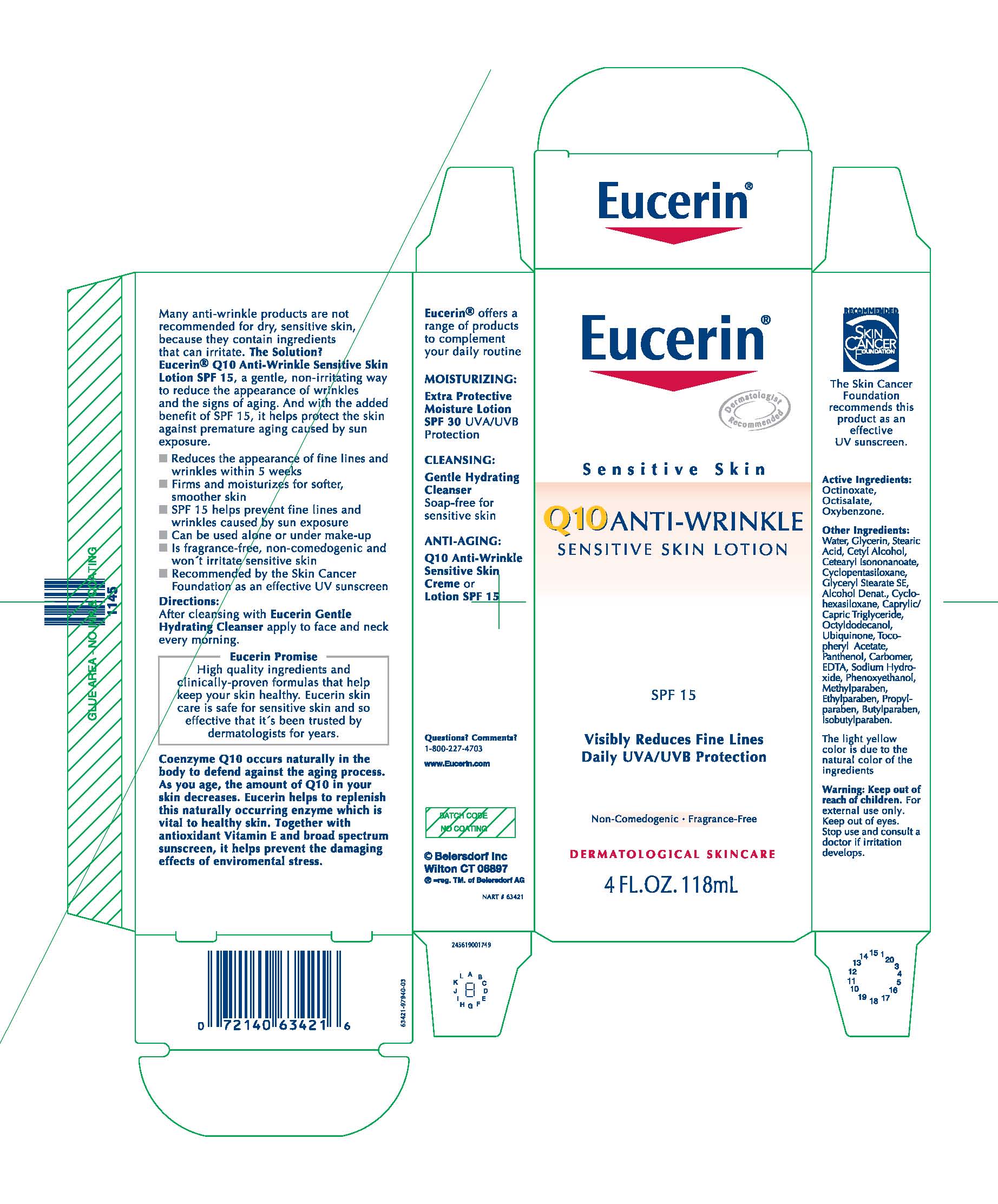
EUCERIN Q10 ANTI-WRINKLE SENSITIVE SKIN
-
octinoxate,
octisalate and
oxybenzone lotion
Beiersdorf Inc
----------
Eucerin Q10 Anti-Wrinkle Sensitive SkinOctinoxate, Octisalate, Oxybenzone.
Water, Glycerin, Stearic Acid, Cetyl Alcohol, Cetearyl Isononanoate, Cyclopentasiloxane, Glyceryl Stearate SE,
Alcohol Denat., Cyclohexasiloxane, Caprylic/Capric Triglyceride, Octyldodecanol, Ubiquinone, Tocopheryl
Acetate, Panthenol, Carbomer, EDTA, Sodium Hydroxide, Phenoxyethanol, Methylparaben, Ethylparaben, Propylparaben,
Butylparaben, Isobutylparaben.
Many anti-wrinkle products are not recommended for dry, sensitive skin,
because they contain ingredients that can irritate. The Solution?
Eucerin® Q10 Anti-Wrinkle Sensitive Skin Lotion SPF 15, a gentle, non-irritating way
to reduce the appearance of wrinkles and the signs of aging. And with the added
benefit of SPF 15, it helps protect the skin against premature aging caused by sun
exposure.
■ Reduces the appearance of fine lines and wrinkles within 5 weeks
■ Firms and moisturizes for softer, smoother skin
■ SPF 15 helps prevent fine lines and wrinkles caused by sun exposure
■ Can be used alone or under make-up
■ Is fragrance-free, non-comedogenic and won´t irritate sensitive skin
■ Recommended by the Skin Cancer Foundation as an effective UV sunscreen
Eucerin Promise
High quality ingredients and clinically-proven formulas that help
keep your skin healthy. Eucerin skin care is safe for sensitive skin and so
effective that it´s been trusted by dermatologists for years.
As you age, the amount of Q10 in your skin decreases. Eucerin helps to replenish
this naturally occurring enzyme which is vital to healthy skin. Together with
antioxidant Vitamin E and broad spectrum sunscreen, it helps prevent the damaging
effects of enviromental stress.
The Skin Cancer Foundation recommends this product as an
effective UV sunscreen.
The light yellow color is due to the natural color of the ingredients
Directions:
After cleansing with Eucerin Gentle
Hydrating Cleanser apply to face and neck
every morning.
Stop use and consult a doctor if irritation develops.
Keep out of reach of children.
For external use only.
Questions Comments
1-800-227-4703
Eucerin
Sensitive Skin
Q10 Anti-Wrinkle
Sensitive Skin Lotion
SPF 15
Dermatologist recommended
Visibly Reduces Fine LinesDaily UVA/UVB Protection
Non-Comedogenic · Fragrance-Free
DERMATOLOGICAL SKINCARE
4 FL.OZ. 118mL
| EUCERIN Q10 ANTI-WRINKLE SENSITIVE SKIN
octinoxate, octisalate, oxybenzone, lotion |
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part352 | 01/02/2001 | |
| Labeler - Beiersdorf Inc (001177906) |