CETIRIZINE HYDROCHLORIDE
-
cetirizine hydrochloride tablet
Unichem Pharmaceuticals (USA), Inc.
----------
Cetirizine Hydrochloride TabletsACTIVE INGREDIENT (IN EACH TABLET)
For 5 mg
For 10 mg
PURPOSE
USES
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
WARNINGS
Do not use
if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have
liver or kidney disease. Your doctor should determine if you need a different dose.
Ask a doctor or pharmacist before use if you are
taking tranquilizers or sedatives.
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if
an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding:
- if breast-feeding : not recommended
- if pregnant: ask a health professional before use
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
DIRECTIONS
For 5 mg
| adults and children 6 years and over | 1 to 2 tablets once daily depending upon severity of symptoms; do not take more than 2 tablets in 24 hours. |
| adults 65 years and over | 1 tablet once a day; do not take more than 1 tablet in 24 hours. |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
For 10 mg
| adults and children 6 years and over | one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| adults 65 years and over | ask a doctor |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
OTHER INFORMATION
- store between 200 to 250C (680 to 770F)
INACTIVE INGREDIENT
corn starch, hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone and titanium dioxide
QUESTIONS?
UNICHEM LABORATORIES LTD.
Pilerne Ind. Estate, Pilerne, Bardez, Goa 403 511, India
Marketed by:

PRINCIPAL DISPLAY PANEL
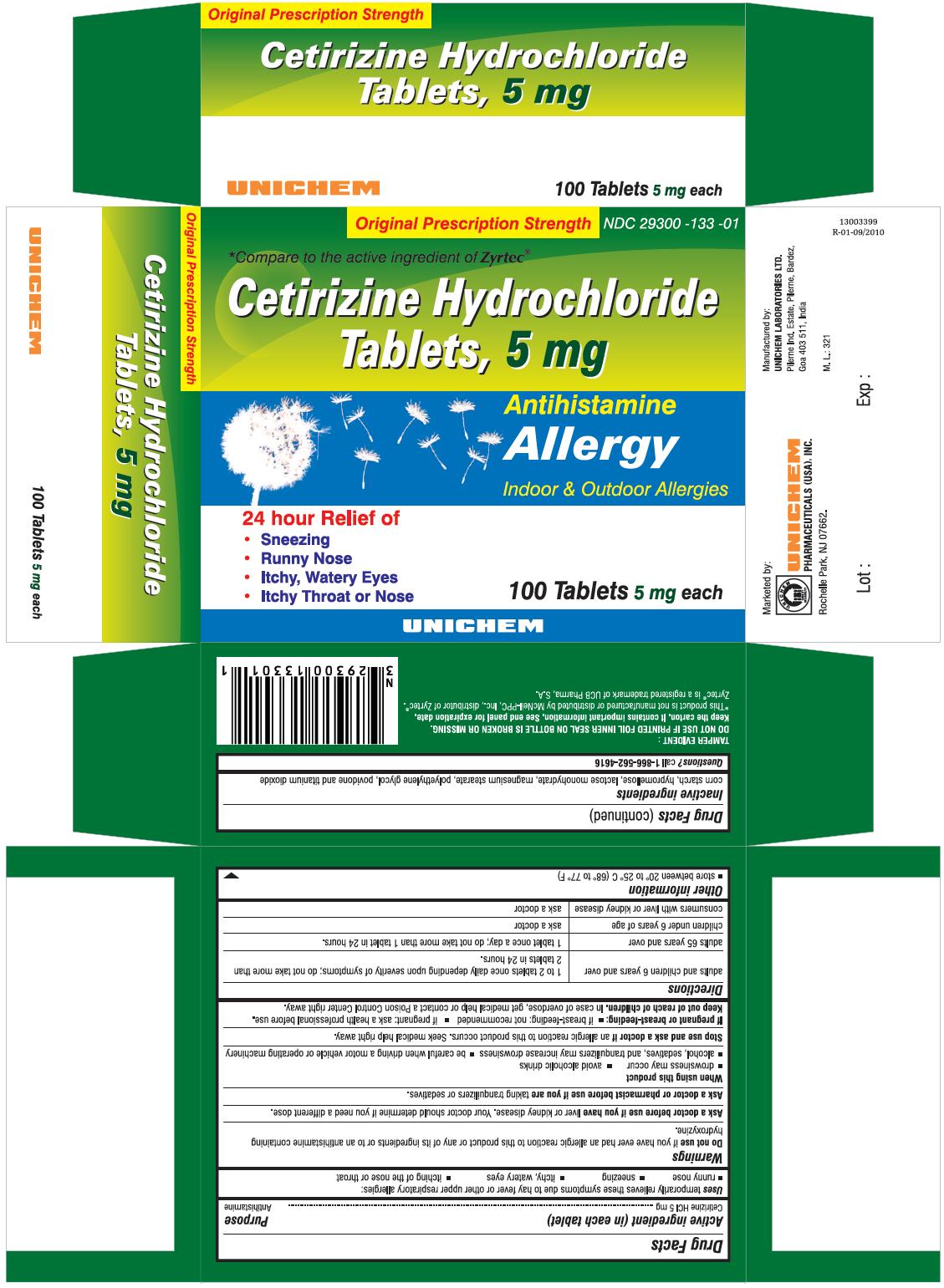
Cetirizine Hydrochloride Tablets 5 mg - Allergy
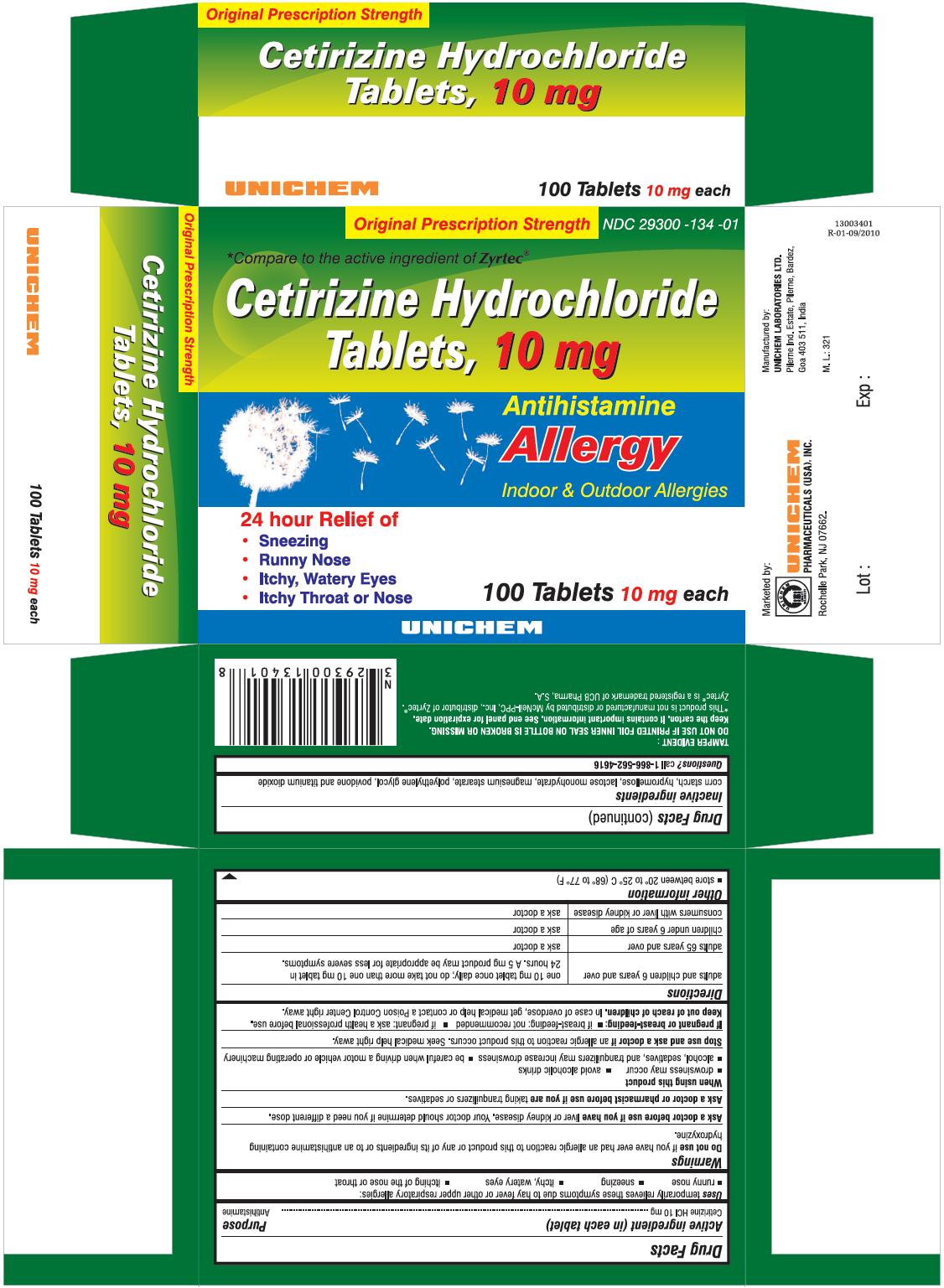
Cetirizine Hydrochloride Tablets 10 mg - Allergy
ACTIVE INGREDIENT (IN EACH TABLET)
For 5 mg
For 10 mg
PURPOSE
USES
relieves itching due to hives (urticaria). This product will not prevent hives or an allergic skin reaction from occurring.
WARNINGS
Severe Allergy Warning
Get emergency help immediately if you have hives along with any of the following symptoms:
- trouble swallowing
- dizziness or loss of consciousness
- swelling of tongue
- swelling in or around mouth
- trouble speaking
- drooling
- wheezing or problems breathing
These symptoms may be signs of anaphylactic shock. This condition can be life threatening if not treated by a health professional immediately. Symptoms of anaphylactic shock may occur when hives first appear or up to a few hours later.
Not a Substitute for Epinephrine.
If your doctor has prescribed an epinephrine injector for "anaphylaxis" or severe allergy symptoms that could occur with your hives, never use this product as a substitute for the epinephrine injector. If you have been prescribed an epinephrine injector, you should carry it with you at all times.
Do not use
▪ to prevent hives from any known cause such as:
- foods
- insect stings
- medicines
- latex or rubber gloves
because this product will not stop hives from occuring. Avoiding the cause of your hives is the only way to prevent them. Hives can sometimes be serious. If you do not know the cause of your hives, see your doctor for a medical exam. Your doctor may be able to help you find a cause.
▪ if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have
- liver or kidney disease. Your doctor should determine if you need a different dose.
- hives that are an unusual color, look bruised or blistered.
- hives that do not itch.
Ask a doctor or pharmacist before use if you are
taking tranquilizers or sedatives.
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives and tranquilizers may increase drowsiness.
- be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve after 3 days of treatment
- the hives have lasted more than 6 weeks.
If pregnant or breast-feeding:
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
DIRECTIONS
For 5 mg
| adults and children 6 years
| 1 to 2 tablets once daily depending upon
|
| and over | severity of symptoms; do not take more than |
|
| 2 tablets in 24 hours. |
| adults 65 years and over
| 1 tablet once a day; do not take more than |
|
| 1 tablet in 24 hours. |
| children under 6 years of age
| ask a doctor |
| consumers with liver or
| ask a doctor |
| kidney disease |
|
For 10 mg
| adults and children 6 years
| one 10 mg tablet once daily; do not take more
|
| and over | than one 10 mg tablet in 24 hours. A 5 mg |
|
| product may be appropriate for less severe |
|
| symptoms. |
| adults 65 years and over
| ask a doctor |
| children under 6 years of age
| ask a doctor |
| consumers with liver or
| ask a doctor |
| kidney disease |
|
OTHER INFORMATION
- store between 20° to 25° C (68° to 77° F)
INACTIVE INGREDIENT
corn starch, hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone and titanium dioxide
QUESTIONS?
UNICHEM LABORATORIES LTD.
Pilerne Ind. Estate, Pilerne, Bardez, Goa 403 511, India
Marketed by:

PRINCIPAL DISPLAY PANEL

Cetirizine Hydrochloride Tablets 5 mg - Hives Relief

Cetirizine Hydrochloride Tablets 10 mg - Hives Relief
| CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tablet |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA078680 | 11/30/2010 | |
| CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tablet |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA078680 | 11/30/2010 | |
| Labeler - Unichem Pharmaceuticals (USA), Inc. (181620514) |