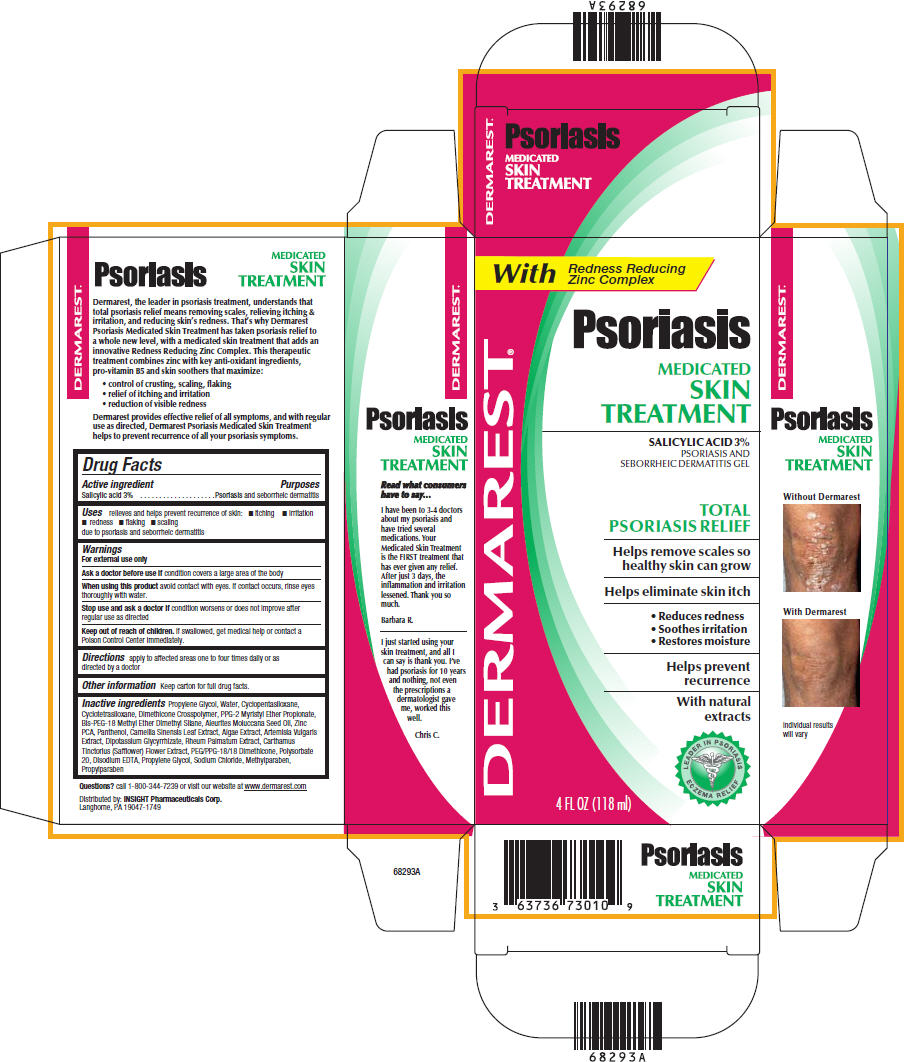
DERMAREST PSORIASIS MEDICATED SKIN TREATMENT
-
salicylic acid gel
Insight Pharmaceuticals
----------
PsoriasisMEDICATED
SKIN
TREATMENT
Drug Facts
Active ingredient
Salicylic acid 3%
Purposes
Psoriasis and seborrheic dermatitis
Uses
relieves and helps prevent recurrence of skin:
- itching
- irritation
- redness
- flaking
- scaling
due to psoriasis and seborrheic dermatitis
Warnings
For external use only
Ask a doctor before use if condition covers a large area of the body
When using this product avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
Stop use and ask a doctor if condition worsens or does not improve after regular use as directed
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Directions
apply to affected areas one to four times daily or as directed by a doctor
Other information
Keep carton for full drug facts.
Inactive ingredients
Propylene Glycol, Water, Cyclopentasiloxane, Cyclotetrasiloxane, Dimethicone Crosspolymer, PPG-2 Myristyl Ether Propionate, Bis-PEG-18 Methyl Ether Dimethyl Silane, Aleurites Moluccana Seed Oil, Zinc PCA, Panthenol, Camellia Sinensis Leaf Extract, Algae Extract, Artemisia Vulgaris Extract, Dipotassium Glycyrrhizate, Rheum Palmatum Extract, Carthamus Tinctorius (Safflower) Flower Extract, PEG/PPG-18/18 Dimethicone, Polysorbate 20, Disodium EDTA, Propylene Glycol, Sodium Chloride, Methylparaben, Propylparaben
Questions?
call 1-800-344-7239 or visit our website at www.dermarest.com
Distributed by: INSIGHT Pharmaceuticals Corp.
Langhorne, PA 19047-1749
PRINCIPAL DISPLAY PANEL - 118 ml Carton
With
Redness Reducing
Zinc Complex
DERMAREST®
Psoriasis
MEDICATED
SKIN
TREATMENT
SALICYLIC ACID 3%
PSORIASIS AND
SEBORRHEIC DERMATITIS GEL
TOTAL
PSORIASIS RELIEF
Helps remove scales so
healthy skin can grow
Helps eliminate skin itch
- Reduces redness
- Soothes irritation
- Restores moisture
Helps prevent
recurrence
With natural extracts
LEADER IN PSORIASIS
ECZEMA RELIEF
4 FL OZ (118 ml)
| DERMAREST
PSORIASIS MEDICATED SKIN TREATMENT
salicylic acid gel |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC MONOGRAPH NOT FINAL | part358 | 10/20/2010 | |
| Labeler - Insight Pharmaceuticals (176792315) |