UMECTA PD UREA
-
urea emulsion
JSJ Pharmaceuticals
----------
Umecta PDDescription
Rx only
For topical use only
Not for ophthalmic use
Umecta is a keratolytic, emollient which is a gentle, yet potent, tissue softener for nails and skin
Each gram of Umecta PD bioadhesive emulsion and topical suspension contains
40% urea, sodium hyaluronate 0.3%, BHT, butyrospermum parkii fruit oil, citric acid,
dimethylacrylamide (and) acrylic acid (and) polystyrene ethyl methacrylate copolymer,
disodium EDTA, glyceryl stearate, glycine soja sterol, helianthus annuus oil,
hydroxypropylmethyl cellulose, purified water, sodium citrate, stearic acid.
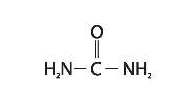
Clinical Pharmacology
Urea gently dissolves the intercellular matrix which results in loosening the horny layer of
skin and shedding scaly skin at regular intervals, thereby softening hyperkeratotic areas. Urea also hydrates
and gently dissolves the intercellular matrix of the nail plate which can result in the softening and eventual debridement
of the nail plate.
Pharmacokinetics
The mechanism of action of topically applied urea is not yet known.
Indications and Uses
For debridement and promotion of normal healing of hyperkeratotic surface lesions, particularly
where healing is retarded by local infection, necrotic tissue, fibrinous or purulent debris or eschar. Urea is useful
for the treatment of hyperkeratotic conditions such as dry, rough skin, dermatitis, psoriasis, xerosis, ichthyosis,
eczema, keratosis, keratoderma, corns, and calluses, as well as damaged, ingrown and devitalized nails.
Contraindications
Known hypersensitivity to any of the listed ingredients.Warnings
For external use only. Avoid contact with eyes, lips or mucous membranes.
Precautions
This medication is to be used as directed by a physician and should not be used to treat anycondition other than that for which it was prescribed. If redness or irritation occurs,
discontinue use.
Pregnancy Category C
Animal reproduction studies have not been conducted with Umecta. It is also not knownwhether Umecta can cause fetal harm when administered to a pregnant woman
or can affect reproductive capacity. Umecta should be given to a pregnant
woman only if clearly needed.
Nursing Mothers
It is not known whether or not this drug is secreted in human milk. Because many drugs are
secreted in human milk, caution should be exercised when Umecta is administered
to a nursing woman.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Adverse Reactions
Transient stinging, burning, itching, or irritation may occur and normally disappear on discontinuing
the medication.
Dosage and Administration
Apply Umecta emulsion/topical suspension or Umecta mousse to affectedskin twice per day or as directed by a physician. Rub in until completely
absorbed.
How Supplied
Umecta PD™ (urea, 40% - sodium hyaluronate 0.3%) bioadhesive emulsion
is available in a:
7 oz. bottle
NDC 68712-017-02
Store at controlled room temperature 15-30°C (59-86°F).
Manufactured for:
JSJ Pharmaceuticals
Charleston, SC 29401
Toll Free: 1-800-499-4468
www.jsjpharm.com
www.umecta.com

| UMECTA PD
UREA
urea emulsion |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| unapproved drug other | 06/01/2007 | ||
| Labeler - JSJ Pharmaceuticals (615074866) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Sonar Products | 104283945 | manufacture | |