FASLODEX
-
fulvestrant injection
AstraZeneca Pharmaceuticals LP
----------
FASLODEX®(fulvestrant)
INJECTION
DESCRIPTION
FASLODEX® (fulvestrant) Injection for intramuscular administration is an estrogen receptor antagonist without known agonist effects. The chemical name is 7-alpha-[9-(4,4,5,5,5-penta fluoropentylsulphinyl) nonyl]estra-1,3,5-(10)- triene-3,17-beta-diol. The molecular formula is C32H47F5O3S and its structural formula is:
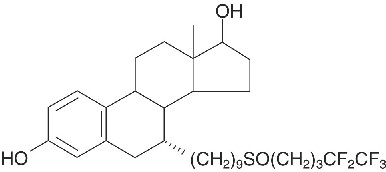
Fulvestrant is a white powder with a molecular weight of 606.77. The solution for injection is a clear, colorless to yellow, viscous liquid.
Each injection contains as inactive ingredients: Alcohol, USP, Benzyl Alcohol, NF, and Benzyl Benzoate, USP, as co-solvents, and Castor Oil, USP as a co-solvent and release rate modifier.
FASLODEX is supplied in sterile single patient pre-filled syringes containing 50-mg/mL fulvestrant either as a single 5 mL or two concurrent 2.5 mL injections to deliver the required monthly dose. FASLODEX is administered as an intramuscular injection of 250 mg once monthly.
CLINICAL PHARMACOLOGY
MECHANISM OF ACTION
Many breast cancers have estrogen receptors (ER), and the growth of these tumors can be stimulated by estrogen. Fulvestrant is an estrogen receptor antagonist that binds to the estrogen receptor in a competitive manner with affinity comparable to that of estradiol. Fulvestrant downregulates the ER protein in human breast cancer cells.
In a clinical study in postmenopausal women with primary breast cancer treated with single doses of FASLODEX 15-22 days prior to surgery, there was evidence of increasing down regulation of ER with increasing dose. This was associated with a dose-related decrease in the expression of the progesterone receptor, an estrogen-regulated protein. These effects on the ER pathway were also associated with a decrease in Ki67 labeling index, a marker of cell proliferation.
In vitro studies demonstrated that fulvestrant is a reversible inhibitor of the growth of tamoxifen-resistant, as well as estrogen-sensitive human breast cancer (MCF-7) cell lines. In in vivo tumor studies, fulvestrant delayed the establishment of tumors from xenografts of human breast cancer MCF-7 cells in nude mice. Fulvestrant inhibited the growth of established MCF-7 xenografts and of tamoxifen-resistant breast tumor xenografts. Fulvestrant resistant breast tumor xenografts may also be cross-resistant to tamoxifen.
Fulvestrant showed no agonist-type effects in in vivo uterotropic assays in immature or ovariectomized mice and rats. In in vivo studies in immature rats and ovariectomized monkeys, fulvestrant blocked the uterotrophic action of estradiol. In postmenopausal women, the absence of changes in plasma concentrations of FSH and LH in response to fulvestrant treatment (250 mg monthly) suggests no peripheral steroidal effects.
PHARMACOKINETICS
Following intravenous administration, fulvestrant is rapidly cleared at a rate approximating hepatic blood flow (about 10.5 mL plasma/min/Kg). After an intramuscular injection plasma concentrations are maximal at about 7 days and are maintained over a period of at least one month, with trough concentration about one-third of Cmax. The apparent half-life was about 40 days. After administration of 250 mg of fulvestrant intramuscularly every month, plasma levels approach steady-state after 3 to 6 doses, with an average 2.5 fold increase in plasma AUC compared to single dose AUC and trough levels about equal to the single dose Cmax (see Table 1).
|
Cmax ng/mL |
Cmin ng/mL |
AUC ng.d/mL |
t½ days |
CL mL/min |
|
|
Single dose |
8.5 ± 5.4 |
2.6 ± 1.1 |
131 ± 62 |
40 ± 11 |
690 ± 226 |
|
Multiple dose steady state |
15.8 ± 2.4 |
7.4 ± 1.7 |
328 ± 48 |
Fulvestrant was subject to extensive and rapid distribution. The apparent volume of distribution at steady state was approximately 3 to 5 L/kg. This suggests that distribution is largely extravascular. Fulvestrant was highly (99%) bound to plasma proteins; VLDL, LDL and HDL lipoprotein fractions appear to be the major binding components. The role of sex hormone-binding globulin, if any, could not be determined.
Metabolism and Excretion:
Biotransformation and disposition of fulvestrant in humans have been determined following intramuscular and intravenous administration of 14C-labeled fulvestrant. Metabolism of fulvestrant appears to involve combinations of a number of possible biotransformation pathways analogous to those of endogenous steroids, including oxidation, aromatic hydroxylation, conjugation with glucuronic acid and/or sulphate at the 2, 3 and 17 positions of the steroid nucleus, and oxidation of the side chain sulphoxide. Identified metabolites are either less active or exhibit similar activity to fulvestrant in antiestrogen models. Studies using human liver preparations and recombinant human enzymes indicate that cytochrome P-450 3A4 (CYP 3A4) is the only P-450 isoenzyme involved in the oxidation of fulvestrant; however, the relative contribution of P-450 and non-P-450 routes in vivo is unknown.
Fulvestrant was rapidly cleared by the hepatobiliary route with excretion primarily via the feces (approximately 90%). Renal elimination was negligible (less than 1%).
SPECIFIC POPULATIONS:
Geriatric:
In patients with breast cancer, there was no difference in fulvestrant pharmacokinetic profile related to age (range 33 to 89 years).
Gender:
Following administration of a single intravenous dose, there were no pharmacokinetic differences between men and women or between premenopausal and postmenopausal women. Similarly, there were no differences between men and postmenopausal women after intramuscular administration.
Race:
In the advanced breast cancer treatment trials, the potential for pharmacokinetic differences due to race have been evaluated in 294 women including 87.4% Caucasian, 7.8% Black, and 4.4% Hispanic. No differences in fulvestrant plasma pharmacokinetics were observed among these groups. In a separate trial, pharmacokinetic data from postmenopausal ethnic Japanese women were similar to those obtained in non-Japanese patients.
Renal Impairment:
Negligible amounts of fulvestrant are eliminated in urine; therefore, a study in patients with renal impairment was not conducted. In the advanced breast cancer trials, fulvestrant concentrations in women with estimated creatinine clearance as low as 30 mL/min were similar to women with normal creatinine.
Hepatic Impairment:
Fulvestrant is metabolized primarily in the liver. In clinical trials in patients with locally advanced or metastatic breast cancer, pharmacokinetic data were obtained following administration of a 250 mg dose of FASLODEX to 261 patients classified as having normal liver function and to 24 patients with mild impairment. Mild impairment was defined as an alanine aminotransferase concentration (at any visit) greater than the upper limit of the normal (ULN) reference range, but less than 2 times the ULN; or if any 2 of the following 3 parameters were between 1- and 2-times the ULN: aspartate aminotransferase, alkaline phosphatase, or total bilirubin.
There was no clear relationship between fulvestrant clearance and hepatic impairment and the safety profile in patients with mild hepatic impairment was similar to that seen in patients with no hepatic impairment. Safety and efficacy have not been evaluated in patients with moderate to severe hepatic impairment (see PRECAUTIONS-Hepatic Impairment and DOSAGE AND ADMINISTRATION-Hepatic Impairment sections).
Pediatric:
The pharmacokinetics of fulvestrant have not been evaluated in pediatric patients.
Drug-Drug Interactions
There are no known drug-drug interactions. Fulvestrant does not significantly inhibit any of the major CYP isoenzymes, including CYP 1A2, 2C9, 2C19, 2D6, and 3A4 in vitro, and studies of co-administration of fulvestrant with midazolam indicate that therapeutic doses of fulvestrant have no inhibitory effects on CYP 3A4 or alter blood levels of drug metabolized by that enzyme. Although fulvestrant is partly metabolized by CYP 3A4, a clinical study with rifampin, an inducer of CYP 3A4, showed no effect on the pharmacokinetics of fulvestrant. Also results from a healthy volunteer study with ketoconazole, a potent inhibitor of CYP 3A4, indicated that ketoconazole had no effect on the pharmacokinetics of fulvestrant and dosage adjustment is not necessary in patients co-prescribed CYP 3A4 inhibitors or inducers.
Clinical Studies
Efficacy of FASLODEX was established by comparison to the selective aromatase inhibitor anastrozole in two randomized, controlled clinical trials (one conducted in North America, the other predominately in Europe) in postmenopausal women with locally advanced or metastatic breast cancer. All patients had progressed after previous therapy with an antiestrogen or progestin for breast cancer in the adjuvant or advanced disease setting. The majority of patients in these trials had ER+ and/or PgR+ tumors. Patients who had ER-/PgR- or unknown disease must have shown prior response to endocrine therapy.
In both trials, eligible patients with measurable and/or evaluable disease were randomized to receive either FASLODEX 250 mg intramuscularly once a month (28 days + 3 days) or anastrozole 1 mg orally once a day. All patients were assessed monthly for the first three months and every three months thereafter. The North American trial was a double-blind, randomized trial in 400 postmenopausal women. The European trial was an open, randomized trial conducted in 451 patients. Patients on the FASLODEX arm of the North American trial received two separate injections (2 X 2.5 mL), whereas FASLODEX patients received a single injection (1 X 5 mL) in the European trial. In both trials, patients were initially randomized to a 125 mg per month dose as well, but interim analysis showed a very low response rate and low dose groups were dropped.
The effectiveness endpoints were response rates (RR), based on the Union Internationale Contre le Cancer (UICC) criteria, and time to progression (TTP). Survival time was also determined. Confidence intervals (95.4%) were calculated for the difference in RR between the FASLODEX and anastrozole groups. The hazard ratio for an unfavorable event, (such as disease progression or death) between FASLODEX and anastrozole groups was also determined.
Table 2 provides the demographics and baseline characteristics of the postmenopausal women randomized to FASLODEX 250 mg or anastrozole 1 mg.
|
North American Trial |
European Trial |
|||
|
Parameter |
FASLODEX 250 mg |
Anastrozole 1 mg |
FASLODEX 250 mg |
Anastrozole 1 mg |
|
No. of Participants |
206 |
194 |
222 |
229 |
|
Median Age (yrs) |
64 |
61 |
64 |
65 |
|
Age Range (yrs) |
33 - 89 |
36 − 94 |
35 - 86 |
33 - 89 |
|
Receptor Status # (%) |
||||
|
ER Positive |
170 (83%) |
156 (80%) |
156 (70%) |
173 (76%) |
|
ER/PgR Positive* |
179 (87%) |
169 (87%) |
163 (73%) |
183 (80%) |
|
ER/PgR Unknown† |
13 (6%) |
15 (8%) |
51 (23%) |
37 (16%) |
|
Previous Therapy |
||||
|
Tamoxifen |
196 (95%) |
187 (96%) |
215 (97%) |
225 (98%) |
|
Adjuvant antiestrogen only |
94 (46%) |
94 (48%) |
95 (43%) |
100 (44%) |
|
Antiestrogen for advanced disease ± adjuvant use |
110 (53%) |
97 (50%) |
126 (57%) |
129 (56%) |
|
Cytotoxic Chemotherapy |
129 (63%) |
122 (63%) |
94 (42%) |
98 (43%) |
|
Site of Metastases |
||||
|
Visceral only‡ |
39 (19%) |
45 (23%) |
30 (14%) |
41 (18%) |
|
Viscera | ||||
|
Liver involvement |
47 (23%) |
45 (23%) |
48 (22%) |
56 (24%) |
|
Lung involvement |
63 (31%) |
60 (31%) |
56 (25%) |
60 (26%) |
|
Bone only |
47 (23%) |
43 (22%) |
38 (17%) |
40 (17%) |
|
Soft Tissue only |
12 (6%) |
13 (7%) |
11 (5%) |
8 (3%) |
|
Skin and soft tissue |
43 (21%) |
41 (21%) |
40 (18%) |
35 (15%) |
Results of the trials, after a minimum follow-up duration of 14.6 months, are summarized in Table 3. The effectiveness of FASLODEX 250 mg was determined by comparing RR and TTP results to anastrozole 1 mg, the active control. With respect to response rate, the two studies ruled out (by one-sided 97.7% confidence limit) inferiority of FASLODEX to anastrozole of 6.3% and 1.4%. There was no statistically significant difference in the survival time between the two treatment groups.
|
North American Trial |
European Trial |
|||
|
Endpoint |
FASLODEX 250 mg (n=206) |
Anastrozole 1 mg (n=194) |
FASLODEX 250 mg (n=222) |
Anastrozole 1 mg (n=229) |
|
Objective tumor response | ||||
|
Number (%) of subjects | ||||
|
35 (17.0) |
33 (17.0) |
45 (20.3) |
34 (14.9) |
|
|
% Difference in Tumor | ||||
|
0.0 |
5.4 |
|||
|
2-sided 95.4% CI¶ |
(-6.3, 8.9) |
(-1.4, 14.8) |
||
|
Time to progression (TTP | ||||
|
Median TTP (days) |
165 |
103 |
166 |
156 |
|
0.9 |
1.0 |
|||
|
2-sided 95.4% CI¶ |
(0.7, 1.1) |
(0.8, 1.2) |
||
|
Stable Disease for ≥ 24 weeks (%) |
26.7 |
19.1 |
24.3 |
30.1 |
|
Survival Time | ||||
|
Died n (%) |
152 (73.8%) |
149 (76.8%) |
167 (75.2%) |
173 (75.5%) |
|
Median Survival (days) |
844 |
913 |
803 |
736 |
|
Hazard Ratio |
0.98 |
0.97 |
||
|
2-sided 95% CI¶ |
(0.78, 1.24) |
(0.78, 1.21) |
||
There are no efficacy data for the use of FASLODEX in premenopausal women with advanced breast cancer (women with functioning ovaries as evidenced by menstruation and/or premenopausal LH, FSH and estradiol levels).
INDICATIONS AND USAGE
FASLODEX is indicated for the treatment of hormone receptor positive metastatic breast cancer in postmenopausal women with disease progression following antiestrogen therapy.
CONTRAINDICATIONS
FASLODEX is contraindicated in pregnant women, and in patients with a known hypersensitivity to the drug or to any of its components.
WARNINGS
Women of childbearing potential should be advised not to become pregnant while receiving FASLODEX. FASLODEX can cause fetal harm when administered to a pregnant woman and has been shown to cross the placenta following single intramuscular doses in rats and in rabbits. In studies in the pregnant rat, intramuscular doses of fulvestrant 100 times lower than the maximum recommended human dose (based on body surface area [BSA]), caused an increased incidence of fetal abnormalities and death. Similarly, rabbits failed to maintain pregnancy and the fetuses showed an increased incidence of skeletal variations when fulvestrant was administered at one-half the recommended human dose (based on BSA).
There are no studies in pregnant women using FASLODEX. If FASLODEX is used during pregnancy or if the patient becomes pregnant while receiving this drug, the patient should be apprised of the potential hazard to the fetus, or potential risk for loss of the pregnancy. See Pregnancy section of PRECAUTIONS.
Because FASLODEX is administered intramuscularly, it should not be used in patients with bleeding diatheses, thrombocytopenia or in patients on anticoagulants.
PRECAUTIONS
General:
Before starting treatment with FASLODEX, pregnancy must be excluded (see WARNINGS).
Hepatic Impairment:
Safety and efficacy have not been evaluated in patients with moderate to severe hepatic impairment (see CLINICAL PHARMACOLOGY-Special Populations-Hepatic Impairment and DOSAGE AND ADMINISTRATION-Hepatic Impairment sections).
Drug Interactions:
There are no known drug-drug interactions. Although, fulvestrant is metabolized by CYP 3A4 in vitro, drug interactions studies with ketoconazole or rifampin did not alter fulvestrant pharmacokinetics. Dose adjustment is not needed in patients co-prescribed CYP3A4 inhibitors or inducers (see CLINICAL PHARMACOLOGY-Drug-Drug Interactions).
Carcinogenesis, Mutagenesis, Impairment of Fertility:
A two-year carcinogenesis study was conducted in female and male rats, at intramuscular doses of 15 mg/kg/30 days, 10 mg/rat/30 days and 10 mg/rat/15 days. These doses correspond to approximately 1-, 3-, and 5-fold (in females) and 1.3-, 1.3-, and 1.6-fold (in males) the systemic exposure [AUC0-30 days] achieved in women receiving the recommended dose of 250 mg/month. An increased incidence of benign ovarian granulosa cell tumors and testicular Leydig cell tumors was evident, in females dosed at 10 mg/rat/15 days and males dosed at 15 mg/rat/30 days, respectively. Induction of such tumors is consistent with the pharmacology-related endocrine feedback alterations in gonadotropin levels caused by an antiestrogen.
Fulvestrant was not mutagenic or clastogenic in multiple in vitro tests with and without the addition of a mammalian liver metabolic activation factor (bacterial mutation assay in strains of Salmonella typhimurium and Escherichia coli, in vitro cytogenetics study in human lymphocytes, mammalian cell mutation assay in mouse lymphoma cells and in vivo micronucleus test in rat).
In female rats, fulvestrant administered at doses ≥ 0.01 mg/kg/day (approximately one-hundredth of the human recommended dose based on body surface area [BSA]), for 2 weeks prior to and for 1 week following mating, caused a reduction in fertility and embryonic survival. No adverse effects on female fertility and embryonic survival were evident in female animals dosed at 0.001 mg/kg/day (approximately one-thousandth of the human dose based on BSA). Restoration of female fertility to values similar to controls was evident following a 29-day withdrawal period after dosing at 2 mg/kg/day (twice the human dose based on BSA). The effects of fulvestrant on the fertility of female rats appear to be consistent with its antiestrogenic activity. The potential effects of fulvestrant on the fertility of male animals were not studied but, in a 6-month toxicology study, male rats treated with intramuscular doses of 15 mg/kg/30 days, 10 mg/rat/30 days, or 10 mg/rat/15 days fulvestrant showed a loss of spermatozoa from the seminiferous tubules, seminiferous tubular atrophy, and degenerative changes in the epididymides. Changes in the testes and epididymides had not recovered 20 weeks after cessation of dosing. These fulvestrant doses correspond to approximately 2-, 3-, and 3-fold the systemic exposure [AUC0-30 days] achieved in women.
Pregnancy:
Pregnancy Category D:
(See WARNINGS.)
In studies in female rats at doses ≥ 0.01 mg/kg/day (IM; approximately one-hundredth of the human recommended dose based on body surface area [BSA]), fulvestrant caused a reversible reduction in female fertility, as well as effects on embryo/fetal development consistent with its antiestrogenic activity. Fulvestrant caused an increased incidence of fetal abnormalities in rats (tarsal flexure of the hind paw at 2 mg/kg/day IM; twice the human dose on BSA) and non-ossification of the odontoid and ventral tubercle of the first cervical vertebra at doses ≥ 0.1 mg/kg/day IM (approximately one-tenth of the human dose on BSA) when administered during the period of organogenesis. Rabbits failed to maintain pregnancy when dosed with 1 mg/kg/day fulvestrant IM (twice the human dose on BSA) during the period of organogenesis. Further, in rabbits dosed at 0.25 mg/kg/day (about one-half the human dose on BSA), increases in placental weight and post-implantation loss were observed but, there were no observed effects on fetal development. Fulvestrant was associated with an increased incidence of fetal variations in rabbits (backwards displacement of the pelvic girdle, and 27 pre-sacral vertebrae at 0.25 mg/kg/day IM; one-half the human dose on BSA) when administered during the period of organogenesis. Because pregnancy could not be maintained in the rabbit following doses of fulvestrant of 1 mg/kg/day and above, this study was inadequate to fully define the possible adverse effects on fetal development at clinically relevant exposures.
Nursing Mothers:
Fulvestrant is found in rat milk at levels significantly higher (approximately 12-fold) than plasma after administration of 2 mg/kg. Drug exposure in rodent pups from fulvestrant-treated lactating dams was estimated as 10% of the administered dose. It is not known if fulvestrant is excreted in human milk. Because many drugs are excreted in human milk, and because of the potential for serious adverse reactions from FASLODEX in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug taking into account the importance of the drug to the mother.
Pediatric Use:
The safety and efficacy of FASLODEX in pediatric patients have not been established.
Geriatric Use:
When tumor response was considered by age, objective responses were seen in 24% and 22% of patients under 65 years of age and in 16% and 11% of patients 65 years of age and older, who were treated with FASLODEX in the European and North American trials, respectively.
ADVERSE REACTIONS
The most commonly reported adverse experiences in the FASLODEX and anastrozole treatment groups, regardless of the investigator’s assessment of causality, were gastrointestinal symptoms (including nausea, vomiting, constipation, diarrhea and abdominal pain), headache, back pain, vasodilatation (hot flushes), and pharyngitis.
Injection site reactions with mild transient pain and inflammation were seen with FASLODEX and occurred in 7% of patients (1% of treatments) given the single 5 mL injection (predominately European Trial) and in 27% of patients (4.6% of treatments) given the 2 x 2.5 mL injections (North American Trial).
Table 4 lists adverse experiences reported with an incidence of 5% or greater, regardless of assessed causality, from the two controlled clinical trials comparing the administration of FASLODEX 250 mg intramuscularly once a month with anastrozole 1 mg orally once a day.
|
Body system and adverse event* |
FASLODEX 250 mg N=423 (%) |
Anastrozole 1 mg N=423 (%) |
|
Body as a whole |
68.3 |
67.6 |
|
Asthenia |
22.7 |
27.0 |
|
Pain |
18.9 |
20.3 |
|
Headache |
15.4 |
16.8 |
|
Back pain |
14.4 |
13.2 |
|
Abdominal pain |
11.8 |
11.6 |
|
Injection site pain† |
10.9 |
6.6 |
|
Pelvic Pain |
9.9 |
9.0 |
|
Chest pain |
7.1 |
5.0 |
|
Flu syndrome |
7.1 |
6.4 |
|
Fever |
6.4 |
6.4 |
|
Accidental injury |
4.5 |
5.7 |
|
Cardiovascular system |
30.3 |
27.9 |
|
Vasodilatation |
17.7 |
17.3 |
|
Digestive system |
51.5 |
48.0 |
|
Nausea |
26.0 |
25.3 |
|
Vomiting |
13.0 |
11.8 |
|
Constipation |
12.5 |
10.6 |
|
Diarrhea |
12.3 |
12.8 |
|
Anorexia |
9.0 |
10.9 |
|
Hemic and lymphatic Systems |
13.7 |
13.5 |
|
Anemia |
4.5 |
5.0 |
|
Metabolic and Nutritional disorders |
18.2 |
17.7 |
|
Peripheral edema |
9.0 |
10.2 |
|
Musculoskeletal system |
25.5 |
27.9 |
|
Bone pain |
15.8 |
13.7 |
|
Arthritis |
2.8 |
6.1 |
|
Nervous system |
34.3 |
33.8 |
|
Dizziness |
6.9 |
6.6 |
|
Insomnia |
6.9 |
8.5 |
|
Paresthesia |
6.4 |
7.6 |
|
Depression |
5.7 |
6.9 |
|
Anxiety |
5.0 |
3.8 |
|
Respiratory system |
38.5 |
33.6 |
|
Pharyngitis |
16.1 |
11.6 |
|
Dyspnea |
14.9 |
12.3 |
|
Cough increased |
10.4 |
10.4 |
|
Skin and appendages |
22.2 |
23.4 |
|
Rash |
7.3 |
8.0 |
|
Sweating |
5.0 |
5.2 |
|
Urogenital system |
18.2 |
14.9 |
|
Urinary tract infection |
6.1 |
3.5 |
Other adverse events reported as drug-related and seen infrequently (<1%) include thromboembolic phenomena, myalgia, vertigo, leukopenia and hypersensitivity reactions including angioedema and urticaria.
Vaginal bleeding has been reported infrequently (<1%), mainly in patients during the first 6 weeks after changing from existing hormonal therapy to treatment with FASLODEX. If bleeding persists, further evaluation should be considered.
OVERDOSAGE
Animal studies have shown no effects other than those related directly or indirectly to antiestrogen activity with intramuscular doses of fulvestrant higher than the recommended human dose. There is no clinical experience with overdosage in humans. No adverse effects were seen in healthy male and female volunteers who received intravenous fulvestrant, which resulted in peak plasma concentrations at the end of the infusion, that were approximately 10 to 15 times those seen after intramuscular injection.
DOSAGE AND ADMINISTRATION
Adults (including the elderly)
The recommended dose is 250 mg to be administered intramuscularly into the buttock at intervals of one month as either a single 5 mL injection or two concurrent 2.5 mL injections (see HOW SUPPLIED). The injection should be administered slowly.
Patients with Hepatic Impairment
FASLODEX has not been studied in patients with moderate or severe hepatic compromise. No dosage adjustment is recommended in patients with mild hepatic impairment (see CLINICAL PHARMACOLOGY-Special Populations-Hepatic Impairment and PRECAUTIONS-Hepatic Impairment sections).
Instructions for Intramuscular use, handling and disposal
1. Remove glass syringe barrel from tray and check that it is not damaged.
2. Remove perforated patient record label from syringe.
3. Peel open the safety needle (SafetyGlide™) outer packaging. For complete SafetyGlide™ instructions refer below to the "Directions for Use of SafetyGlide™".
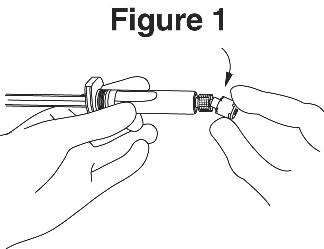
4. Break the seal of the white plastic cover on the syringe luer connector to remove the cover with the attached rubber tip cap (see Figure 1).
5. Twist to lock the needle to the luer connector.
6. Remove needle sheath.
7. Remove excess gas from the syringe (a small gas bubble may remain).
8. Administer intramuscularly slowly in the buttock.
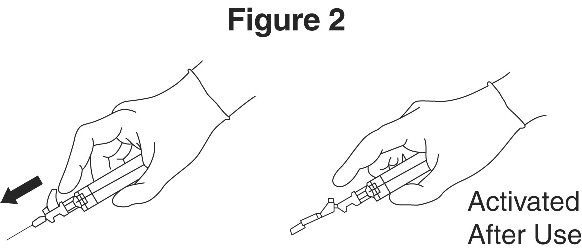
9. Immediately activate needle protection device upon withdrawal from patient by pushing lever arm completely forward until needle tip is fully covered (see Figure 2).
10. Visually confirm that the lever arm has fully advanced and the needle tip is covered. If unable to activate, discard immediately into an approved sharps collector.
11. Repeat steps 1 through 10 for second syringe.
For the 2 x 2.5 mL syringe package only, both syringes must be administered to receive the 250 mg recommended monthly dose.
SAFETYGLIDE™ INSTRUCTIONS FROM BECTON DICKINSON
SafetyGlide™ is a trademark of Becton Dickinson and Company
Reorder number 305917
CAUTION CONCERNING SAFETYGLIDE™
Federal (USA) law restricts this device to sale by or on the order of a physician. To help avoid HIV (AIDS), HBV (Hepatitis), and other infectious diseases due to accidental needlesticks, contaminated needles should not be recapped or removed, unless there is no alternative or that such action is required by a specific medical procedure.
WARNING CONCERNING SAFETYGLIDE™
Do not autoclave SafetyGlide™ Needle before use. Hands must remain behind the needle at all times during use and disposal.
DIRECTIONS FOR USE OF SAFETYGLIDE™
Peel apart packaging of the SafetyGlide™, break the seal of the white plastic cover on the syringe Luer connector and attach the SafetyGlide™ needle to the Luer Lock of the syringe by twisting.
Transport filled syringe to point of administration.
Pull shield straight off needle to avoid damaging needle point.
Administer injection following package instruction.
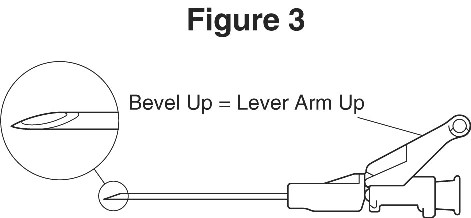
For user convenience, the needle ‘bevel up’ position is orientated to the lever arm, as shown in Figure 3.
Immediately activate needle protection device upon withdrawal from patient by pushing lever arm completely forward until needle tip is fully covered (Figure 2).
Visually confirm that the lever arm has fully advanced and the needle tip is covered. If unable to activate, discard immediately into an approved sharps collector.
Activation of the protective mechanism may cause minimal splatter of fluid that may remain on the needle after injection.
For greatest safety, use a one-handed technique and activate away from self and others.
After single use, discard in an approved sharps collector in accordance with applicable regulations and institutional policy.
Becton Dickinson guarantees the contents of their unopened or undamaged packages to be sterile, non-toxic and non-pyrogenic.
HOW SUPPLIED
FASLODEX is supplied in two different packaging configurations:
1. FASLODEX is supplied as one clear neutral glass (Type 1) barrel containing 250 mg/5mL (50 mg/mL) FASLODEX injection for intramuscular injection and fitted with a tamper evident closure.
NDC 0310-0720-50
2. FASLODEX is also supplied as two clear neutral glass (Type 1) barrels each containing 125 mg/2.5 mL (50 mg/mL) FASLODEX Injection for intramuscular injection and fitted with a tamper-evident closure. PLEASE NOTE: THE SYRINGES ARE SUPPLIED HALF FULL. BOTH SYRINGES MUST BE ADMINISTERED TO RECEIVE THE 250 MG RECOMMENDED MONTHLY DOSE.
NDC 0310-0720-25
The syringes are presented in a tray with polystyrene plunger rod and a safety needles (SafetyGlide™) for connection to the barrel.
Storage:
REFRIGERATE, 2°-8°C (36°-46°F). TO PROTECT FROM LIGHT, STORE IN THE ORIGINAL CARTON UNTIL TIME OF USE.
SafetyGlide™ is a trademark of Becton Dickinson and Company
All other trademarks are the property of the AstraZeneca group of companies
©AstraZeneca 2002, 2004
Distributed by:
AstraZeneca Pharmaceuticals LP
Wilmington, DE 19850
Manufactured for:
AstraZeneca UK Limited
Macclesfield, Cheshire, England
By: Vetter Pharma-Fertigung GMBH & Co. KG
Ravensburg, Germany
Made in Germany
Rev 08–04 SIC 31003–00
PACKAGE LABEL PRINCIPAL DISPLAY PANEL




| FASLODEX
fulvestrant injection |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA021344 | 08/12/2010 | |
| Labeler - AstraZeneca Pharmaceuticals LP (054743190) |
| Registrant - AstraZeneca PLC (230790719) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Vetter Pharma Fertigung GMBH & Co. KG | 316126754 | MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| AstraZeneca UK Limited | 232784079 | API MANUFACTURE | |