BENZOYL PEROXIDE
-
benzoyl peroxide liquid
River's Edge Pharmaceuticals, LLC
----------
Benzoyl Peroxide Wash 7%INDICATIONS AND USAGE
Benzoyl Peroxide Wash 7% is indicated for use in the topical treatment of mild to moderate acne vulgaris.DOSAGE AND ADMINISTRATION
Benzoyl Peroxide Wash 7% should be used once or twice daily on the affected areas. Frequency of use should be adjusted to obtain the desired clinical response. Clinically visible improvement will normally occur by the third week of therapy. Maximum lesion reduction may be expected after approximately eight to twelve weeks of drug use. Continuing use of the drug is normally required to maintain a satisfactory clinical response. Shake well before using. Wet skin areas to be treated; apply Benzoyl Peroxide Wash 7% by massaging gently into skin for 10-20 seconds, working into a full lather. Rinse thoroughly and pat dry.DOSAGE FORM AND STRENGTH
Benzoyl peroxide 7% topical preparationCONTRAINDICATIONS
Benzoyl Peroxide Wash 7% should not be used in patients who have shown hypersensitivity to benzoyl peroxide or to any of the other ingredients in the products.WARNINGS AND PRECAUTIONS
KEEP OUT OF REACH OF CHILDREN. When using this product, avoid unnecessary sun exposure and use a sunscreen.General: For external use only. Avoid contact with eyes and mucous membranes. If severe irritation develops, discontinue use and institute appropriate therapy.
Information for the Patient: Avoid contact with eyes, eyelids, lips and mucous membranes. If accidental contact occurs, rinse with water. AVOID CONTACT WITH HAIR, FABRICS OR CARPETING AS BENZOYL PEROXIDE WILL CAUSE BLEACHING OR DISCOLORATION. If excessive irritation develops, discontinue use and consult your physician.
ADVERSE REACTIONS
Allergic contact dermatitis and dryness have been reported with topical benzoyl peroxide therapy.To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
The use of tretinoin and hydroquinone topical preparations should be avoided when using this product.
USE IN SPECIFIC POPULATIONS
Pregnancy
Category C - Animal reproduction studies have not been conducted with benzoyl peroxide. It is also not known whether benzoyl peroxide can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Benzoyl peroxide should be used by a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when benzoyl peroxide is administered to a nursing woman.
Pediatric Use
Safety and effectiveness in children below the age of 12 have not been established.
OVERDOSAGE
If excessive scaling, erythema or edema occurs, the use of these preparations should be discontinued. To hasten resolution of the adverse effects, cool compresses may be used. After symptoms and signs subside, a reduced dosage schedule may be cautiously tried if the reaction is judged to be due to excessive use and not allergenicity.
DESCRIPTION
Benzoyl Peroxide Wash 7% is a topical preparation containing benzoyl peroxide as the active ingredient incorporated into a series of microscopic concentric vesicules of oil and water. This system results in the release of the active ingredient into the skin over a sustained period of time. Ingredients include: Citric Acid, cocamidopropyl betaine, cocamine oxide, disodium laureth sulfosuccinate, disodium EDTA, Fragrance Lemon, Glycerin, Hydrogenated Castor Oil, Hypromellose, Magnesium Aluminum Silicate, Cetearyl Alcohol and PEG-3 Distearoylamidoethylmonium Methosulfate and Polysorbate 60, Methylparaben, PEG-150 Pentaerythrityl Tetrastearate and Aqua and PEG-6 Caprylic/Capric Glycerides, PEG-40 Hydrogenated Castor Oil, Poloxamer 182, Purified Water and Xanthan Gum.
Benzoyl peroxide is an oxidizing agent that possesses antibacterial properties and is classified as a keratolytic.
Benzoyl peroxide (C14H10O4) is represented by the following structure:
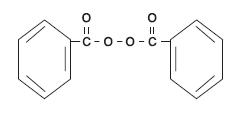
CLINICAL PHARMACOLOGY
The exact method of action of benzoyl peroxide in acne vulgaris is not known. Benzoyl peroxide is an antibacterial agent with demonstrated activity against Propionibacterium acnes. This action, combined with the mild keratolytic effect of benzoyl peroxide is believed to be responsible for its usefulness in acne. Benzoyl peroxide is absorbed by the skin where it is metabolized to benzoic acid and excreted as benzoate in the urine.NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of FertilityBased upon all available evidence, benzoyl peroxide is not considered to be a carcinogen. However, data from a study using mice known to be highly susceptible to cancer suggest that benzoyl peroxide acts as a tumor promoter. The clinical significance of the findings is not known.
HOW SUPPLIED/STORAGE AND HANDLING
Benzoyl Peroxide Wash 7% is supplied as follows:
SIZE: 6.35 oz. (180 g) bottle
NDC #: 68032-420-18
Storage Conditions:
Store between 15-30° C (59°- 86°F). Protect from freezing.
PATIENT COUNSELING INFORMATION
GENERAL INFORMATION
Patients should be advised of the following:
• Avoid contact with eyes, eyelids, lips, and mucous membranes while using benzoyl peroxide. If accidental contact occurs, rinse with water.
• If excessive irritation develops, discontinue use and consult their physician.
• When using this product, avoid unnecessary sun exposure and use a sunscreen.
• Avoid contact with hair, fabrics, or carpeting as benzoyl peroxide will cause bleaching or discoloration.
ADVERSE REACTIONS
Patients should be informed that treatment with topical benzoyl peroxide may lead to adverse reactions that include allergic contact dermatitis and dryness.
EXCESSIVE USE OF TOPICAL BENZOYL PEROXIDE
Patients should be advised to discontinue use and consult their physician if excessive scaling, erythema, or edema occurs. Advise the use of cool compresses to hasten resolution of the adverse effects. After symptoms and signs subside, a reduced dosage schedule may be cautiously tried if the reaction is judged to be due to excessive use and not allergenicity.
Manufactured for:
River’s Edge Pharmaceuticals, LLC.
Suwanee, GA 30024
Rev. 07/09 420-115
PACKAGING
NDC 68032-420-18Rx Only
Benzoyl Peroxide Wash 7%
7% benzoyl peroxide
Net Wt. 6.35 oz. (180 g)
DIRECTIONS: Shake well before using. Wet skin areas to be treated; apply Benzoyl Peroxide Wash 7% by massaging gently into skin for 10-20 seconds, working into a full lather. Rinse thoroughly and pat dry. Avoid contact with eyes, eyelids, and lips.
ACTIVE INGREDIENTS: Benzoyl Peroxide 7%
INACTIVE INGREDIENTS: Citric Acid, cocamidopropyl betaine, cocamine oxide, disodium laureth sulfosuccinate, disodium EDTA, Fragrance Lemon, Glycerin, Hydrogenated Castor Oil, Hypromellose, Magnesium Aluminum Silicate, Cetearyl Alcohol and PEG-3 Distearoylamidoethylmonium Methosulfate and Polysorbate 60, Methylparaben, PEG-150 Pentaerythrityl Tetrastearate and Aqua and PEG-6 Caprylic/Capric Glycerides, PEG-40 Hydrogenated Castor Oil, Poloxamer 182, Purified Water and Xanthan Gum.
WARNINGS: KEEP OUT OF REACH OF CHILDREN. For External Use Only.
When using this product, avoid unnecessary sun exposure and use a sunscreen. Benzoyl peroxide may bleach colored or dyed fabric.
See package insert for full prescribing information.
Store between 15°-30°C (59°-86°F). Protect from freezing.
Manufacuted for:
River’s Edge Pharmaceuticals, LLC.
Suwanee, GA 30024
Iss. 6/09 420-26

| BENZOYL PEROXIDE
benzoyl peroxide 7% liquid |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| unapproved drug other | 06/01/2009 | ||
| Labeler - River's Edge Pharmaceuticals, LLC (133879135) |