ammonium lactate (Ammonium Lactate) lotion
[PERRIGO]
Rx only
For Dermatologic use only.
Not for ophthalmic, oral or intravaginal use.
DESCRIPTION
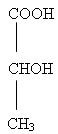
This product specially formulates 12% lactic acid neutralized with ammonium hydroxide, as ammonium lactate to provide a lotion pH of 4.5 - 5.5. It also contains light mineral oil, glyceryl stearate, PEG-100 stearate, propylene glycol, polyoxyl 40 stearate, glycerin, magnesium aluminum silicate, laureth-4, cetyl alcohol, methyl and propyl parabens, methylcellulose, fragrance and water. Lactic acid is a racemic mixture of 2-hydroxypropanoic acid and has the following structural formula:
CLINICAL PHARMACOLOGY
It is generally accepted that the water content of the stratum corneum is a controlling factor in maintaining skin flexibility. When the stratum corneum contains more than 10% water it remains soft and pliable; however, when the water content drops below 10% the stratum corneum becomes less flexible and rough, and may exhibit scaling and cracking and the underlying skin may become irritatied.1,2
Symptomatic relief of dry skin is provided by skin protectants containing hygroscopic substances (humectants) which increase skin moisture. Lactic acid, an α-hydroxy acid, is reported to be one of the most effective naturally occurring humectants in the skin3. The α-hydroxy acids (and their salts), in addition to having beneficial effects on dry skin, have also been shown to reduce excessive epidermal keratinization in patients with hyperkeratotic conditions (e.g., ichthyosis).4
Pharmacokinetics
The mechanism of action of topically applied neutralized lactic acid is not yet known.
Indications and Usage
Ammonium lactate lotion, 12% is indicated for the treatment of dry, scaly skin (xerosis) and ichthyosis vulgaris and for the temporary relief of itching associated with these conditions.
CONTRAINDICATIONS
Known hypersensitivity to any of the label ingredients.
WARNING
Sun exposure (natural or artificial sunlight) to areas of the skin treated with ammonium lactate lotion, 12% should be minimized or avoided (see PRECAUTIONS section).
PRECAUTIONS
General
For external use only. Stinging or burning may occur when applied to skin with fissures, erosions, or that is otherwise abraded (for example, after shaving the legs). Caution is advised when used on the face because of the potential for irritation. The potential for post-inflammatory hypo- or hyperpigmentation has not been studied.
Information for patients
Patients using ammonium lactate lotion, 12% should receive the following information and instructions:
- This medication is to be used as directed by the physician, and should not be used for any disorder other than for which it was prescribed. It is for external use only. Avoid contact with eyes, lips, or mucous membranes.
- Patients should minimize or avoid use of this product on areas of the skin that may be exposed to natural or artificial sunlight, including the face. If sun exposure is unavoidable, clothing should be worn to protect the skin.
- This medication may cause transient stinging or burning when applied to skin with fissures, erosions, or abrasions (for example, after shaving the legs).
- If the skin condition worsens with treatment, the medication should be promptly discontinued.
Carcinogenesis, Mutagenesis, Impairment of Fertility
The topical treatment of CD-1 mice with 12%, 21% or 30% ammonium lactate formulations for two-years did not produce a significant increase in dermal or systemic tumors in the absence of increased exposure to ultraviolet radiation. The maximum systemic exposure of the mice in this study was 0.7 times the maximum possible systemic exposure in humans. However, a long-term photocarcinogenicity study in hairless albino mice suggested that topically applied 12% ammonium lactate formulations enhanced the rate of ultraviolet light-induced skin tumor formation.
The mutagenic potential of ammonium lactate formulations was evaluated in the Ames assay and in the mouse in vivo micronucleus assay, both of which were negative.
In dermal Segment I and III studies with ammonium lactate formulations there were no effects observed in fertility or pre- or post-natal development parameters in rats at dose levels of 300 mg/kg/day (1800 mg/m2/day), approximately 0.4 times the human topical dose.
Pregnancy
Teratogenic effects
Pregnancy Category B
Animal reproduction studies have been performed in rats and rabbits at doses up to 0.7 and 1.5 times the human dose, respectively (600 mg/kg/day, corresponding to 3600 mg/m2/day in the rat and 7200 mg/m2/day in the rabbit) and have revealed no evidence of impaired fertility or harm to the fetus due to ammonium lactate formulations. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, ammonium lactate lotion, 12% should be used during pregnancy only if clearly needed.
Nursing Mothers
Although lactic acid is a normal constituent of blood and tissues, it is not known to what extent this drug affects normal lactic acid levels in human milk. Because many drugs are excreted in human milk, caution should be exercised when ammonium lactate is administered to a nursing woman.
Pediatric Use
Safety and effectiveness of ammonium lactate have been demonstrated in infants and children. No unusual toxic effects were reported.
Geriatric Use
Clinical studies of ammonium lactate lotion, 12%, did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious.
ADVERSE REACTIONS
The most frequent adverse experiences in patients with xerosis are transient stinging (1 in 30 patients), burning (1 in 30 patients), erythema (1 in 50 patients) and peeling (1 in 60 patients). Other adverse reactions which occur less frequently are irritation, eczema, petechiae, dryness and hyperpigmentation.
Due to the more severe initial skin conditions associated with ichthyosis, there was a higher incidence of transient stinging, burning and erythema (each occurring in 1 in 10 patients).
OVERDOSAGE
The oral administration of ammonium lactate to rats and mice showed this drug to be practically non-toxic LD50>15 mL/kg).
DOSAGE AND ADMINISTRATION
Shake well. Apply to the affected areas and rub in thoroughly. Use twice daily or as directed by a physician.
HOW SUPPLIED
225 g plastic bottle and 400 g plastic bottle.
Store at controlled room temperature 15°– 30° C (59 – 86° F).
REFERENCES
- Blank IH: Further observation on factors which influence the water content of the stratum corneum. J Invest Dermatol 21: 259-271, 1953.
- Blank IH: Factors which influence the water content of the stratum corneum. J Invest Dermatol 18: 433-440, 1952.
- Middleton JD: Sodium lactate as a moisturizer. Cosmetics and Toiletries 93: 85-86, 1978.
- VanScott EJ and Yu RJ: Modulations of keratinization with α-hydroxy acids and related compounds in: Recent Advances In Dermatopharmacology, P. Frost, E.E. Gomez and N. Zaias (eds) Spectrum Publications, Inc. NY, 211-217, 1977
DISTRIBUTED BY PERRIGO®, ALLEGAN, MI 49010
0K5A7 RC J1 / 0K5A8 RC J1
| Ammonium Lactate (Ammonium Lactate) | |||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Revised: 03/2007