medroxyprogesterone acetate (Medroxyprogesterone Acetate) tablet
[BARR LABORATORIES, INC.]
AUGUST 2001
21008720103
(Three Patient Information Leaflets Enclosed-Tear at Perforation)
Rx only
DESCRIPTION:
Medroxyprogesterone Acetate Tablets contain medroxyprogesterone acetate, which is a derivative of progesterone. It is a white to off-white, odorless, crystalline powder, stable in air, melting between 200 and 210°C. It is freely soluble in chloroform, soluble in acetone and in dioxane, sparingly soluble in alcohol and in methanol, slightly soluble in ether, and insoluble in water.
The chemical name for medroxyprogesterone acetate is Pregn-4-ene-3,20-dione, 17-(acetyloxy)-6-methyl-, (6α)-. The structural formula is:
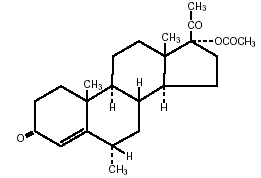
C24H34O4 Molecular Weight: 386.53O mm
Each tablet, for oral administration, contains 2.5 mg, 5 mg or 10 mg of medroxyprogesterone acetate. In addition, each tablet contains the following inactive ingredients: crospovidone, lactose monohydrate, magnesium stearate, methylcellulose, pregelatinized starch, and sodium lauryl sulfate.
CLINICAL PHARMACOLOGY:
Medroxyprogesterone acetate (MPA), administered orally or parenterally in the recommended doses to women with adequate endogenous estrogen, transforms proliferative into secretory endometrium. Androgenic and anabolic effects have been noted, but the drug is apparently devoid of significant estrogenic activity. While parenterally administered MPA inhibits gonadotropin production, which in turn prevents follicular maturation and ovulation, available data indicate that this does not occur when the usually recommended oral dosage is given as single daily doses.
Pharmacokinetics:
The pharmacokinetics of MPA were determined in 20 postmenopausal women following a single-dose administration of eight Medroxyprogesterone Acetate Tablets 2.5 mg or a single administration of two medroxyprogesterone acetate tablets 10 mg under fasting conditions. In another study, the steady-state pharmacokinetics of MPA were determined under fasting conditions in 30 postmenopausal women following daily administration of one medroxyprogesterone acetate tablet 10 mg for 7 days. In both studies, MPA was quantitated in serum using a validated gas chromatography-mass spectrometry (GC-MS) method. Estimates of the pharmacokinetic parameters of MPA after single and multiple doses of medroxyprogesterone acetate tablets were highly variable and are summarized in Table 1.
|
|||||||
|
Tablet Strength |
Cmax (ng/mL) |
Tmax (h) |
Auc0-∞ (ng*h/mL) |
t½ (h) |
Vd/f (L) |
CL/f (mL/min) |
|
| Single Dose | |||||||
| 2 x 10 mg | 1.01 (0.599) | 2.65 (1.41) | 6.95 (3.39) | 12.1 (3.49) |
78024 (47220) |
64110 (42662) |
|
| 8 x 2.5 mg | 0.805 (0.413) | 2.22 (1.39) | 5.62 (2.79) | 11.6 (2.81) |
62748 (40146) |
74123 (35126) |
|
| Multiple Dose | |||||||
| 10 mg* | 0.71 (0.35) | 2.83 (1.83) | 6.01 (3.16) | 16.6 (15.0) |
40564 (38256) |
41963 (38402) |
|
Absorption:
No specific investigation on the absolute bioavailability of MPA in humans has been conducted. MPA is rapidly absorbed from the gastrointestinal tract, and maximum MPA concentrations are obtained between 2 to 4 hours after oral administration.
Effect of Food:
Administration of medroxyprogesterone acetate with food increases the bioavailability of MPA. A 10-mg dose of medroxyprogesterone acetate, taken immediately before or after a meal, increased MPA Cmax(50 to 70%) and AUC (18 to 33%).
The half-life of MPA was not changed with food.
Distribution:
MPA is approximately 90% protein bound, primarily to albumin; no MPA binding occurs with sex-hormone binding globulin. The unbound MPA modulates pharmacologic responses.
Metabolism:
Following oral dosing, MPA is extensively metabolized in the liver via ring A and/or side-chain hydroxylation, with subsequent conjugation and elimination in the urine. At least 16 MPA metabolites have been identified.
Excretion:
Most MPA metabolites are excreted in the urine as glucuronide conjugates with only minor amounts excreted as sulfates. Mean percent dose excreted in the 24-hour urine of patients with fatty liver as intact MPA after a 10-mg or 100-mg dose was 7.3% and 6.4%, respectively.
Special Populations:
Renal Insufficiency :
The pharmacokinetics of MPA in patients with varying degrees of renal insufficiency have not been investigated. The renal clearance of MPA is negligible and a decrease in total body clearance is not expected in patients with renal insufficiency.
Hepatic Insufficiency:
MPA is almost exclusively eliminated via hepatic metabolism. In 14 patients with advanced liver disease, MPA disposition was significantly altered (reduced elimination). As such, medroxyprogesterone acetate is contraindicated in patients with severe hepatic disease (see CONTRAINDICATIONS). However, for patients with mild-moderate degree of hepatic impairment, a lower dose of medroxyprogesterone acetate or a less frequent administration should be considered.
Drug-Drug Interactions:
No formal pharmacokinetic drug-drug interaction studies have been conducted with medroxyprogesterone acetate. However, published literature indicates that coadministration of conjugated estrogens with MPA does not affect the pharmacokinetic profile of MPA; similarly, MPA does not affect the pharmacokinetic profile of the conjugated or unconjugated estrogens. Literature data also indicate that concomitant administration with aminoglutethimide would significantly reduce serum concentrations of MPA, likely by increasing the clearance of the drug.
CLINICAL STUDIES:
The use of unopposed estrogen therapy has been associated with an increased risk of endometrial hyperplasia, a possible precursor of endometrial carcinoma.1 The incidence of estrogen-associated endometrial hyperplasia and endometrial cancer was assessed in two large, long-term, randomized clinical trials. The histological results of the clinical studies indicate that the addition of medroxyprogesterone acetate to an estrogen replacement regimen for 12 to 14 days per cycle reduces the incidence of endometrial hyperplasia in women with intact uteri. The addition of a progestin to 0.625 mg conjugated estrogen has not been shown to interfere with the efficacy of 0.625 mg conjugated estrogen for its approved indications.1-3
A 3-year, double-blind, placebo-controlled study of nonhysterectomized, postmenopausal women between the ages of 45 and 64 years were randomized to receive placebo, conjugated estrogen only, or conjugated estrogen plus cyclic medroxyprogesterone acetate. The treatment group receiving 10 mg medroxyprogesterone acetate plus 0.625 mg conjugated estrogens showed a significantly lower rate of hyperplasia in comparison to the group given 0.625 mg conjugated estrogens only. The 3-year histological results are summarized in Table 2.
| * Includes most extreme abnormal result | |||
|
Histological Results |
Placebo (n=119) |
CEE* (n=119) |
Medroxyprogesterone |
|
Normal/No hyperplasia (%) |
116 (97) | 45 (38) | 112 (95) |
|
Simple (cystic) hyperplasia (%) |
1 (1) | 33 (28) | 4 (3) |
|
Complex (adenomatous) hyperplasia (%) |
1 (1) | 27 (22) | 2 (2) |
| Atypia(%) | 0 | 14 (12) | 0 |
|
Adenocarcinoma (%) |
1 (1) | 0 | 0 |
In a second study, postmenopausal women between the ages of 45 and 65 years were enrolled in a 1-year, double-blind study. All patients received conjugated estrogen 0.625 mg every day of a 28-day cycle, and were randomized to receive cyclic MPA 5 mg, cyclic MPA 10 mg, or conjugated estrogen only. The treatment groups receiving MPA 5 or 10 mg plus conjugated estrogens showed a significantly lower rate of hyperplasia in comparison to the group given conjugated estrogens only. The incidence of endometrial hyperplasia is shown in Table 3.
| CEE* | MPA† + CEE* | ||
| (n=283) |
MPA 5 mg (n=277) |
MPA 10 mg (n=272) |
|
|
Cystic hyperplasia (%) |
55 (19) | 3 (1) | 0 |
|
Adenomatous hyperplasia without atypia |
2 (1) | 0 | 0 |
INDICATIONS AND USAGE:
Medroxyprogesterone Acetate Tablets are indicated for secondary amenorrhea and for abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology, such as fibroids or uterine cancer. Medroxyprogesterone Acetate Tablets are also indicated to reduce the incidence of endometrial hyperplasia in nonhysterectomized postmenopausal women receiving 0.625 mg conjugated estrogen.
CONTRAINDICATIONS:
- Thrombophlebitis, thromboembolic disorders, cerebral apoplexy or patients with a past history of these conditions.
- Liver dysfunction or disease.
- Known or suspected malignancy of breast or genital organs.
- Undiagnosed vaginal bleeding.
- Missed abortion.
- As a diagnostic test for pregnancy.
- Known sensitivity to medroxyprogesterone acetate tablets.
- Known or suspected pregnancy.
WARNINGS:
- The physician should be alert to the earliest manifestations of thrombotic disorders (thrombophlebitis, cerebrovascular disorders, pulmonary embolism, and retinal thrombosis). Should any of these occur or be suspected, the drug should be discontinued immediately.
- Beagle dogs treated with medroxyprogesterone acetate developed mammary nodules some of which were malignant. Although nodules occasionally appeared in control animals, they were intermittent in nature, whereas the nodules in the drug-treated animals were larger, more numerous, persistent, and there were some breast malignancies with metastases. Their significance with respect to humans has not been established.
- Discontinue medication pending examination if there is sudden partial or complete loss of vision, or if there is a sudden onset of proptosis, diplopia or migraine. If examination reveals papilledema or retinal vascular lesions, medication should be withdrawn.
- Detectable amounts of progestin have been identified in the milk of mothers receiving the drug. The effect of this on the nursing neonate and infant has not been determined.
- Usage in pregnancy is contraindicated.
- Retrospective studies of morbidity and mortality in Great Britain and studies of morbidity in the United States have shown a statistically significant association between thrombophlebitis, pulmonary embolism, and cerebral thrombosis and embolism and the use of oral contraceptives.4‑7 The estimate of the relative risk of thromboembolism in the study by Vessey and Doll6 was about sevenfold, while Sartwell and associates7 in the United States found a relative risk of 4.4, meaning that the users are several times as likely to undergo thromboembolic disease without evident cause as nonusers. The American study also indicated that the risk did not persist after discontinuation of administration, and that it was not enhanced by long continued administration. The American study was not designed to evaluate a difference between products.
PRECAUTIONS:
General:
- The pretreatment physical examination should include special reference to breast and pelvic organs, as well as Papanicolaou smear.
- Because progestogens may cause some degree of fluid retention, conditions which might be influenced by this factor, such as epilepsy, migraine, asthma, cardiac or renal dysfunction, require careful observation.
- In cases of breakthrough bleeding, as in all cases of irregular bleeding per vaginum, nonfunctional causes should be borne in mind. In cases of undiagnosed vaginal bleeding, adequate diagnostic measures are indicated.
- Patients who have a history of psychic depression should be carefully observed and the drug discontinued if the depression recurs to a serious degree.
- Any possible influence of prolonged progestin therapy on pituitary, ovarian, adrenal, hepatic or uterine functions awaits further study.
- Diabetic patients should be carefully observed while receiving progestin therapy.
- The age of the patient constitutes no absolute limiting factor although treatment with progestins may mask the onset of the climacteric.
- The pathologist should be advised of progestin therapy when relevant specimens are submitted.
- Because of the occurrence of thrombotic disorders, (thrombophlebitis, pulmonary embolism, retinal thrombosis, and cerebrovascular disorders) in patients taking estrogen-progestin combinations and since the mechanism is obscure, the physician should be alert to the earliest manifestation of these disorders.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Long-term intramuscular administration of medroxyprogesterone acetate has been shown to produce mammary tumors in beagle dogs. There was no evidence of a carcinogenic effect associated with the oral administration of medroxyprogesterone to rats and mice. Medroxyprogesterone acetate was not mutagenic in a battery of in vitro or in vivo genetic toxicity assays.
Medroxyprogesterone acetate at high doses is an antifertility drug and high doses would be expected to impair fertility until the cessation of treatment.
Information for the Patient:
See Patient Information at the end of the brochure.
Pregnancy:
Pregnancy Category X:
Medroxyprogesterone acetate tablets are contraindicated during pregnancy. Several reports suggest an association between intrauterine exposure to progestational drugs in the first trimester of pregnancy and genital abnormalities in male and female fetuses. The risk of hypospadias in male fetuses may be doubled with exposure to these drugs. Some progestational drugs induce mild virilization of the external genitalia of female fetuses.
Nursing Mothers:
The administration of any drug to nursing mothers should be done only when clearly necessary since many drugs are excreted in human milk. Detectable amounts of progestin have been identified in the milk of nursing mothers receiving progestins. The effect of this on the nursing infant has not been determined.
Pediatric Use:
The safety and effectiveness of medroxyprogesterone acetate tablets in pediatric patients has not been established.
ADVERSE REACTIONS:
Breast:
Breast tenderness or galactorrhea has been reported.
Skin:
Sensitivity reactions consisting of urticaria, pruritus, edema and generalized rash have occurred. Acne, alopecia and hirsutism have been reported.
Thromboembolic Phenomena:
Thromboembolic phenomena including thrombophlebitis and pulmonary embolism have been reported.
Other:
The following adverse reactions have been observed in women taking progestins, including medroxyprogesterone acetate tablets:
breakthrough bleeding
cholestatic jaundice
spotting
anaphylactoid reactions and anaphylaxis rash (allergic) with and without pruritus
change in menstrual flow
amenorrhea
edema
change in weight (increase or decrease)
mental depression
pyrexia
changes in cervical erosion and cervical secretions
insomnia
nausea
somnolence
Although available evidence is suggestive of an association, such a relationship has been neither confirmed nor refuted for the following serious adverse reactions:
neuro-ocular lesions, e.g., retinal thrombosis and optic neuritis.
The following adverse reactions have been observed in patients receiving estrogen-progestin combination drugs:
rise in blood pressure in susceptible individuals
fatigue
backache
hirsutism
premenstrual-like syndrome
loss of scalp hair
erythema multiforme
changes in libido
erythema nodosum
hemorrhagic eruption
changes in appetite
cystitis-like syndrome
headache
itching
nervousness
dizziness
Laboratory Tests:
The following laboratory results may be altered by the use of estrogen-progestin combination drugs:
Increased sulfobromophthalein retention and other hepatic function tests.
Coagulation tests: increase in prothrombin factors VII, VIII, IX and X.
Metyrapone test.
Pregnanediol determination.
Thyroid function: increase in PBI, and butanol extractable protein bound iodine and decrease in T3 uptake values.
DOSAGE AND ADMINISTRATION:
Secondary Amenorrhea:
Medroxyprogesterone acetate tablets may be given in dosages of 5 or 10 mg daily for 5 to 10 days. A dose for inducing an optimum secretory transformation of an endometrium that has been adequately primed with either endogenous or exogenous estrogen is 10 mg of medroxyprogesterone acetate daily for 10 days. In cases of secondary amenorrhea, therapy may be started at any time. Progestin withdrawal bleeding usually occurs within three to seven days after discontinuing medroxyprogesterone therapy.
Abnormal Uterine Bleeding Due to Hormonal Imbalance in the Absence of Organic Pathology:
Beginning on the calculated 16th or 21st day of the menstrual cycle, 5 or 10 mg of medroxyprogesterone acetate may be given daily for 5 to 10 days. To produce an optimum secretory transformation of an endometrium that has been adequately primed with either endogenous or exogenous estrogen, 10 mg of medroxyprogesterone acetate daily for 10 days beginning on the 16th day of the cycle is suggested. Progestin withdrawal bleeding usually occurs within three to seven days after discontinuing therapy with medroxyprogesterone. Patients with a past history of recurrent episodes of abnormal uterine bleeding may benefit from planned menstrual cycling with medroxyprogesterone.
Reduction of endometrial hyperplasia in postmenopausal women receiving 0.625 mg conjugated estrogens:
Medroxyprogesterone acetate tablets may be given in dosages of 5 or 10 mg daily for 12 to 14 consecutive days per month, either beginning on the 1st day of the cycle or the 16th day of the cycle.
HOW SUPPLIED:
Medroxyprogesterone Acetate Tablets, USP are available as:
| 2.5 mg: | White, round, scored, biconvex tablet. Debossed with 555/872 on the scored side and stylized b on the other side. | |
| Available in bottles of: | ||
| 30 | NDC 0555-0872-01 | |
| 100 | NDC 0555-0872-02 | |
| 500 | NDC 0555-0872-04 | |
| 1000 | NDC 0555-0872-05 | |
| 5 mg: | White, round, scored, biconvex tablet. Debossed with 555/873 on the scored side and stylized b on the other side. | |
| Available in bottles of: | ||
| 30 | NDC 0555-0873-01 | |
| 100 | NDC 0555-0873-02 | |
| 500 | NDC 0555-0873-04 | |
| 1000 | NDC 0555-0873-05 | |
| 10 mg: | White, round, scored, biconvex tablet. Debossed with 555/779 on the scored side and stylized b on the other side. | |
| Available in bottles of: | ||
| 30 | NDC 0555-0779-01 | |
| 100 | NDC 0555-0779-02 | |
| 500 | NDC 0555-0779-04 | |
| 1000 | NDC 0555-0779-05 | |
Dispense with a child-resistant closure in a tight container as defined in the USP.
Store at controlled room temperature 15°-30°C (59°-86°F) [see USP].
REFERENCES:
- Writing Group for the PEPI Trial: Effects of hormone replacement therapy on endometrial histology in postmenopausal women. JAMA 275:370-375, 1996.
- Woodruff JD, Pickar JH: Incidence of endometrial hyperplasia in postmenopausal women taking conjugated estrogens (Premarin) with medroxyprogesterone acetate or conjugated estrogens alone (The Menopause Study Group). Am J Obstet Gynecol 170:1213-1223, 1994.
- Speroff L, Rowan J, Symons J, et.al.: The comparative effect on bone density, endometrium, and lipids of continuous hormones as replacement therapy (CHART Study) JAMA 276:1397-1403, 1996.
- Royal College of General Practitioners: Oral contraception and thromboembolic disease. J Coll Gen Pract 13:267-279, 1967.
- Inman WHW, Vessey MP: Investigation of deaths from pulmonary, coronary, and cerebral thrombosis and embolism in women of child-bearing age. Br Med J 2:193-199, 1968.
- Vessey MP, Doll R: Investigation of relation between use of oral contraceptives and thromboembolic disease. A further report. Br Med J 2:651-657, 1969.
- Sartwell PE, Masi AT, Arthes FG, et.al.: Thromboembolism and oral contraceptives: An epidemiological case-control study. Am J Epidemiol 90:365-380, 1969.
The text of the patient insert for progesterone and progesterone-like drugs is set forth below:
PATIENT INFORMATION:
Medroxyprogesterone acetate tablets contain medroxyprogesterone acetate, a progesterone. The information below is that which the U.S. Food and Drug Administration requires be provided for all patients taking progesterones. The information below relates only to the risk to the unborn child associated with use of progesterone during pregnancy. For further information on the use, side effects and other risks associated with this product, ask your doctor.
WARNING FOR WOMEN:
Progesterone or progesterone-like drugs have been used to prevent miscarriage in the first few months of pregnancy. No adequate evidence is available to show that they are effective for this purpose. Furthermore, most cases of early miscarriage are due to causes which could not be helped by these drugs.
There is an increased risk of minor birth defects in children whose mothers take this drug during the first 4 months of pregnancy. Several reports suggest an association between mothers who take these drugs in the first trimester of pregnancy and genital abnormalities in male and female babies. The risk to the male baby is the possibility of being born with a condition in which the opening of the penis is on the underside rather than the tip of the penis (hypospadias). Hypospadias occurs in about 5 to 8 per 1,000 male births and is about doubled with exposure to these drugs. There is not enough information to quantify the risk to exposed female fetuses, but enlargement of the clitoris and fusion of the labia may occur, although rarely.
Therefore, since drugs of this type may induce mild masculinization of the external genitalia of the female fetus, as well as hypospadias in the male fetus, it is wise to avoid using the drug during the first trimester of pregnancy.
These drugs have been used as a test for pregnancy but such use is no longer considered safe because of possible damage to a developing baby. Also, more rapid methods for testing for pregnancy are now available.
If you take medroxyprogesterone acetate and later find you were pregnant when you took it, be sure to discuss this with your doctor as soon as possible.
MANUFACTURED BY
BARR LABORATORIES, INC.
POMONA, NY 10970
AUGUST 2001
BR-872, 873, 779
| Medroxyprogesterone Acetate (Medroxyprogesterone Acetate) | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Medroxyprogesterone Acetate (Medroxyprogesterone Acetate) | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Medroxyprogesterone Acetate (Medroxyprogesterone Acetate) | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
Revised: 10/2006BARR LABORATORIES, INC.