LIDOCAINE HCI HYDROCORTISONE ACETATE
-
lidocaine hydrochloride and
hydrocortisone acetate cream
Kylemore Pharmaceuticals, LLC
----------
Lidocaine HCI 3% - Hydrocortisone Acetate 0.5% CreamDESCRIPTION:
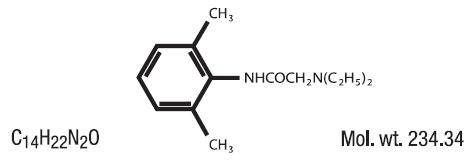
Lidocaine is chemically designated as acetamide, 2-(diethylamino)- N-(2,6-dimethylphenyl), and has the following structure:
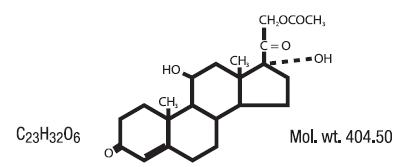
Hydrocortisone acetate has a chemical name pregn-4-ene-3, 20-dione, 21-(acetyloxy)-11,17-dihydroxy-(11ß)-. It has the following structural formula:
INGREDIENTS: Lidocaine HCI 3% - Hydrocortisone Acetate 0.5% Cream
Each 7g tube of Lidocaine HCl 3% - Hydrocortisone Acetate 0.5% contains 30 mg of Lidocaine HCl and 5 mg of Hydrocortisone Acetate per gram. Inactive ingredients include: Purified Water, White Petrolatum USP, Stearic Acid NF, Cetyl Alcohol NF, Propylene Glycol USP, Stearyl Alcohol NF, Mineral Oil USP, Polysorbate 60, Sorbitan Monostearate, Methylparaben NF, Propylparaben NF, Sodium Hydroxide NF, Aluminum Sulfate and Calcium Acetate
CLINICAL PHARMACOLOGY:
MECHANISM OF ACTION: Product releases lidocaine to stabilize the neuronal membrane by inhibiting the ionic fluxes required for initiation and conduction of impulses, thereby effecting local anesthetic action. Hydrocortisone acetate provides relief of inflammatory and pruritic manifestations of corticosteroid responsive dermatoses.PHARMACOKINETICS:
Lidocaine may be absorbed following topical administration to mucous membranes, its rate and extent of absorption depending upon the specific site of application, duration of exposure, concentration, and total dosage. In general, the rate of absorption of local anesthetic agents following topical application occurs most rapidly after intratracheal administration. Lidocaine is also well-absorbed from the gastrointestinal tract, but little intact drug appears in the circulation because of biotransformation in the liver.Lidocaine is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 g of free base per mL, 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Studies of lidocaine metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 g free base per mL. In the rhesus monkey arterial blood levels of 18-21 g/mL have been shown to be threshold for convulsive activity.
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses.
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma protein in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
INDICATIONS:
Product is used for the anti-inflammatory and anesthetic relief of itching, pain, soreness and discomfort due to hemorrhoids, anal fissures, pruritus and similar conditions of the anal area.
CONTRAINDICATIONS:
These products should not be used in patients with a history of sensitivity to any of its ingredients or adverse reactions to lidocaine or amide anesthetics, which usually do not cross-react with “caine” ester type anesthetics. If excessive irritation and significant worsening occur, discontinue use and seek the advice of your physician. These products and topical lidocaine should be used cautiously in those with impaired liver function, as well as the very ill or very elderly and those with significant liver disease. These products should be used with caution in patients receiving antiarrhythmic drugs of Class I since the adverse effects are additive and generally synergistic. These products are contraindicated for tuberculous or fungal lesions or skin vaccinia, varicella and acute herpes simplex. Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.WARNINGS:
For external use only. Not for ophthalmic use.These products and used applicators could harm small children if chewed or swallowed. Individual tubes are NOT child resistant. Keep the product inside the child resistant blister until ready to use.Keep products, cleansing wipes and applicators out of the reach of children.
Topical formulations of lidocaine may be absorbed to a greater extent through mucous membranes and abraded, fissured or irritated skin than through intact skin. These products should not be ingested or applied into the mouth, inside of the nose or in the eyes. These products should not be used in the ears. Any situation where lidocaine penetrates beyond the tympanic membrane into the middle ear is contraindicated because of ototoxicity associated with lidocaine observed in animals when instilled in the middle ear. These products should not come into contact with the eye or be applied into the eye because of the risk of severe eye irritation and the loss of eye surface sensation which reduces protective reflexes and can lead to corneal irritation and possibly abrasion. If eye contact occurs, rinse out the eye immediately with saline or water and protect the eye surface until sensation is restored.
PRECAUTIONS:
If irritation or sensitivity occurs or infection appears, discontinue use and institute appropriate therapy. If extensive areas are treated, the possibility of systemic absorption exists. Systemic absorption of topical steroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glycosuria in some patients. Conditions which augment systemic absorption include the application of the more potent steroids, use over large surface areas, prolonged use, and the addition of occlusive dressings. Therefore, patients receiving a large dose of potent topical steroids applied to a large surface area, or under an occlusive dressing, should be evaluated periodically for evidence of HPA axis suppression. If noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Recovery of the HPA axis function is generally prompt and complete upon discontinuation of the drug. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids. Children may absorb proportionately larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity. If irritation develops, topical steroids should be discontinued and appropriate therapy instituted. In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, the corticosteroid should be discontinued until the infection has been adequately controlled.CARCINOGENESIS, MUTAGENESIS, AND IMPAIRMENT OF FERTILITY:
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of topical corticosteroids.Studies to determine mutagenicity with prednisolone and hydrocortisone have revealed negative results.
Studies of lidocaine in animals to evaluate the carcinogenic and mutagenic potential of the effect on fertility have not been conducted.
USE IN PREGNANCY:
Teratogenic Effects: Pregnancy Category C: Reproduction studies have been performed for lidocaine in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by lidocaine. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering lidocaine to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place. Corticosteroids are generally terato-genic in laboratory animals when administered systemically at relatively low dosage levels. The more potent corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.NURSING MOTHERS:
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when this drug is administered to a nursing women.PEDIATRIC USE:
Safety and efficacy in children have not been established.ADVERSE REACTIONS:
During or immediately following application of product, there may be transient stinging or burning from open areas of skin, or transient blanching (lightening), or erythema (redness) of the skin.Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
DOSAGE AND ADMINISTRATION:
Apply product to the affected area(s) 2-3 times per day, or as directed by a physician. The cap should be removed from the tube and the applicator tip firmly screwed onto the end of the tube and tightened. While holding the tube, squeeze the tube to fill the applicator until a small amount of cream/gel shows and lubricate the end of the tip with cream/gel. Gently insert the applicator tip with attached tube into anal area. Continue squeezing the body of the tube as it is moved around the areas of discomfort, and lastly, around and in the anal opening (if directed by physician).Do not completely insert the applicator and tube into the anus or insert deep into the rectum. Do not insert a loose applicator tip into the anus or rectum. Once application is completed, the tube and applicator tip should be gently removed from the area and disposed. Note that an adequate amount of product for an application to the anal and peri-anal area will be applied through the applicator tip by gently squeezing the tube during application. These products should not be used in excess of recommendations or for prolonged use in the anal canal. If the condition does not respond to repeated courses of product or should worsen, discontinue use and seek the advice of your physician.
HOW SUPPLIED:
Lidocaine HCI 3% - Hydrocortisone Acetate 0.5% Cream contains 14 1/4 oz. (7 g) Tubes and Applicators. NDC 49769-111-01.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Store at controlled room temperature 20º-25ºC (68º-77ºF). Protect from freezing.
Manufactured for:
Kylemore Pharmaceuticals
Suwanee, GA 30024
Rev. 12/09 111-10
PACKAGING:
Sample carton:

Sample label:

| LIDOCAINE HCI HYDROCORTISONE ACETATE
lidocaine hydrochloride, hydrocortisone acetate cream |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| unapproved drug other | 01/12/2010 | ||
| Labeler - Kylemore Pharmaceuticals, LLC (831892471) |