifest itself as excessive CNS stimulation resulting in excitement, tremor, restlessness, and insomnia. Other effects may include hyperpyrexia, hypertension, mydriasis, hyperglycemia and urinary retention. Severe hyperkalemia can occur, probably due to a compartmental shift.
No organ damage or significant metabolic derangement is associated with overdosage.
General Treatment: Treatment is symptomatic and supportive with possible utilization of the following:
• Induction of emesis (syrup of Ipecac recommended); however, precaution against aspiration is necessary, especially in infants and children.
• Gastric lavage (isotonic or 0.45% sodium chloride solution) if patient is unable to vomit within three hours of ingestion
• Saline Cathartics (milk of magnesia) may be used.
• Vasopressors to treat hypotension; however epinephrine should not be used since it may further lower blood pressure.
• For excessive hypertensive effect an α-adrenergic blocker, such as phentolamine, may be administered.
• Hyperpyrexia, especially in children, may require treatment with tepid water sponge bath.
• Excessive CNS stimulation may be counteracted with parenteral diazepam.
• Oxygen and intravenous fluids.
• Precaution against the use of stimulants (analeptic agents) is recommended because they may cause seizures.
• Excitement to a degree which demands attention may be managed with sodium thiopental 2% solution given slowly intravenously or chloral hydrate (100-200 mL of a 2% solution) by rectal infusion.
In severe cases of overdosage it is essential to monitor both the heart (by electrocardiograph) and plasma electrolytes, and to give intravenous potassium as indicated. In the event of progression of the respiration should be instituted and maintained until effective respiratory action returns.
2 DOSAGE & ADMINISTRATION
Adults and children 12 years of age and older: One (1) tablet every 12 hours.
Children 6 to under 12 years: One-half (1/2) tablet every 12 hours.
Not recommended for children under 6 years of age. Tablets may be broken in half for ease of administration but should not be crushed or chewed.
Note: Geriatric patients may be more sensitive to the effects of the usual adult dose. Adjust adult dose accordingly.
16 HOW SUPPLIED
Supplied as white, capsule-shaped tablets scored and debossed “110” on top, Prasco Laboratories logo on bottom. Bottles of 100 tablets, NDC 66993-110-02.
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN. IN CASE OF ACCIDENTAL OVERDOSE, CALL A DOCTOR OR CONTACT A POISON CONTROL CENTER IMMEDIATELY.
Dispense in tight, light-resistant containers as described in the USP/NF. Store at controlled room temperature 59°-86°F (15°-30°C).
Manufactured for:
PRASCO LABORATORIES
Cincinnati, OH 45249
by:
Sovereign Pharmaceuticals, Ltd.
Forth Worth, TX 76118
Rev. 10/2004 500068
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
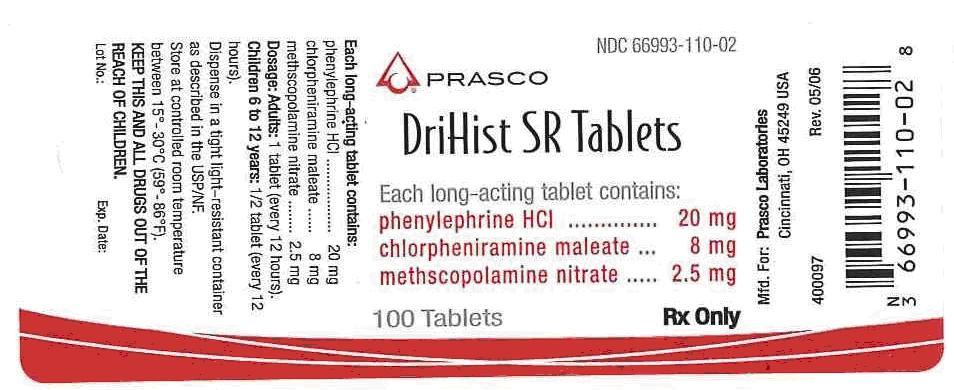
| DRIHIST
SR
phenylephrine hydrochloride, chlorpheniramine maleate, methscopolamine nitrate tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| Unapproved drug other | 01/18/2011 | 01/19/2011 | |
| Labeler - Prasco Laboratories (065969375) |