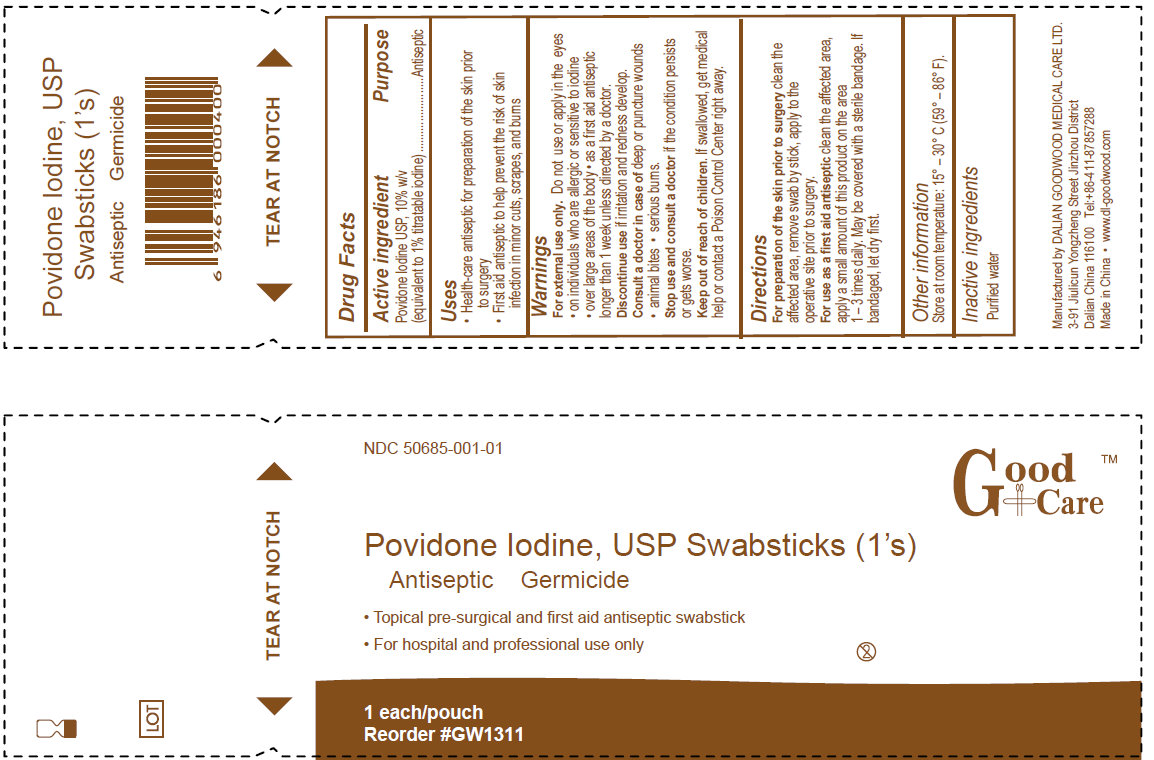
GOOD CARE POVIDONE IODINE
-
povidone-iodine swab
Dalian Goodwood Medical Care Ltd.
----------
Povidone Iodine, USP SwabsticksActive Ingredient
Povidone Iodine USP, 10% w/v
(equivalent to 1% titratable iodine)
Purpose
Antiseptic
Uses
- Health-care antiseptic for preparation of the skin prior to surgery
- First aid antiseptic to help prevent the risk of skin infection in minor cuts, scrapes and burns
Warnings
For external use only.
Do not use or apply in the eyes- on individuals who are allergic or sensitive to iodine
- over large areas of the body
- as a first aid antiseptic longer than 1 week unless directed by a doctor
Stop use and consult a doctor if the condition persists or gets worse.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
For preparation of the skin prior to surgery clean the affected area, remove swab by the stick, apply to the operative site prior to surgery.
For use as a first aid antiseptic clean the affected area, apply a small amount of this product on the area 1-3 times daily. May be covered with a sterile bandage. If bandaged, let dry first.
Other information
Store at room temperature: 15 degrees - 30 degrees C (59 degrees - 86 degrees F)
Inactive ingredients
citric acid, glycerin, nonoxynol-8, purified water, sodium hydroxide
3-91 Jiulicun Yongzheng Street Jinzhou District Dalian China 116100
Tel: +86-411-87857288
Made in China www.dl-goodwood.com
Povidone Iodine, USP Swabsticks
Antiseptic Germicide
- Topical pre-surgical and first aid antiseptic swabstick
- For hospital and professional use only
| GOOD CARE POVIDONE IODINE
povidone-iodine swab |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part333A | 10/01/2010 | |
| Labeler - Dalian Goodwood Medical Care Ltd. (529575699) |
| Registrant - Dalian Goodwood Medical Care Ltd. (529575699) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Dalian Goodwood Medical Care Ltd. | 529575699 | manufacture | |