CLOZARIL
-
clozapine tablet
Novartis Pharmaceuticals Corporation
----------
ClozarilCLOZARIL®
(clozapine) Tablets
Rx only
Prescribing Information
Before prescribing CLOZARIL® (clozapine), the physician should be thoroughly familiar with the details of this prescribing information.
BOXED WARNING
1. AGRANULOCYTOSIS
BECAUSE OF A SIGNIFICANT RISK OF AGRANULOCYTOSIS, A POTENTIALLY LIFE-THREATENING ADVERSE EVENT, CLOZARIL® (CLOZAPINE) SHOULD BE RESERVED FOR USE IN (1) THE TREATMENT OF SEVERELY ILL PATIENTS WITH SCHIZOPHRENIA WHO FAIL TO SHOW AN ACCEPTABLE RESPONSE TO ADEQUATE COURSES OF STANDARD ANTIPSYCHOTIC DRUG TREATMENT, OR (2) FOR REDUCING THE RISK OF RECURRENT SUICIDAL BEHAVIOR IN PATIENTS WITH SCHIZOPHRENIA OR SCHIZOAFFECTIVE DISORDER WHO ARE JUDGED TO BE AT RISK OF REEXPERIENCING SUICIDAL BEHAVIOR.
PATIENTS BEING TREATED WITH CLOZAPINE MUST HAVE A BASELINE WHITE BLOOD CELL (WBC) COUNT AND ABSOLUTE NEUTROPHIL COUNT (ANC) BEFORE INITIATION OF TREATMENT AS WELL AS REGULAR WBC COUNTS AND ANCs DURING TREATMENT AND FOR AT LEAST 4 WEEKS AFTER DISCONTINUATION OF TREATMENT (SEE WARNINGS).
CLOZAPINE IS AVAILABLE ONLY THROUGH A DISTRIBUTION SYSTEM THAT ENSURES MONITORING OF WBC COUNT AND ANC ACCORDING TO THE SCHEDULE DESCRIBED BELOW PRIOR TO DELIVERY OF THE NEXT SUPPLY OF MEDICATION (SEE WARNINGS).
2. SEIZURES
SEIZURES HAVE BEEN ASSOCIATED WITH THE USE OF CLOZAPINE. DOSE APPEARS TO BE AN IMPORTANT PREDICTOR OF SEIZURE, WITH A GREATER LIKELIHOOD AT HIGHER CLOZAPINE DOSES. CAUTION SHOULD BE USED WHEN ADMINISTERING CLOZAPINE TO PATIENTS HAVING A HISTORY OF SEIZURES OR OTHER PREDISPOSING FACTORS. PATIENTS SHOULD BE ADVISED NOT TO ENGAGE IN ANY ACTIVITY WHERE SUDDEN LOSS OF CONSCIOUSNESS COULD CAUSE SERIOUS RISK TO THEMSELVES OR OTHERS. (SEE WARNINGS.)
3. MYOCARDITIS
ANALYSES OF POSTMARKETING SAFETY DATABASES SUGGEST THAT CLOZAPINE IS ASSOCIATED WITH AN INCREASED RISK OF FATAL MYOCARDITIS, ESPECIALLY DURING, BUT NOT LIMITED TO, THE FIRST MONTH OF THERAPY. IN PATIENTS IN WHOM MYOCARDITIS IS SUSPECTED, CLOZAPINE TREATMENT SHOULD BE PROMPTLY DISCONTINUED. (SEE WARNINGS.)
4. OTHER ADVERSE CARDIOVASCULAR AND RESPIRATORY EFFECTS
ORTHOSTATIC HYPOTENSION, WITH OR WITHOUT SYNCOPE, CAN OCCUR WITH CLOZAPINE TREATMENT. RARELY, COLLAPSE CAN BE PROFOUND AND BE ACCOMPANIED BY RESPIRATORY AND/OR CARDIAC ARREST. ORTHOSTATIC HYPOTENSION IS MORE LIKELY TO OCCUR DURING INITIAL TITRATION IN ASSOCIATION WITH RAPID DOSE ESCALATION. IN PATIENTS WHO HAVE HAD EVEN A BRIEF INTERVAL OFF CLOZAPINE, i.e., 2 OR MORE DAYS SINCE THE LAST DOSE, TREATMENT SHOULD BE STARTED WITH 12.5 mg ONCE OR TWICE DAILY. (SEE WARNINGS and DOSAGE AND ADMINISTRATION.)
SINCE COLLAPSE, RESPIRATORY ARREST AND CARDIAC ARREST DURING INITIAL TREATMENT HAS OCCURRED IN PATIENTS WHO WERE BEING ADMINISTERED BENZODIAZEPINES OR OTHER PSYCHOTROPIC DRUGS, CAUTION IS ADVISED WHEN CLOZAPINE IS INITIATED IN PATIENTS TAKING A BENZODIAZEPINE OR ANY OTHER PSYCHOTROPIC DRUG. (SEE WARNINGS.)
5. INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS TREATED WITH ANTIPSYCHOTIC DRUGS ARE AT AN INCREASED RISK OF DEATH. ANALYSES OF SEVENTEEN PLACEBO-CONTROLLED TRIALS (MODAL DURATION OF 10 WEEKS), LARGELY IN PATIENTS TAKING ATYPICAL ANTIPSYCHOTIC DRUGS, REVEALED A RISK OF DEATH IN THE DRUG-TREATED PATIENTS OF BETWEEN 1.6 TO 1.7 TIMES THE RISK OF DEATH IN PLACEBO-TREATED PATIENTS. OVER THE COURSE OF A TYPICAL 10-WEEK CONTROLLED TRIAL, THE RATE OF DEATH IN DRUG-TREATED PATIENTS WAS ABOUT 4.5%, COMPARED TO A RATE OF ABOUT 2.6% IN THE PLACEBO GROUP. ALTHOUGH THE CAUSES OF DEATH WERE VARIED, MOST OF THE DEATHS APPEARED TO BE EITHER CARDIOVASCULAR (e.g., HEART FAILURE, SUDDEN DEATH) OR INFECTIOUS (e.g., PNEUMONIA) IN NATURE. OBSERVATIONAL STUDIES SUGGEST THAT, SIMILAR TO ATYPICAL ANTIPSYCHOTIC DRUGS, TREATMENT WITH CONVENTIONAL ANTIPSYCHOTIC DRUGS MAY INCREASE MORTALITY. THE EXTENT TO WHICH THE FINDINGS OF INCREASED MORTALITY IN OBSERVATIONAL STUDIES MAY BE ATTRIBUTED TO THE ANTIPSYCHOTIC DRUG AS OPPOSED TO SOME CHARACTERISTIC(S) OF THE PATIENTS IS NOT CLEAR. CLOZARIL® (CLOZAPINE) IS NOT APPROVED FOR THE TREATMENT OF PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS (see WARNINGS).
DESCRIPTION
CLOZARIL® (clozapine), an atypical antipsychotic drug, is a tricyclic dibenzodiazepine derivative, 8-chloro-11-(4-methyl-1-piperazinyl)-5H-dibenzo [b,e] [1,4] diazepine.
The structural formula is
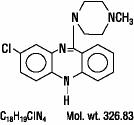
CLOZARIL is available in pale yellow tablets of 25 mg and 100 mg for oral administration.
25 mg and 100 mg Tablets
Active Ingredient: clozapine is a yellow, crystalline powder, very slightly soluble in water.
Inactive Ingredients: colloidal silicon dioxide, lactose, magnesium stearate, povidone, starch (corn), and talc.
CLINICAL PHARMACOLOGY
Pharmacodynamics
CLOZARIL® (clozapine) is classified as an ‘atypical’ antipsychotic drug because its profile of binding to dopamine receptors and its effects on various dopamine-mediated behaviors differ from those exhibited by more typical antipsychotic drug products. In particular, although CLOZARIL does interfere with the binding of dopamine at D1, D2, D3 and D5 receptors, and has a high affinity for the D4 receptor, it does not induce catalepsy nor inhibit apomorphine-induced stereotypy. This evidence, consistent with the view that CLOZARIL is preferentially more active at limbic than at striatal dopamine receptors, may explain the relative freedom of CLOZARIL from extrapyramidal side effects.
CLOZARIL also acts as an antagonist at adrenergic, cholinergic, histaminergic and serotonergic receptors.
Absorption, Distribution, Metabolism and Excretion
In man, CLOZARIL tablets (25 mg and 100 mg) are equally bioavailable relative to a clozapine solution. Following a dosage of 100 mg b.i.d., the average steady-state peak plasma concentration was 319 ng/mL (range: 102-771 ng/mL), occurring at the average of 2.5 hours (range: 1-6 hours) after dosing. The average minimum concentration at steady state was 122 ng/mL (range: 41-343 ng/mL), after 100 mg b.i.d. dosing. Food does not appear to affect the systemic bioavailability of CLOZARIL. Thus, CLOZARIL may be administered with or without food.
Clozapine is approximately 97% bound to serum proteins. The interaction between CLOZARIL and other highly protein-bound drugs has not been fully evaluated but may be important. (See PRECAUTIONS.)
Clozapine is almost completely metabolized prior to excretion and only trace amounts of unchanged drug are detected in the urine and feces. Approximately 50% of the administered dose is excreted in the urine and 30% in the feces. The demethylated, hydroxylated and N-oxide derivatives are components in both urine and feces. Pharmacological testing has shown the desmethyl metabolite to have only limited activity, while the hydroxylated and N-oxide derivatives were inactive.
The mean elimination half-life of clozapine after a single 75-mg dose was 8 hours (range: 4-12 hours), compared to a mean elimination half-life, after achieving steady state with 100 mg b.i.d. dosing, of 12 hours (range: 4-66 hours). A comparison of single-dose and multiple-dose administration of clozapine showed that the elimination half-life increased significantly after multiple dosing relative to that after single-dose administration, suggesting the possibility of concentration-dependent pharmacokinetics. However, at steady state, linearly dose-proportional changes with respect to AUC (area under the curve), peak and minimum clozapine plasma concentrations were observed after administration of 37.5 mg, 75 mg, and 150 mg b.i.d.
Human Pharmacology
In contrast to more typical antipsychotic drugs, CLOZARIL therapy produces little or no prolactin elevation.
As is true of more typical antipsychotic drugs, clinical EEG studies have shown that CLOZARIL increases delta and theta activity and slows dominant alpha frequencies. Enhanced synchronization occurs, and sharp wave activity and spike and wave complexes may also develop. Patients, on rare occasions, may report an intensification of dream activity during CLOZARIL therapy. REM sleep was found to be increased to 85% of the total sleep time. In these patients, the onset of REM sleep occurred almost immediately after falling asleep.
Clinical Trial Data (Reducing the Risk of Recurrent Suicidal Behavior in Patients with Schizophrenia or Schizoaffective Disorder Who are Judged to be at Risk of Reexperiencing Suicidal Behavior)
The effectiveness of CLOZARIL in reducing the risk of recurrent suicidal behavior was assessed in the International Suicide Prevention Trial (InterSePT™), which was a prospective, randomized, international, parallel-group comparison of CLOZARIL vs. Zyprexa®* (olanzapine) in patients with schizophrenia or schizoaffective disorder (DSM-IV) who were judged to be at risk for reexperiencing suicidal behavior. Only about one-fourth of these patients (27%) were considered resistant to standard antipsychotic drug treatment, and the remainder were not. Patients met one of the following criteria:
- They had attempted suicide within the 3 years prior to their baseline evaluation.
- They had been hospitalized to prevent a suicide attempt within the 3 years prior to their baseline evaluation.
- They demonstrated moderate-to-severe suicidal ideation with a depressive component within 1 week prior to their baseline evaluation.
- They demonstrated moderate-to-severe suicidal ideation accompanied by command hallucinations to do self-harm within 1 week prior to their baseline evaluation.
Dosing regimens for each treatment group were determined by individual investigators and were individualized by patient. Dosing was flexible, with a dose range of 200-900 mg/day for CLOZARIL and 5-20 mg/day for Zyprexa. For the 956 patients who received CLOZARIL or Zyprexa in this study, there was extensive use of concomitant psychotropics: 84% with antipsychotics; 65% with anxiolytics; 53% with antidepressants, and 28% with mood stabilizers. There was significantly greater use of concomitant psychotropic medications among the patients in the Zyprexa group.
The primary efficacy measure was time to (1) a significant suicide attempt, including a completed suicide, (2) hospitalization due to imminent suicide risk (including increased level of surveillance for suicidality for patients already hospitalized), or (3) worsening of suicidality severity as demonstrated by “much worsening” or “very much worsening” from baseline in the Clinical Global Impression of Severity of Suicidality as assessed by the Blinded Psychiatrist (CGI-SS-BP) scale. A determination of whether or not a reported event met criterion 1 or 2 above was made by the Suicide Monitoring Board (SMB, a group of experts blinded to patient data).
A total of 980 patients were randomized to the study and 956 received study medication. Sixty-two percent of the patients were diagnosed with schizophrenia, and the remainder (38%) were diagnosed with schizoaffective disorder. Only about one-fourth of the total patient population (27%) was identified as “treatment resistant” at baseline. There were more males than females in the study (61% of all patients were male). The mean age of patients entering the study was 37 years (range 18-69). Most patients were Caucasian (71%), 15% were Black, 1% were Oriental, and 13% were classified as being of “other” races.
Data from this study indicate that CLOZARIL had a statistically significant longer delay in the time to recurrent suicidal behavior in comparison with Zyprexa. This result should be interpreted only as evidence of the effectiveness of CLOZARIL in delaying time to recurrent suicidal behavior, and not a demonstration of the superior efficacy of CLOZARIL over Zyprexa.
The probability of experiencing (1) a significant suicide attempt, including a completed suicide, or (2) hospitalization due to imminent suicide risk (including increased level of surveillance for suicidality for patients already hospitalized) was lower for CLOZARIL patients than for Zyprexa patients at Week 104: CLOZARIL 24% vs. Zyprexa 32%; 95% C.I. of the difference: 2%, 14% (Figure 1).
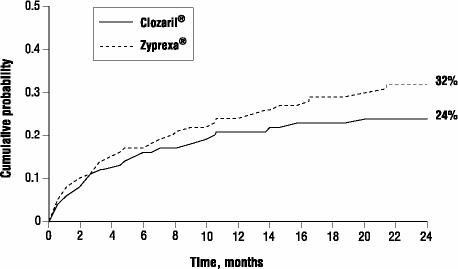
Figure 1: Kaplan-Meier Estimates of Cumulative Probability of a Significant Suicide Attempt or Hospitalization to Prevent Suicide.
INDICATIONS AND USAGE
Treatment-Resistant Schizophrenia
CLOZARIL® (clozapine) is indicated for the management of severely ill schizophrenic patients who fail to respond adequately to standard drug treatment for schizophrenia. Because of the significant risk of agranulocytosis and seizure associated with its use, CLOZARIL should be used only in patients who have failed to respond adequately to treatment with appropriate courses of standard drug treatments for schizophrenia, either because of insufficient effectiveness or the inability to achieve an effective dose due to intolerable adverse effects from those drugs. (See WARNINGS.)
The effectiveness of CLOZARIL in a treatment-resistant schizophrenic population was demonstrated in a 6-week study comparing CLOZARIL and chlorpromazine. Patients meeting DSM-III criteria for schizophrenia and having a mean BPRS total score of 61 were demonstrated to be treatment resistant by history and by open, prospective treatment with haloperidol before entering into the double-blind phase of the study. The superiority of CLOZARIL to chlorpromazine was documented in statistical analyses employing both categorical and continuous measures of treatment effect.
Because of the significant risk of agranulocytosis and seizure, events which both present a continuing risk over time, the extended treatment of patients failing to show an acceptable level of clinical response should ordinarily be avoided. In addition, the need for continuing treatment in patients exhibiting beneficial clinical responses should be periodically reevaluated.
Reduction in the Risk of Recurrent Suicidal Behavior in Schizophrenia or Schizoaffective Disorders
CLOZARIL is indicated for reducing the risk of recurrent suicidal behavior in patients with schizophrenia or schizoaffective disorder who are judged to be at chronic risk for reexperiencing suicidal behavior, based on history and recent clinical state. Suicidal behavior refers to actions by a patient that puts him/herself at risk for death.
The effectiveness of CLOZARIL in reducing the risk of recurrent suicidal behavior was demonstrated over a 2-year treatment period in the InterSePT Trial (see Clinical Trial Data under CLINICAL PHARMACOLOGY). Therefore, CLOZARIL treatment to reduce the risk of suicidal behavior should be continued for at least 2 years (see DOSAGE AND ADMINISTRATION).
The prescriber should be aware that a majority of patients in both treatment groups in InterSePT received other treatments as well to reduce suicide risk, such as antidepressants and other medications, hospitalization, and/or psychotherapy. The contributions of these additional measures are unknown.
CONTRAINDICATIONS
CLOZARIL® (clozapine) is contraindicated in patients with a previous hypersensitivity to clozapine or any other component of this drug, in patients with myeloproliferative disorders, uncontrolled epilepsy, paralytic ileus, or a history of CLOZARIL-induced agranulocytosis or severe granulocytopenia. As with more typical antipsychotic drugs, CLOZARIL is contraindicated in severe central nervous system depression or comatose states from any cause.
CLOZARIL should not be used simultaneously with other agents having a well-known potential to cause agranulocytosis or otherwise suppress bone marrow function. The mechanism of CLOZARIL-induced agranulocytosis is unknown; nonetheless, it is possible that causative factors may interact synergistically to increase the risk and/or severity of bone marrow suppression.
WARNINGS
General
INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS TREATED WITH ANTIPSYCHOTIC DRUGS ARE AT AN INCREASED RISK OF DEATH. CLOZARIL® (clozapine) IS NOT APPROVED FOR THE TREATMENT OF PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS (SEE BOXED WARNING).
AGRANULOCYTOSIS
BECAUSE OF THE SIGNIFICANT RISK OF AGRANULOCYTOSIS, A POTENTIALLY LIFE-THREATENING ADVERSE EVENT (SEE FOLLOWING), CLOZARIL® (clozapine) SHOULD BE RESERVED FOR USE IN THE FOLLOWING INDICATIONS: 1) FOR TREATMENT OF SEVERELY ILL SCHIZOPHRENIC PATIENTS WHO FAIL TO SHOW AN ACCEPTABLE RESPONSE TO ADEQUATE COURSES OF STANDARD DRUG TREATMENT FOR SCHIZOPHRENIA, EITHER BECAUSE OF INSUFFICIENT EFFECTIVENESS OR THE INABILITY TO ACHIEVE AN EFFECTIVE DOSE DUE TO INTOLERABLE ADVERSE EFFECTS FROM THOSE DRUGS. CONSEQUENTLY, BEFORE INITIATING TREATMENT WITH CLOZARIL® (clozapine); IT IS STRONGLY RECOMMENDED THAT A PATIENT BE GIVEN AT LEAST 2 TRIALS, EACH WITH A DIFFERENT STANDARD DRUG PRODUCT FOR SCHIZOPHRENIA, AT AN ADEQUATE DOSE, AND FOR AN ADEQUATE DURATION. 2) FOR REDUCING THE RISK FOR RECURRENT SUICIDAL BEHAVIOR IN PATIENTS WITH SCHIZOPHRENIA OR SCHIZOAFFECTIVE DISORDER WHO ARE JUDGED TO BE AT RISK OF REEXPERIENCING SUICIDAL BEHAVIOR.
CLOZARIL® (clozapine) IS AVAILABLE ONLY THROUGH A DISTRIBUTION SYSTEM THAT ENSURES MONITORING OF WHITE BLOOD CELL (WBC) COUNT AND ABSOLUTE NEUTROPHIL COUNT (ANC) ACCORDING TO THE SCHEDULE DESCRIBED BELOW PRIOR TO DELIVERY OF THE NEXT SUPPLY OF MEDICATION.
AS DESCRIBED IN TABLE 1, PATIENTS WHO ARE BEING TREATED WITH CLOZARIL® (clozapine) MUST HAVE A BASELINE WBC COUNT AND ANC BEFORE INITIATION OF TREATMENT, AND A WBC COUNT AND ANC EVERY WEEK FOR THE FIRST 6 MONTHS. THEREAFTER, IF ACCEPTABLE WBC COUNTS AND ANC (WBC ≥3500/mm3 and ANC ≥2000/mm3) HAVE BEEN MAINTAINED DURING THE FIRST 6 MONTHS OF CONTINUOUS THERAPY, WBC COUNTS AND ANC CAN BE MONITORED EVERY 2 WEEKS FOR THE NEXT 6 MONTHS. THEREAFTER, IF ACCEPTABLE WBC COUNTS AND ANC (WBC ≥3500/mm3 and ANC ≥2000/mm3) HAVE BEEN MAINTAINED DURING THE SECOND 6 MONTHS OF CONTINUOUS THERAPY, WBC COUNT AND ANC CAN BE MONITORED EVERY 4 WEEKS.
WHEN TREATMENT WITH CLOZARIL® (clozapine) IS DISCONTINUED (REGARDLESS OF THE REASON), WBC COUNT AND ANC MUST BE MONITORED WEEKLY FOR AT LEAST 4 WEEKS FROM THE DAY OF DISCONTINUATION OR UNTIL WBC ≥3500/mm3 AND ANC ≥2000/mm3.
Agranulocytosis
Background
Agranulocytosis, defined as an ANC of less than 500/mm3, has been estimated to occur in association with CLOZARIL® (clozapine) use at a cumulative incidence at 1 year of approximately 1.3%, based on the occurrence of 15 U.S. cases out of 1,743 patients exposed to CLOZARIL® (clozapine) during its clinical testing prior to domestic marketing. All of these cases occurred at a time when the need for close monitoring of WBC counts was already recognized. Agranulocytosis could prove fatal if not detected early and therapy interrupted. Of the 149 cases of agranulocytosis reported world wide in association with CLOZARIL® (clozapine) use as of December 31, 1989, 32% were fatal. However, few of these deaths occurred since 1977, at which time the knowledge of CLOZARIL® (clozapine)-induced agranulocytosis became more widespread, and close monitoring of WBC counts more widely practiced. In the U.S., under a weekly WBC count monitoring system with CLOZARIL® (clozapine), there have been 585 cases of agranulocytosis as of August 21, 1997; 19 were fatal (3%). During this period 150,409 patients received CLOZARIL® (clozapine). A hematologic risk analysis was conducted based upon the available information in the Clozaril® National Registry (CNR) for U.S. patients. Based upon a cut-off date of April 30, 1995, the incidence rates of agranulocytosis based upon a weekly monitoring schedule, rose steeply during the first two months of therapy, peaking in the third month. Among CLOZARIL® (clozapine) patients who continued the drug beyond the third month, the weekly incidence of agranulocytosis fell to a substantial degree. After 6 months, the weekly incidence of agranulocytosis declines still further, however, it never reaches zero. It should be noted that any type of reduction in the frequency of monitoring WBC counts may result in an increased incidence of agranulocytosis.
Risk Factors
Experience from clinical development, as well as from examples in the medical literature, suggest that patients who have developed agranulocytosis during CLOZARIL® (clozapine) therapy are at increased risk of subsequent episodes of agranulocytosis. Analysis of WBC count data from the Clozaril® National Registry also suggests that patients who have an initial episode of moderate leukopenia (3000/mm3 >WBC ≥2000/mm3) are at an increased risk of subsequent episodes of agranulocytosis. Except for bone marrow suppression during initial CLOZARIL® (clozapine) therapy, there are no other established risk factors, based on worldwide experience, for the development of agranulocytosis in association with CLOZARIL® (clozapine) use. However, a disproportionate number of the U.S. cases of agranulocytosis occurred in patients of Jewish background compared to the overall proportion of such patients exposed during domestic development of CLOZARIL® (clozapine). Most of the U.S. cases of agranulocytosis occurred within 4-10 weeks of exposure but neither dose nor duration is a reliable predictor of this problem. Agranulocytosis associated with other antipsychotic drugs has been reported to occur with a greater frequency in women, the elderly, and in patients who are cachectic or have a serious underlying medical illness; such patients may also be at particular risk with CLOZARIL® (clozapine), although this has not been definitely demonstrated.
WBC Count and ANC Monitoring Schedule
Table 1 provides a summary of the frequency of monitoring that should occur based on various stages of therapy (e.g., initiation of therapy) or results from WBC count and ANC monitoring tests (e.g., moderate leukopenia). The text that follows should be consulted for additional details regarding the treatment of patients under the various conditions (e.g., severe leukopenia).
Patients should be advised to report immediately the appearance of lethargy, weakness, fever, sore throat or any other signs of infection occurring at any time during CLOZARIL® (clozapine) therapy. Such patients should have a WBC count and ANC performed promptly.
| Situation | Hematological Values for Monitoring | Frequency of WBC and ANC Monitoring |
| Initiation of therapy | WBC ≥3500/mm3
ANC ≥2000/mm3 Note: Do not initiate in patients with 1) history of myeloproliferative disorder or 2) Clozaril® (clozapine) induced agranulocytosis or granulocytopenia | Weekly for 6 months |
| 6 months – 12 months of therapy | All results for WBC ≥3500/mm3 and ANC ≥2000/mm3 | Every 2 weeks for 6 months |
| 12 months of therapy | All results for WBC ≥3500/mm3 and ANC ≥2000/mm3 | Every 4 weeks ad infinitum |
| Immature forms present | N/A | Repeat WBC and ANC |
| Discontinuation of Therapy | N/A | Weekly for at least 4 weeks from day of discontinuation or until WBC ≥3500/mm3 and ANC >2000/mm3 |
| Substantial drop in WBC or ANC | Single Drop or cumulative drop within 3 weeks of WBC ≥3000/mm3 or ANC ≥1500/mm3 | 1. Repeat WBC and ANC 2. If repeat values are 3000/mm3 ≤WBC ≤3500/mm3 and ANC <2000/mm3, then monitor twice weekly |
| Mild Leukopenia -------------------------- Mild Granulocytopenia | 3500/mm3 >WBC ≥3000/mm3
and/or 2000/mm3 >ANC ≥1500/mm3 | Twice-weekly until WBC >3500/mm3 and ANC >2000/mm3 then return to previous monitoring frequency |
| Moderate Leukopenia --------------------------- Moderate Granulocytopenia | 3000/mm3 >WBC ≥2000/mm3
and/or 1500/mm3 > ANC ≥1000/mm3 | 1.
Interrupt therapy 2. Daily until WBC >3000/mm3 and ANC >1500/mm3 3. Twice-weekly until WBC >3500/mm3 and ANC >2000/mm3 4. May rechallenge when WBC >3500/mm3 and ANC >2000/mm3 5. If rechallenged, monitor weekly for 1 year before returning to the usual monitoring schedule of every 2 weeks for 6 months and then every 4 weeks ad infinitum |
| Severe Leukopenia ------------------------ Severe Granulocytopenia | WBC <2000/mm3
and/or ANC <1000/mm3 | 1.
Discontinue treatment and do not rechallenge patient 2. Monitor until normal and for at least four weeks from day of discontinuation as follows: Daily until WBC >3000/mm3 and ANC >1500/mm3 Twice weekly until WBC >3500/mm3 and ANC >2000/mm3 Weekly after WBC >3500/mm3 |
| Agranulocytosis | ANC ≤500/mm3 | 1.
Discontinue treatment and do not rechallenge patient 2. Monitor until normal and for at least four weeks from day of discontinuation as follows: Daily until WBC >3000/mm3 and ANC >1500/mm3 Twice weekly until WBC >3500/mm3 and ANC >2000/mm3 Weekly after WBC >3500/mm3 |
| *WBC=white blood cell count; ANC=absolute neutrophil count | ||
Decrements in WBC Count and/or ANC
Consult Table 1 above to determine how to monitor patients who experience decrements in WBC count and ANC at any point during treatment. Additionally, patients should be carefully monitored for flu-like symptoms or other symptoms suggestive of infection.
Nonrechallengeable Patients
If the total WBC count falls below 2000/mm3 or the ANC falls below 1000/mm3, bone marrow aspiration should be considered to ascertain granulopoietic status and patients should not be rechallenged with CLOZARIL® (clozapine). Protective isolation with close observation may be indicated if granulopoiesis is determined to be deficient. Should evidence of infection develop, the patient should have appropriate cultures performed and an appropriate antibiotic regimen instituted.
Patients discontinued from CLOZARIL® (clozapine) therapy due to significant granulopoietic suppression have been found to develop agranulocytosis upon rechallenge, often with a shorter latency on reexposure. To reduce the chances of rechallenge occurring in patients who have experienced significant bone marrow suppression during CLOZARIL® (clozapine) therapy, a single, national master file (i.e., Nonrechallengeable Database) is maintained confidentially.
Treatment of Rechallengeable Patients
Patients may be rechallenged with CLOZARIL® (clozapine) if their WBC count does not fall below 2000/mm3 and the ANC does not fall below 1000/mm3. However, analysis of data from the Clozaril® National Registry suggests that patients who have an initial episode of moderate leukopenia (3000/mm3 >WBC ≥2000/mm3) have up to a 12-fold increased risk of having a subsequent episode of agranulocytosis when rechallenged compared to the full cohort of patients treated with CLOZARIL® (clozapine). Although CLOZARIL® (clozapine) therapy may be resumed if no symptoms of infection develop, and when the WBC count rises above 3500/mm3 and the ANC rises above 2000/mm3, prescribers are strongly advised to consider whether the benefit of continuing CLOZARIL® (clozapine) treatment outweighs the increased risk of agranulocytosis.
Analyses of the Clozaril® National Registry have shown an increased risk of having a subsequent episode of granulopoietic suppression up to a year after recovery from the initial episode. Therefore, as noted in Table 1 above, patients must undergo weekly WBC count and ANC monitoring for one year following recovery from an episode of moderate leukopenia and/or moderate granulocytopenia regardless of when the episode develops. If acceptable WBC counts and ANC (WBC ≥3500/mm3 and ANC ≥2000/mm3) have been maintained during the year of weekly monitoring, WBC counts can be monitored every 2 weeks for the next 6 months. If acceptable WBC counts and ANC (WBC ≥3500/mm3 and ANC ≥2000/mm3) continue to be maintained during the 6 months of every 2 week monitoring, WBC counts can be monitored every 4 weeks thereafter, ad infinitum.
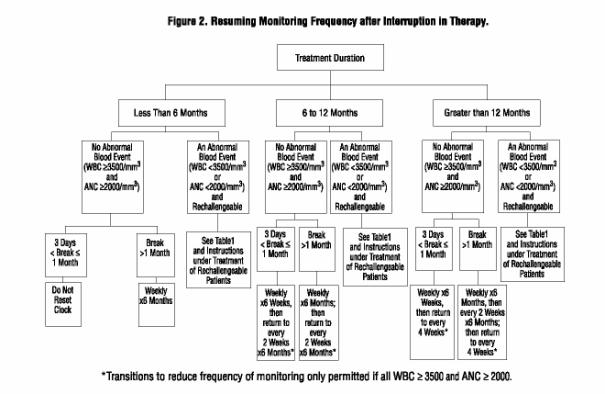
Interruptions in Therapy
Figure 2 provides instructions regarding reinitiating therapy and subsequently the frequency of WBC count and ANC monitoring after a period of interruption.
Eosinophilia
In clinical trials, 1% of patients developed eosinophilia, which, in rare cases, can be substantial. If a differential count reveals a total eosinophil count above 4000/mm3, CLOZARIL therapy should be interrupted until the eosinophil count falls below 3000/mm3.
Seizures
Seizure has been estimated to occur in association with CLOZARIL use at a cumulative incidence at one year of approximately 5%, based on the occurrence of one or more seizures in 61 of 1,743 patients exposed to CLOZARIL during its clinical testing prior to domestic marketing (i.e., a crude rate of 3.5%). Dose appears to be an important predictor of seizure, with a greater likelihood of seizure at the higher CLOZARIL doses used.
Caution should be used in administering CLOZARIL to patients having a history of seizures or other predisposing factors. Because of the substantial risk of seizure associated with CLOZARIL use, patients should be advised not to engage in any activity where sudden loss of consciousness could cause serious risk to themselves or others, e.g., the operation of complex machinery, driving an automobile, swimming, climbing, etc.
Myocarditis
Postmarketing surveillance data from four countries that employ hematological monitoring of clozapine-treated patients revealed: 30 reports of myocarditis with 17 fatalities in 205,493 U.S. patients (August 2001); 7 reports of myocarditis with 1 fatality in 15,600 Canadian patients (April 2001); 30 reports of myocarditis with 8 fatalities in 24,108 U.K. patients (August 2001); 15 reports of myocarditis with 5 fatalities in 8,000 Australian patients (March 1999). These reports represent an incidence of 5.0, 16.3, 43.2, and 96.6 cases/100,000 patient-years, respectively. The number of fatalities represent an incidence of 2.8, 2.3, 11.5, and 32.2 cases/100,000 patient-years, respectively.
The overall incidence rate of myocarditis in patients with schizophrenia treated with antipsychotic agents is unknown. However, for the established market economies (WHO), the incidence of myocarditis is 0.3 cases/100,000 patient-years and the fatality rate is 0.2 cases/100,000 patient-years. Therefore, the rate of myocarditis in clozapine-treated patients appears to be 17-322 times greater than the general population and is associated with an increased risk of fatal myocarditis that is 14-161 times greater than the general population.
The total reports of myocarditis for these four countries was 82 of which 51 (62%) occurred within the first month of clozapine treatment, 25 (31%) occurred after the first month of therapy and 6 (7%) were unknown. The median duration of treatment was 3 weeks. Of 5 patients rechallenged with clozapine, 3 had a recurrence of myocarditis. Of the 82 reports, 31 (38%) were fatal and 25 patients who died had evidence of myocarditis at autopsy. These data also suggest that the incidence of fatal myocarditis may be highest during the first month of therapy.
Therefore, the possibility of myocarditis should be considered in patients receiving CLOZARIL who present with unexplained fatigue, dyspnea, tachypnea, fever, chest pain, palpitations, other signs or symptoms of heart failure, or electrocardiographic findings such as ST-T wave abnormalities or arrhythmias. It is not known whether eosinophilia is a reliable predictor of myocarditis. Tachycardia, which has been associated with CLOZARIL treatment, has also been noted as a presenting sign in patients with myocarditis. Therefore, tachycardia during the first month of therapy warrants close monitoring for other signs of myocarditis.
Prompt discontinuation of CLOZARIL treatment is warranted upon suspicion of myocarditis. Patients with clozapine-related myocarditis should not be rechallenged with CLOZARIL.
Other Adverse Cardiovascular and Respiratory Effects
Orthostatic hypotension with or without syncope can occur with CLOZARIL treatment and may represent a continuing risk in some patients. Rarely (approximately 1 case per 3,000 patients), collapse can be profound and be accompanied by respiratory and/or cardiac arrest. Orthostatic hypotension is more likely to occur during initial titration in association with rapid dose escalation and may even occur on first dose. In one report, initial doses as low as 12.5 mg were associated with collapse and respiratory arrest. When restarting patients who have had even a brief interval off CLOZARIL, i.e., 2 days or more since the last dose, it is recommended that treatment be reinitiated with one-half of a 25-mg tablet (12.5 mg) once or twice daily. (See DOSAGE AND ADMINISTRATION.)
Some of the cases of collapse/respiratory arrest/cardiac arrest during initial treatment occurred in patients who were being administered benzodiazepines; similar events have been reported in patients taking other psychotropic drugs or even CLOZARIL by itself. Although it has not been established that there is an interaction between CLOZARIL and benzodiazepines or other psychotropics, caution is advised when clozapine is initiated in patients taking a benzodiazepine or any other psychotropic drug.
Tachycardia, which may be sustained, has also been observed in approximately 25% of patients taking CLOZARIL, with patients having an average increase in pulse rate of 10-15 bpm. The sustained tachycardia is not simply a reflex response to hypotension, and is present in all positions monitored. Either tachycardia or hypotension may pose a serious risk for an individual with compromised cardiovascular function.
A minority of CLOZARIL-treated patients experience ECG repolarization changes similar to those seen with other antipsychotic drugs, including S-T segment depression and flattening or inversion of T waves, which all normalize after discontinuation of CLOZARIL. The clinical significance of these changes is unclear. However, in clinical trials with CLOZARIL, several patients experienced significant cardiac events, including ischemic changes, myocardial infarction, arrhythmias and sudden death. In addition, there have been postmarketing reports of congestive heart failure, pericarditis, and pericardial effusions. Causality assessment was difficult in many of these cases because of serious preexisting cardiac disease and plausible alternative causes. Rare instances of sudden death have been reported in psychiatric patients, with or without associated antipsychotic drug treatment, and the relationship of these events to antipsychotic drug use is unknown.
CLOZARIL should be used with caution in patients with known cardiovascular and/or pulmonary disease, and the recommendation for gradual titration of dose should be carefully observed.
Hyperglycemia and Diabetes Mellitus
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics including CLOZARIL. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of treatment-emergent hyperglycemia-related adverse events in patients treated with the atypical antipsychotics. Precise risk estimates for hyperglycemia-related adverse events in patients treated with atypical antipsychotics are not available.
Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients with risk factors for diabetes mellitus (e.g., obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of treatment and periodically during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required continuation of antidiabetic treatment despite discontinuation of the suspect drug.
Neuroleptic Malignant Syndrome (NMS)
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology.
The management of NMS should include 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
There have been several reported cases of NMS in patients receiving CLOZARIL alone or in combination with lithium or other CNS-active agents.
Tardive Dyskinesia
A syndrome consisting of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of treatment, which patients are likely to develop the syndrome.
There are several reasons for predicting that CLOZARIL may be different from other antipsychotic drugs in its potential for inducing tardive dyskinesia, including the preclinical finding that it has a relatively weak dopamine-blocking effect and the clinical finding of a low incidence of certain acute extrapyramidal symptoms, e.g., dystonia. A few cases of tardive dyskinesia have been reported in patients on CLOZARIL who had been previously treated with other antipsychotic agents, so that a causal relationship cannot be established. There have been no reports of tardive dyskinesia directly attributable to CLOZARIL alone. Nevertheless, it cannot be concluded, without more extended experience, that CLOZARIL is incapable of inducing this syndrome.
Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses. There is no known treatment for established cases of tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic drug treatment is withdrawn. Antipsychotic drug treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibly mask the underlying process. The effect that symptom suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, CLOZARIL should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. As with any antipsychotic drug, chronic CLOZARIL use should be reserved for patients who appear to be obtaining substantial benefit from the drug. In such patients, the smallest dose and the shortest duration of treatment should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient on CLOZARIL, drug discontinuation should be considered. However, some patients may require treatment with CLOZARIL despite the presence of the syndrome.
PRECAUTIONS
General
Because of the significant risk of agranulocytosis and seizure, both of which present a continuing risk over time, the extended treatment of patients failing to show an acceptable level of clinical response should ordinarily be avoided. In addition, the need for continuing treatment in patients exhibiting beneficial clinical responses should be periodically reevaluated. Although it is not known whether the risk would be increased, it is prudent either to avoid CLOZARIL® (clozapine) or use it cautiously in patients with a previous history of agranulocytosis induced by other drugs.
Cardiomyopathy
Cases of cardiomyopathy have been reported in patients treated with clozapine. The reporting rate for cardiomyopathy in clozapine-treated patients in the U.S. (8.9 per 100,000 person-years) was similar to an estimate of the cardiomyopathy incidence in the U.S. general population derived from the 1999 National Hospital Discharge Survey data (9.7 per 100,000 person-years). Approximately 80% of clozapine-treated patients in whom cardiomyopathy was reported were less than 50 years of age; the duration of treatment with clozapine prior to cardiomyopathy diagnosis varied, but was >6 months in 65% of the reports. Dilated cardiomyopathy was most frequently reported, although a large percentage of reports did not specify the type of cardiomyopathy. Signs and symptoms suggestive of cardiomyopathy, particularly exertional dyspnea, fatigue, orthopnea, paroxysmal nocturnal dyspnea, and peripheral edema should alert the clinician to perform further investigations. If the diagnosis of cardiomyopathy is confirmed, the prescriber should discontinue clozapine unless the benefit to the patient clearly outweighs the risk.
Fever
During CLOZARIL therapy, patients may experience transient temperature elevations above 100.4°F (38°C), with the peak incidence within the first 3 weeks of treatment. While this fever is generally benign and self-limiting, it may necessitate discontinuing patients from treatment. On occasion, there may be an associated increase or decrease in WBC count. Patients with fever should be carefully evaluated to rule out the possibility of an underlying infectious process or the development of agranulocytosis. In the presence of high fever, the possibility of Neuroleptic Malignant Syndrome (NMS) must be considered. There have been several reports of NMS in patients receiving CLOZARIL, usually in combination with lithium or other CNS-active drugs. (See Neuroleptic Malignant Syndrome [NMS], under WARNINGS.)
Pulmonary Embolism
The possibility of pulmonary embolism should be considered in patients receiving CLOZARIL who present with deep vein thrombosis, acute dyspnea, chest pain or with other respiratory signs and symptoms. As of December 31, 1993 there were 18 cases of fatal pulmonary embolism in association with CLOZARIL therapy in users 10-54 years of age. Based upon the extent of use observed in the Clozaril® National Registry, the mortality rate associated with pulmonary embolus was 1 death per 3,450 person-years of use. This rate was about 27.5 times higher than that in the general population of a similar age and gender (95% Confidence Interval; 17.1, 42.2). Deep vein thrombosis has also been observed in association with CLOZARIL therapy. Whether pulmonary embolus can be attributed to CLOZARIL or some characteristic(s) of its users is not clear, but the occurrence of deep vein thrombosis or respiratory symptomatology should suggest its presence.
Hepatitis
Caution is advised in patients using CLOZARIL who have concurrent hepatic disease. Hepatitis has been reported in both patients with normal and preexisting liver function abnormalities. In patients who develop nausea, vomiting, and/or anorexia during CLOZARIL treatment, liver function tests should be performed immediately. If the elevation of these values is clinically relevant or if symptoms of jaundice occur, treatment with CLOZARIL should be discontinued.
Anticholinergic Toxicity
Eye: CLOZARIL has potent anticholinergic effects and care should be exercised in using this drug in the presence of narrow angle glaucoma.
Gastrointestinal: CLOZARIL use has been associated with varying degrees of impairment of intestinal peristalsis, ranging from constipation to intestinal obstruction, fecal impaction and paralytic ileus (see ADVERSE REACTIONS). On rare occasions, these cases have been fatal. Constipation should be initially treated by ensuring adequate hydration, and use of ancillary therapy such as bulk laxatives. Consultation with a gastroenterologist is advisable in more serious cases.
Prostate: CLOZARIL has potent anticholinergic effects and care should be exercised in using this drug in the presence of prostatic enlargement.
Interference with Cognitive and Motor Performance
Because of initial sedation, CLOZARIL may impair mental and/or physical abilities, especially during the first few days of therapy. The recommendations for gradual dose escalation should be carefully adhered to, and patients cautioned about activities requiring alertness.
Cerebrovascular Adverse Events
An increased risk of cerebrovascular adverse events has been observed in dementia patients treated with some atypical antipsychotics. The mechanism for this increased risk is not known. An increased risk cannot be excluded for dementia patients or other patients treated with CLOZARIL. CLOZARIL should be used with caution in patients with dementia or risk factors for stroke.
Use in Patients with Concomitant Illness
Clinical experience with CLOZARIL in patients with concomitant systemic diseases is limited. Nevertheless, caution is advisable in using CLOZARIL in patients with renal or cardiac disease.
Use in Patients Undergoing General Anesthesia
Caution is advised in patients being administered general anesthesia because of the CNS effects of CLOZARIL. Check with the anesthesiologist regarding continuation of CLOZARIL therapy in a patient scheduled for surgery.
Information for Patients
Physicians are advised to discuss the following issues with patients for whom they prescribe CLOZARIL:
- Patients who are to receive CLOZARIL should be warned about the significant risk of developing agranulocytosis. Patients should be advised to report immediately the appearance of lethargy, weakness, fever, sore throat, malaise, mucous membrane ulceration or other possible signs of infection. Particular attention should be paid to any flu-like complaints or other symptoms that might suggest infection.
- Patients should be informed that CLOZARIL tablets will be made available only through a special program designed to ensure the required blood monitoring in order to reduce the risk of developing agranulocytosis. Patients should be informed that their WBC count and ANC will be monitored as follows:
- Weekly blood tests are required for the first 6 months.
- If acceptable WBC counts and ANCs (WBC ≥3500/mm3 and ANC ≥2000/mm3) have been maintained during the first 6 months of continuous therapy, then WBC counts and ANCs can be monitored every 2 weeks for the next 6 months.
- Thereafter, if acceptable WBC counts and ANCs have been maintained during the second 6 months of continuous therapy, WBC counts and ANCs can be monitored every 4 weeks.
- Weekly blood tests are required for the first 6 months.
- Patients should be informed that CLOZARIL tablets will be made available only through a special program designed to ensure the required blood monitoring in order to reduce the risk of developing agranulocytosis. Patients should be informed that their WBC count and ANC will be monitored as follows:
- Patients should be informed of the significant risk of seizure during CLOZARIL treatment, and they should be advised to avoid driving and any other potentially hazardous activity while taking CLOZARIL.
- Patients should be advised of the risk of orthostatic hypotension, especially during the period of initial dose titration.
- Patients should be informed that if they miss taking CLOZARIL for more than 2 days, they should not restart their medication at the same dosage, but should contact their physician for dosing instructions.
- Patients should notify their physician if they are taking, or plan to take, any prescription or over-the-counter drugs or alcohol.
- Patients should notify their physician if they become pregnant or intend to become pregnant during therapy.
- Patients should not breast-feed an infant if they are taking CLOZARIL.
Drug Interactions
The risks of using CLOZARIL in combination with other drugs have not been systematically evaluated.
Pharmacodynamic-Related Interactions
The mechanism of CLOZARIL-induced agranulocytosis is unknown; nonetheless, the possibility that causative factors may interact synergistically to increase the risk and/or severity of bone marrow suppression warrants consideration. Therefore, CLOZARIL should not be used with other agents having a well-known potential to suppress bone marrow function.
Given the primary CNS effects of CLOZARIL, caution is advised in using it concomitantly with other CNS-active drugs or alcohol.
Orthostatic hypotension in patients taking clozapine can, in rare cases (approximately 1 case per 3,000 patients), be accompanied by profound collapse and respiratory and/or cardiac arrest. Some of the cases of collapse/respiratory arrest/cardiac arrest during initial treatment occurred in patients who were being administered benzodiazepines; similar events have been reported in patients taking other psychotropic drugs or even CLOZARIL by itself. Although it has not been established that there is an interaction between CLOZARIL and benzodiazepines or other psychotropics, caution is advised when clozapine is initiated in patients taking a benzodiazepine or any other psychotropic drug.
CLOZARIL may potentiate the hypotensive effects of antihypertensive drugs and the anticholinergic effects of atropine-type drugs. The administration of epinephrine should be avoided in the treatment of drug-induced hypotension because of a possible reverse epinephrine effect.
Pharmacokinetic-Related Interactions
Clozapine is a substrate for many CYP450 isozymes, in particular 1A2, 2D6, and 3A4. The risk of metabolic interactions caused by an effect on an individual isoform is therefore minimized. Nevertheless, caution should be used in patients receiving concomitant treatment with other drugs that are either inhibitors or inducers of these enzymes.
Concomitant administration of drugs known to induce cytochrome P450 enzymes may decrease the plasma levels of clozapine. Phenytoin, tobacco smoke, and rifampin may decrease CLOZARIL plasma levels, resulting in a decrease in effectiveness of a previously effective CLOZARIL dose.
Concomitant administration of drugs known to inhibit the activity of cytochrome P450 isozymes may increase the plasma levels of clozapine. Cimetidine, caffeine, citalopram, ciprofloxacin, and erythromycin may increase plasma levels of CLOZARIL, potentially resulting in adverse effects. Although concomitant use of CLOZARIL and carbamazepine is not recommended, it should be noted that discontinuation of concomitant carbamazepine administration may result in an increase in CLOZARIL plasma levels.
In a study of schizophrenic patients who received clozapine under steady-state conditions, fluvoxamine or paroxetine was added in 16 and 14 patients, respectively. After 14 days of coadministration, mean trough concentrations of clozapine and its metabolites, N-desmethylclozapine and clozapine N-oxide, were elevated with fluvoxamine by about three-fold compared to baseline concentrations. Paroxetine produced only minor changes in the levels of clozapine and its metabolites. However, other published reports describe modest elevations (less than two-fold) of clozapine and metabolite concentrations when clozapine was taken with paroxetine, fluoxetine, and sertraline. Therefore, such combined treatment should be approached with caution and patients should be monitored closely when CLOZARIL is combined with these drugs, particularly with fluvoxamine. A reduced CLOZARIL dose should be considered.
A subset (3%-10%) of the population has reduced activity of certain drug metabolizing enzymes such as the cytochrome P450 isozyme P450 2D6. Such individuals are referred to as “poor metabolizers” of drugs such as debrisoquin, dextromethorphan, the tricyclic antidepressants, and clozapine. These individuals may develop higher than expected plasma concentrations of clozapine when given usual doses. In addition, certain drugs that are metabolized by this isozyme, including many antidepressants (clozapine, selective serotonin reuptake inhibitors, and others), may inhibit the activity of this isozyme, and thus may make normal metabolizers resemble poor metabolizers with regard to concomitant therapy with other drugs metabolized by this enzyme system, leading to drug interaction.
Concomitant use of clozapine with other drugs metabolized by cytochrome P450 2D6 may require lower doses than usually prescribed for either clozapine or the other drug. Therefore, coadministration of clozapine with other drugs that are metabolized by this isozyme, including antidepressants, phenothiazines, carbamazepine, and Type 1C antiarrhythmics (e.g., propafenone, flecainide and encainide), or that inhibit this enzyme (e.g., quinidine), should be approached with caution.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenic potential was demonstrated in long-term studies in mice and rats at doses approximately 7 times the typical human dose on a mg/kg basis. Fertility in male and female rats was not adversely affected by clozapine. Clozapine did not produce genotoxic or mutagenic effects when assayed in appropriate bacterial and mammalian tests.
Pregnancy Category B
Reproduction studies have been performed in rats and rabbits at doses of approximately 2-4 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to clozapine. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, and in view of the desirability of keeping the administration of all drugs to a minimum during pregnancy, this drug should be used only if clearly needed.
Non-Teratogenic Effects
Neonates exposed to antipsychotic drugs, during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress and feeding disorder in these neonates. These complications have varied in severity; while in some cases symptoms have been self-limited, in other cases neonates have required intensive care unit support and prolonged hospitalization.
Antipsychotic drugs, including Clozaril, should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
Animal studies suggest that clozapine may be excreted in breast milk and have an effect on the nursing infant. Therefore, women receiving CLOZARIL should not breast-feed.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatric Use
Clinical studies of clozapine did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently from younger subjects.
Orthostatic hypotension can occur with CLOZARIL treatment and tachycardia, which may be sustained, has been observed in about 25% of patients taking CLOZARIL (see BOXED WARNING, Other Adverse Cardiovascular and Respiratory Effects). Elderly patients, particularly those with compromised cardiovascular functioning, may be more susceptible to these effects.
Also, elderly patients may be particularly susceptible to the anticholinergic effects of CLOZARIL, such as urinary retention and constipation. (See PRECAUTIONS, Anticholinergic Toxicity.)
Dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. Other reported clinical experience does suggest that the prevalence of tardive dyskinesia appears to be highest among the elderly, especially elderly women. (See WARNINGS, Tardive Dyskinesia.)
ADVERSE REACTIONS
Associated with Discontinuation of Treatment
Sixteen percent of 1,080 patients who received CLOZARIL® (clozapine) in premarketing clinical trials discontinued treatment due to an adverse event, including both those that could be reasonably attributed to CLOZARIL treatment and those that might more appropriately be considered intercurrent illness. The more common events considered to be causes of discontinuation included: CNS, primarily drowsiness/sedation, seizures, dizziness/syncope; cardiovascular, primarily tachycardia, hypotension and ECG changes; gastrointestinal, primarily nausea/vomiting; hematologic, primarily leukopenia/ granulocytopenia/ agranulocytosis; and fever. None of the events enumerated accounts for more than 1.7% of all discontinuations attributed to adverse clinical events.
Extrapyramidal Symptoms
Dystonia
Class Effect: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups. Clozapine, an atypical antipsychotic, is associated with a low incidence of dystonia (see WARNINGS, Tardive Dyskinesia).
Commonly Observed
Adverse events observed in association with the use of CLOZARIL in clinical trials at an incidence of greater than 5% were: central nervous system complaints, including drowsiness/sedation, dizziness/vertigo, headache and tremor; autonomic nervous system complaints, including salivation, sweating, dry mouth and visual disturbances; cardiovascular findings, including tachycardia, hypotension and syncope; and gastrointestinal complaints, including constipation and nausea; and fever. Complaints of drowsiness/sedation tend to subside with continued therapy or dose reduction. Salivation may be profuse, especially during sleep, but may be diminished with dose reduction.
Incidence in Clinical Trials
The following table enumerates adverse events that occurred at a frequency of 1% or greater among CLOZARIL patients who participated in clinical trials. These rates are not adjusted for duration of exposure.
| Body System
Adverse Eventa | Percent |
| Central Nervous System | |
| Drowsiness/Sedation | 39 |
| Dizziness/Vertigo | 19 |
| Headache | 7 |
| Tremor | 6 |
| Syncope | 6 |
| Disturbed Sleep/Nightmares | 4 |
| Restlessness | 4 |
| Hypokinesia/Akinesia | 4 |
| Agitation | 4 |
| Seizures (convulsions) | 3b |
| Rigidity | 3 |
| Akathisia | 3 |
| Confusion | 3 |
| Fatigue | 2 |
| Insomnia | 2 |
| Hyperkinesia | 1 |
| Weakness | 1 |
| Lethargy | 1 |
| Ataxia | 1 |
| Slurred Speech | 1 |
| Depression | 1 |
| Epileptiform Movements/Myoclonic Jerks | 1 |
| Anxiety | 1 |
| Cardiovascular | |
| Tachycardia | 25b |
| Hypotension | 9 |
| Hypertension | 4 |
| Chest Pain/Angina | 1 |
| ECG Change/Cardiac Abnormality | 1 |
| Gastrointestinal | |
| Constipation | 14 |
| Nausea | 5 |
| Abdominal Discomfort/Heartburn | 4 |
| Nausea/Vomiting | 3 |
| Vomiting | 3 |
| Diarrhea | 2 |
| Liver test Abnormality | 1 |
| Anorexia | 1 |
| Urogenital | |
| Urinary Abnormalities | 2 |
| Incontinence | 1 |
| Abnormal Ejaculation | 1 |
| Urinary Urgency/Frequency | 1 |
| Urinary Retention | 1 |
| Autonomic Nervous System | |
| Salivation | 31 |
| Sweating | 6 |
| Dry Mouth | 6 |
| Visual Disturbances | 5 |
| Integumentary (Skin) | |
| Rash | 2 |
| Musculoskeletal | |
| Muscle Weakness | 1 |
| Pain (Back, Neck, Legs) | 1 |
| Muscle Spasm | 1 |
| Muscle Pain, Ache | 1 |
| Respiratory | |
| Throat Discomfort | 1 |
| Dyspnea, Shortness of Breath | 1 |
| Nasal Congestion | 1 |
| Hemic/Lymphatic | |
| Leukopenia/Decreased WBC/Neutropenia | 3 |
| Agranulocytosis | 1b |
| Eosinophilia | 1 |
| Miscellaneous | |
| Fever | 5 |
| Weight Gain | 4 |
| Tongue Numb/Sore | 1 |
| a Events reported by at least 1% of CLOZARIL patients are included. | |
| b Rate based on population of approximately 1,700 exposed during premarket clinical evaluation of CLOZARIL. | |
The following table enumerates adverse events that occurred at a frequency of 10% for either treatment group in patients who took at least 1 dose of study medication during their participation in InterSePT, which was an adequate and well-controlled 2-year study evaluating the efficacy of CLOZARIL relative to Zyprexa in reducing the risk of emergent suicidal behavior in patients with schizophrenia or schizoaffective disorder. These rates are not adjusted for duration of exposure.
| Clozaril®
N=479 % Reporting | Zyprexa®
N=477 % Reporting |
|
| Adverse Events | ||
| Salivary hypersecretion | 48% | 6% |
| Somnolence | 46% | 25% |
| Weight increased | 31% | 56% |
| Dizziness (excluding vertigo) | 27% | 12% |
| Constipation | 25% | 10% |
| Insomnia NEC | 20% | 33% |
| Nausea | 17% | 10% |
| Vomiting NOS | 17% | 9% |
| Dyspepsia | 14% | 8% |
| 1AEs are listed by frequency in CLOZARIL group, and included in the table are those for which the risk ratio of CLOZARIL over Zyprexa or of Zyprexa over CLOZARIL was greater than 1.5. | ||
| NEC - not elsewhere classified | ||
| NOS - not otherwise specified | ||
Other Events Observed During the Premarketing Evaluation of CLOZARIL® (clozapine)
This section reports additional, less frequent adverse events which occurred among the patients taking CLOZARIL in clinical trials. Various adverse events were reported as part of the total experience in these clinical studies; a causal relationship to CLOZARIL treatment cannot be determined in the absence of appropriate controls in some of the studies. The table above enumerates adverse events that occurred at a frequency of at least 1% of patients treated with CLOZARIL. The list below includes all additional adverse experiences reported as being temporally associated with the use of the drug which occurred at a frequency less than 1%, enumerated by organ system.
Central Nervous System: loss of speech, amentia, tics, poor coordination, delusions/hallucinations, involuntary movement, stuttering, dysarthria, amnesia/memory loss, histrionic movements, libido increase or decrease, paranoia, shakiness, Parkinsonism, and irritability.
Cardiovascular System: edema, palpitations, phlebitis/thrombophlebitis, cyanosis, premature ventricular contraction, bradycardia, and nosebleed.
Gastrointestinal System: abdominal distention, gastroenteritis, rectal bleeding, nervous stomach, abnormal stools, hematemesis, gastric ulcer, bitter taste, and eructation.
Urogenital System: dysmenorrhea, impotence, breast pain/discomfort, and vaginal itch/infection.
Autonomic Nervous System: numbness, polydipsia, hot flashes, dry throat, and mydriasis.
Integumentary (Skin): pruritus, pallor, eczema, erythema, bruise, dermatitis, petechiae, and urticaria.
Musculoskeletal System: twitching and joint pain.
Respiratory System: coughing, pneumonia/pneumonia-like symptoms, rhinorrhea, hyperventilation, wheezing, bronchitis, laryngitis, and sneezing.
Hemic and Lymphatic System: anemia and leukocytosis.
Miscellaneous: chills/chills with fever, malaise, appetite increase, ear disorder, hypothermia, eyelid disorder, bloodshot eyes, and nystagmus.
Postmarketing Clinical Experience
Postmarketing experience has shown an adverse experience profile similar to that presented above. Voluntary reports of adverse events temporally associated with CLOZARIL not mentioned above that have been received since market introduction and that may have no causal relationship with the drug include the following:
Central Nervous System: delirium; EEG abnormal; exacerbation of psychosis; myoclonus; overdose; paresthesia; possible mild cataplexy, and status epilepticus, and obsessive compulsive symptoms.
Cardiovascular System: atrial or ventricular fibrillation and periorbital edema.
Gastrointestinal System: acute pancreatitis; dysphagia; fecal impaction; intestinal obstruction/paralytic ileus; and salivary gland swelling.
Hepatobiliary System: cholestasis; hepatitis; jaundice.
Hepatic System: cholestasis.
Urogenital System: acute interstitial nephritis and priapism.
Integumentary (Skin): hypersensitivity reactions: photosensitivity, vasculitis, erythema multiforme, and Stevens-Johnson Syndrome.
Metabolic and Nutritional Disorders: hypercholesterolemia, hypertriglyceridemia and new onset diabetes.
Musculoskeletal System: myasthenic syndrome and rhabdomyolysis.
Respiratory System: aspiration, pleural effusion, and pneumonia and lower respiratory tract infection which may be fatal.
Hemic and Lymphatic System: deep vein thrombosis; elevated hemoglobin/hematocrit; ESR increased; pulmonary embolism; sepsis; thrombocytosis; and thrombocytopenia.
Vision Disorders: narrow angle glaucoma.
Miscellaneous: CPK elevation; hyperglycemia; hyperuricemia; hyponatremia; and weight loss.
DRUG ABUSE AND DEPENDENCE
Physical and psychological dependence have not been reported or observed in patients taking CLOZARIL® (clozapine).
OVERDOSAGE
Human Experience
The most commonly reported signs and symptoms associated with CLOZARIL® (clozapine) overdose are: altered states of consciousness, including drowsiness, delirium and coma; tachycardia; hypotension; respiratory depression or failure; hypersalivation. Aspiration pneumonia and cardiac arrhythmias have also been reported. Seizures have occurred in a minority of reported cases. Fatal overdoses have been reported with CLOZARIL, generally at doses above 2500 mg. There have also been reports of patients recovering from overdoses well in excess of 4 g.
Management of Overdose
Establish and maintain an airway; ensure adequate oxygenation and ventilation. Activated charcoal, which may be used with sorbitol, may be as or more effective than emesis or lavage, and should be considered in treating overdosage. Cardiac and vital signs monitoring is recommended along with general symptomatic and supportive measures. Additional surveillance should be continued for several days because of the risk of delayed effects. Avoid epinephrine and derivatives when treating hypotension, and quinidine and procainamide when treating cardiac arrhythmia.
There are no specific antidotes for CLOZARIL. Forced diuresis, dialysis, hemoperfusion and exchange transfusion are unlikely to be of benefit.
In managing overdosage, the physician should consider the possibility of multiple drug involvement.
Up-to-date information about the treatment of overdose can often be obtained from a certified Regional Poison Control Center. Telephone numbers of certified Poison Control Centers are listed in the Physicians’ Desk Reference®.**
DOSAGE AND ADMINISTRATION
Treatment-Resistant Schizophrenia
Upon initiation of CLOZARIL® (clozapine) therapy, up to a 1-week supply of additional CLOZARIL tablets may be provided to the patient to be held for emergencies (e.g., weather, holidays).
Initial Treatment:
It is recommended that treatment with CLOZARIL begin with one-half of a 25-mg tablet (12.5 mg) once or twice daily and then be continued with daily dosage increments of 25-50 mg/day, if well tolerated, to achieve a target dose of 300-450 mg/day by the end of 2 weeks. Subsequent dosage increments should be made no more than once or twice weekly, in increments not to exceed 100 mg. Cautious titration and a divided dosage schedule are necessary to minimize the risks of hypotension, seizure, and sedation.
In the multicenter study that provides primary support for the effectiveness of CLOZARIL in patients resistant to standard drug treatment for schizophrenia, patients were titrated during the first 2 weeks up to a maximum dose of 500 mg/day, on a t.i.d. basis, and were then dosed in a total daily dose range of 100-900 mg/day, on a t.i.d. basis thereafter, with clinical response and adverse effects as guides to correct dosing.
Therapeutic Dose Adjustment:
Daily dosing should continue on a divided basis as an effective and tolerable dose level is sought. While many patients may respond adequately at doses between 300-600 mg/day, it may be necessary to raise the dose to the 600-900 mg/day range to obtain an acceptable response. (Note: In the multicenter study providing the primary support for the superiority of CLOZARIL in treatment-resistant patients, the mean and median CLOZARIL doses were both approximately 600 mg/day.)
Because of the possibility of increased adverse reactions at higher doses, particularly seizures, patients should ordinarily be given adequate time to respond to a given dose level before escalation to a higher dose is contemplated. CLOZARIL can cause EEG changes, including the occurrence of spike and wave complexes. It lowers the seizure threshold in a dose-dependent manner and may induce myoclonic jerks or generalized seizures. These symptoms may be likely to occur with rapid dose increase and in patients with preexisting epilepsy. In this case, the dose should be reduced and, if necessary, anticonvulsant treatment initiated.
Dosing should not exceed 900 mg/day.
Because of the significant risk of agranulocytosis and seizure, events which both present a continuing risk over time, the extended treatment of patients failing to show an acceptable level of clinical response should ordinarily be avoided.
Maintenance Treatment:
While the maintenance effectiveness of CLOZARIL in schizophrenia is still under study, the effectiveness of maintenance treatment is well established for many other drugs used to treat schizophrenia. It is recommended that responding patients be continued on CLOZARIL, but at the lowest level needed to maintain remission. Because of the significant risk associated with the use of CLOZARIL, patients should be periodically reassessed to determine the need for maintenance treatment.
Discontinuation of Treatment:
In the event of planned termination of CLOZARIL therapy, gradual reduction in dose is recommended over a 1-2-week period. However, should a patient’s medical condition require abrupt discontinuation (e.g., leukopenia), the patient should be carefully observed for the recurrence of psychotic symptoms and symptoms related to cholinergic rebound such as headache, nausea, vomiting, and diarrhea.
Reinitiation of Treatment in Patients Previously Discontinued:
When restarting patients who have had even a brief interval off CLOZARIL, i.e., 2 days or more since the last dose, it is recommended that treatment be reinitiated with one-half of a 25-mg tablet (12.5 mg) once or twice daily (see WARNINGS). If that dose is well tolerated, it may be feasible to titrate patients back to a therapeutic dose more quickly than is recommended for initial treatment. However, any patient who has previously experienced respiratory or cardiac arrest with initial dosing, but was then able to be successfully titrated to a therapeutic dose, should be retitrated with extreme caution after even 24 hours of discontinuation.
Certain additional precautions seem prudent when reinitiating treatment. The mechanisms underlying CLOZARIL-induced adverse reactions are unknown. It is conceivable, however, that reexposure of a patient might enhance the risk of an untoward event’s occurrence and increase its severity. Such phenomena, for example, occur when immune-mediated mechanisms are responsible. Consequently, during the reinitiation of treatment, additional caution is advised. Patients discontinued for WBC counts below 2000/mm3 or an ANC below 1000/mm3 must not be restarted on CLOZARIL. (See WARNINGS.)
Reducing the Risk of Recurrent Suicidal Behavior in Patients with Schizophrenia or Schizoaffective Disorder
The dosage and administration recommendations outlined above regarding the use of CLOZARIL in patients with treatment-resistant schizophrenia should also be followed when treating patients with schizophrenia or schizoaffective disorder at risk for recurrent suicidal behavior.
The InterSePT study demonstrated the efficacy of CLOZARIL in treatment of patients with schizophrenia or schizoaffective disorder at risk for recurrent suicidal behavior where the mean daily dose was about 300 mg (range 12.5 to 900 mg).
Patients previously treated with other antipsychotics were cross-titrated to CLOZARIL over a one-month interval; the dose of the previous antipsychotic was gradually decreased simultaneous with a gradual increase in CLOZARIL dose over the first month of the study. Patients on depot antipsychotic medication began CLOZARIL after one full dosing interval since the last injection.
Recommendations to Reduce the Risk of Recurrent Suicidal Behavior in Patients Who Otherwise Previously Responded to Treatment of Schizophrenia or Schizoaffective Disorder with Another Antipsychotic Medication:
The results of the InterSePT study demonstrated that, for a 2-year treatment period, the probability of a suicide attempt or a hospitalization due to imminent suicide risk is stable at approximately 24% after one year of treatment with CLOZARIL (Figure 1, Clinical Trial Data Section). A course of treatment with CLOZARIL of at least 2 years is therefore recommended in order to maintain the reduction of risk for suicidal behavior. After 2 years, it is recommended that the patient’s risk of suicidal behavior be assessed. If the physician’s assessment indicates that a significant risk for suicidal behavior is still present, treatment with CLOZARIL should be continued. Thereafter, the decision to continue treatment with CLOZARIL should be revisited at regular intervals, based on thorough assessments of the patient’s risk for suicidal behavior during treatment. If the physician determines that the patient is no longer at risk for suicidal behavior, treatment with CLOZARIL may be discontinued (see recommendations above regarding discontinuation of treatment) and treatment of the underlying disorder with an antipsychotic medication to which the patient has previously responded may be resumed.
HOW SUPPLIED
CLOZARIL® (clozapine) is available as 25 mg and 100 mg round, pale-yellow, uncoated tablets with a facilitated score on one side.
CLOZARIL® (clozapine) Tablets
25 mg
Engraved with “CLOZARIL” once on the periphery of one side.
Engraved with a facilitated score and “25” once on the other side.
Bottle of 100 NDC 0078-0126-05
Bottle of 500 NDC 0078-0126-08
Unit dose packages of 100: 2 x 5 strips, 10 blisters per strip NDC 0078-0126-06
100 mg
Engraved with “CLOZARIL” once on the periphery of one side.
Engraved with a facilitated score and “100” once on the other side.
Bottle of 100 NDC 0078-0127-05
Bottle of 500 NDC 0078-0127-08
Unit dose packages of 100: 2 x 5 strips, 10 blisters per strip NDC 0078-0127-06
Store and Dispense
Storage temperature should not exceed 30°C (86°F). Drug dispensing should not ordinarily exceed a weekly supply.
If a patient is eligible for White Blood Cell (WBC) count and Absolute Neutrophil Count (ANC) testing every 2 weeks, then a two-week supply of CLOZARIL can be dispensed. If a patient is eligible for WBC count and ANC testing every 4 weeks, then a four-week supply of CLOZARIL can be dispensed. Dispensing should be contingent upon the WBC count and ANC test results.
*Zyprexa® (olanzapine) is a registered trademark of Eli Lilly and Company.
**Trademark of Thomson Healthcare, Inc.
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
REV: DECEMBER 2010 T2010-122
© Novartis
PRINCIPAL DISPLAY PANEL
Package Label – 25 mg
Rx Only NDC 0078-0126-06
CLOZARIL® (clozapine)
Unit Dose Package
100 Tablets
This unit dose package is intended for institutional
In-patient use only. If dispensed for out-patient use,
an appropriate safety closure should be provided.

PRINCIPAL DISPLAY PANEL
Package Label – 100 mg
Rx Only NDC 0078-0127-06
CLOZARIL® (clozapine)
Unit Dose Package
100 Tablets
This unit dose package is intended for institutional
In-patient use only. If dispensed for out-patient use,
an appropriate safety closure should be provided

| CLOZARIL
clozapine tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA019758 | 09/30/1989 | |
| CLOZARIL
clozapine tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA019758 | 09/30/1989 | |
| Labeler - Novartis Pharmaceuticals Corporation (002147023) |