GREEN GUARD SORE THROAT RELIEF
-
hexylresorcinol and
menthol lozenge
Unifirst First Aid Corporation
----------
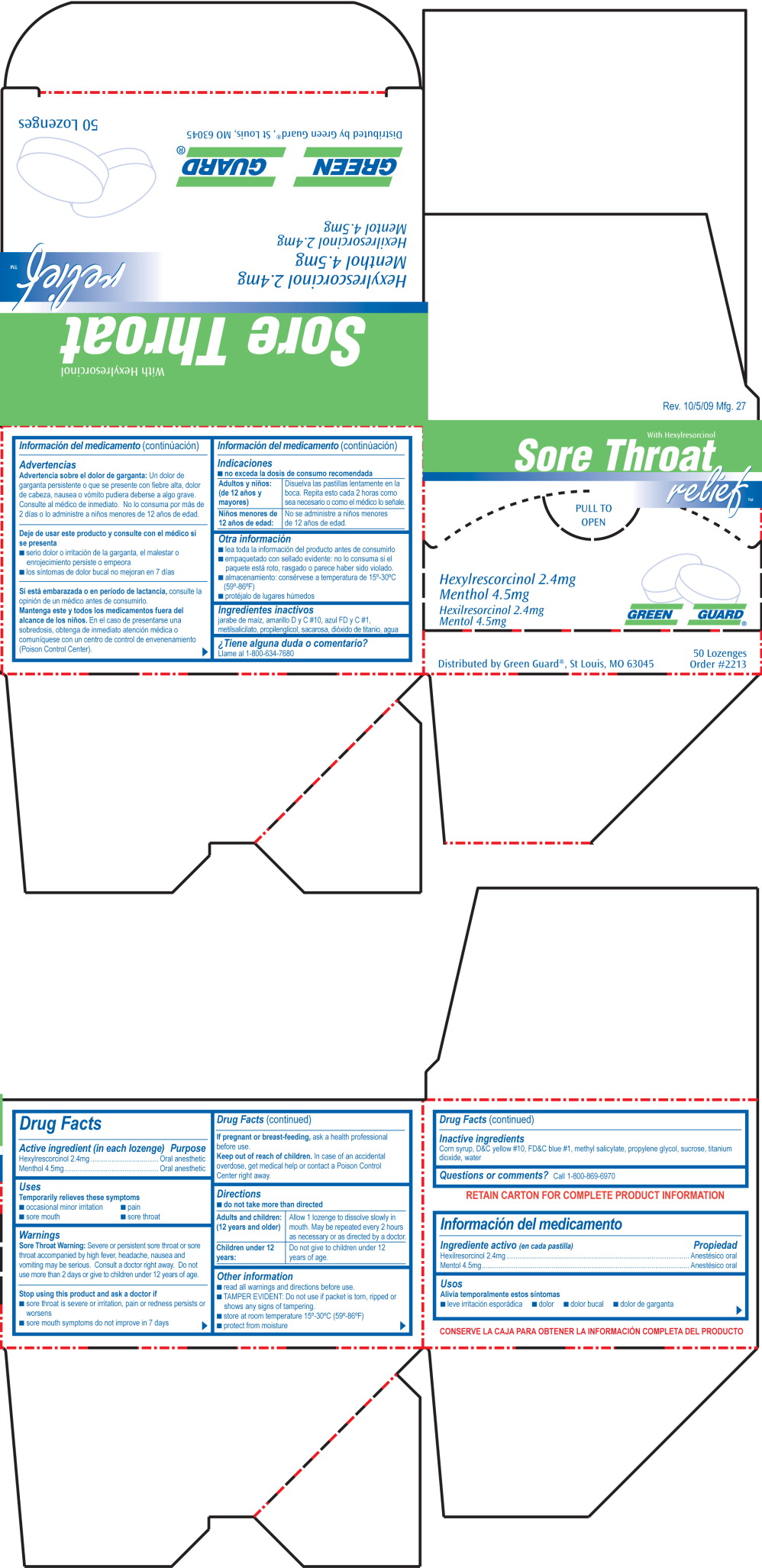
Green Guard Sore Throat ReliefDrug Facts
Active ingredient (in each lozenge)
Hexylrescorcinol 2.4mg
Menthol 4.5mg
Purpose
Oral anesthetic
Oral anesthetic
Uses
Temporarily relieves these symptoms
- occasional minor irritation
- pain
- sore mouth
- sore throat
Warnings
Sore Throat Warning: Severe or persistent sore throat or sore throat accompanied by high fever, headache, nausea and vomiting may be serious. Consult a doctor right away. Do not use more than 2 days or give to children under 12 years of age.
Stop using this product and ask a doctor if
- sore throat is severe or irritation, pain or redness persists or worsens
- sore mouth symptoms do not improve in 7 days
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of an accidental overdose, get medical help or contact a Poison Control Center right away.
Directions
- do not take more than directed
Adults and children: (12 years and older) Allow 1 lozenge to dissolve slowly in mouth. May be repeated every 2 hours as necessary or as directed by a doctor.
Children under 12 years: Do not give to children under 12 years of age.
Other information
- read all warnings and directions before use.
- TAMPER EVIDENT: Do not use if packet is torn, ripped or shows any signs of tampering.
- store at room temperature 15º-30ºC (59º-86ºF)
- protect from moisture
Inactive ingredients
Corn syrup, D&C yellow #10, FD&C blue #1, methyl salicylate, propylene glycol, sucrose, titanium dioxide, water
Questions or comments? Call 1-800-869-6970
909B Green Guard Sore Throat Relief Label
With Hexylresorcinol
Sore Throat relief™
Pull to Open
Hexylrescorcinol 2.4 mg
Menthol 4.5 mg
Hexilresorcinol 2.4 mg
Mentol 4.5 mg
Green Guard®
50 Lozenges
Order #2213
Distributed by Green Guard®, St Louis, MO 6345
| GREEN GUARD SORE THROAT RELIEF
hexylresorcinol, menthol lozenge |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part356 | 12/30/2008 | |
| Labeler - Unifirst First Aid Corporation (832947092) |