nexium (Esomeprazole magnesium) capsule, delayed release
[AstraZeneca LP]
DESCRIPTION
The active ingredient in NEXIUM® (esomeprazole magnesium) Delayed-Release Capsules is bis(5-methoxy-2-[(S)-[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfinyl]-1H-benzimidazole-1-yl) magnesium trihydrate, a compound that inhibits gastric acid secretion. Esomeprazole is the S-isomer of omeprazole, which is a mixture of the S- and R- isomers. Its empirical formula is (C17H18N3O3S)2Mg x 3 H2O with molecular weight of 767.2 as a trihydrate and 713.1 on an anhydrous basis. The structural formula is:
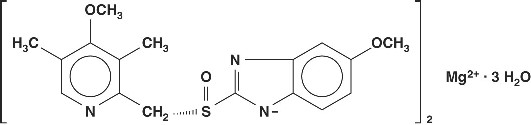
The magnesium salt is a white to slightly colored crystalline powder. It contains 3 moles of water of solvation and is slightly soluble in water.
The stability of esomeprazole magnesium is a function of pH; it rapidly degrades in acidic media, but it has acceptable stability under alkaline conditions. At pH 6.8 (buffer), the half-life of the magnesium salt is about 19 hours at 25°C and about 8 hours at 37°C.
NEXIUM is supplied as Delayed-Release Capsules for oral administration. Each delayed-release capsule contains 20 mg or 40 mg of esomeprazole (present as 22.3 mg or 44.5 mg esomeprazole magnesium trihydrate) in the form of enteric-coated pellets with the following inactive ingredients: glyceryl monostearate 40-50, hydroxypropyl cellulose, hypromellose, magnesium stearate, methacrylic acid copolymer type C, polysorbate 80, sugar spheres, talc, and triethyl citrate. The capsule shells have the following inactive ingredients: gelatin, FD&C Blue #1, FD&C Red #40, D&C Red #28, titanium dioxide, shellac, ethyl alcohol, isopropyl alcohol, n-butyl alcohol, propylene glycol, sodium hydroxide, polyvinyl pyrrolidone, and D&C Yellow #10.
CLINICAL PHARMACOLOGY
Pharmacokinetics
Absorption:
NEXIUM Delayed-Release Capsules contain an enteric-coated pellet formulation of esomeprazole magnesium. After oral administration peak plasma levels (Cmax) occur at approximately 1.5 hours (Tmax). The Cmax increases proportionally when the dose is increased, and there is a three-fold increase in the area under the plasma concentration-time curve (AUC) from 20 to 40 mg. At repeated once-daily dosing with 40 mg, the systemic bioavailability is approximately 90% compared to 64% after a single dose of 40 mg. The mean exposure (AUC) to esomeprazole increases from 4.32 µmol*hr/L on day 1 to 11.2 µmol*hr/L on day 5 after 40 mg once daily dosing.
The AUC after administration of a single 40 mg dose of esomeprazole is decreased by 43-53% after food intake compared to fasting conditions. Esomeprazole should be taken at least one hour before meals.
The pharmacokinetic profile of esomeprazole was determined in 36 patients with symptomatic gastroesophageal reflux disease following repeated once daily administration of 20 mg and 40 mg capsules of NEXIUM over a period of five days. The results are shown in the following table:
|
||
|
Parameter |
NEXIUM 40 mg |
NEXIUM 20 mg |
|
AUC (μmol*h/L) |
12.6 |
4.2 |
|
Coefficient of variation |
42% |
59% |
|
Cmax (μmol/L) |
4.7 |
2.1 |
|
Tmax (h) |
1.6 |
1.6 |
|
T1/2 (h) |
1.5 |
1.2 |
Distribution:
Esomeprazole is 97% bound to plasma proteins. Plasma protein binding is constant over the concentration range of 2-20 μmol/L. The apparent volume of distribution at steady state in healthy volunteers is approximately 16 L.
Metabolism:
Esomeprazole is extensively metabolized in the liver by the cytochrome P450 (CYP) enzyme system. The metabolites of esomeprazole lack antisecretory activity. The major part of esomeprazole’s metabolism is dependent upon the CYP2C19 isoenzyme, which forms the hydroxy and desmethyl metabolites. The remaining amount is dependent on CYP3A4 which forms the sulphone metabolite. CYP2C19 isoenzyme exhibits polymorphism in the metabolism of esomeprazole, since some 3% of Caucasians and 15-20% of Asians lack CYP2C19 and are termed Poor metabolizers. At steady state, the ratio of AUC in Poor metabolizers to AUC in the rest of the population (Extensive metabolizers) is approximately 2.
Following administration of equimolar doses, the S- and R-isomers are metabolized differently by the liver, resulting in higher plasma levels of the S- than of the R-isomer.
Excretion:
The plasma elimination half-life of esomeprazole is approximately 1-1.5 hours. Less than 1% of parent drug is excreted in the urine. Approximately 80% of an oral dose of esomeprazole is excreted as inactive metabolites in the urine, and the remainder is found as inactive metabolites in the feces.
Special Populations
Geriatric:
The AUC and Cmax values were slightly higher (25% and 18%, respectively) in the elderly as compared to younger subjects at steady state. Dosage adjustment based on age is not necessary.
Pediatric:
The pharmacokinetics of esomeprazole have not been studied in patients < 18 years of age.
Gender:
The AUC and Cmax values were slightly higher (13%) in females than in males at steady state. Dosage adjustment based on gender is not necessary.
Hepatic Insufficiency:
The steady state pharmacokinetics of esomeprazole obtained after administration of 40 mg once daily to 4 patients each with mild (Child Pugh A), moderate (Child Pugh Class B), and severe (Child Pugh Class C) liver insufficiency were compared to those obtained in 36 male and female GERD patients with normal liver function. In patients with mild and moderate hepatic insufficiency, the AUCs were within the range that could be expected in patients with normal liver function. In patients with severe hepatic insufficiency the AUCs were 2 to 3 times higher than in the patients with normal liver function. No dosage adjustment is recommended for patients with mild to moderate hepatic insufficiency (Child Pugh Classes A and B). However, in patients with severe hepatic insufficiency (Child Pugh Class C) a dose of 20 mg once daily should not be exceeded (See DOSAGE AND ADMINISTRATION)
Renal Insufficiency:
The pharmacokinetics of esomeprazole in patients with renal impairment are not expected to be altered relative to healthy volunteers as less than 1% of esomeprazole is excreted unchanged in urine.
Pharmacokinetics: Combination Therapy with Antimicrobials
Esomeprazole magnesium 40 mg once daily was given in combination with clarithromycin 500 mg twice daily and amoxicillin 1000 mg twice daily for 7 days to 17 healthy male and female subjects. The mean steady state AUC and Cmax of esomeprazole increased by 70% and 18%, respectively during triple combination therapy compared to treatment with esomeprazole alone. The observed increase in esomeprazole exposure during co-administration with clarithromycin and amoxicillin is not expected to produce significant safety concerns.
The pharmacokinetic parameters for clarithromycin and amoxicillin were similar during triple combination therapy and administration of each drug alone. However, the mean AUC and Cmax for 14-hydroxyclarithromycin increased by 19% and 22%, respectively, during triple combination therapy compared to treatment with clarithromycin alone. This increase in exposure to 14-hydroxyclarithromycin is not considered to be clinically significant.
Pharmacodynamics
Mechanism of Action:
Esomeprazole is a proton pump inhibitor that suppresses gastric acid secretion by specific inhibition of the H+/K+-ATPase in the gastric parietal cell. The S- and R-isomers of omeprazole are protonated and converted in the acidic compartment of the parietal cell forming the active inhibitor, the achiral sulphenamide. By acting specifically on the proton pump, esomeprazole blocks the final step in acid production, thus reducing gastric acidity. This effect is dose-related up to a daily dose of 20 to 40 mg and leads to inhibition of gastric acid secretion.
Antisecretory Activity:
The effect of esomeprazole on intragastric pH was determined in patients with symptomatic gastroesophageal reflux disease in two separate studies. In the first study of 36 patients, NEXIUM 40 mg and 20 mg capsules were administered over 5 days. The results are shown in the following table:
|
Parameter |
NEXIUM 40 mg |
NEXIUM 20 mg |
|
% Time Gastric pH >4* (Hours) |
70%† (16.8 h) |
53% (12.7 h) |
|
Coefficient of variation |
26% |
37% |
|
Median 24 Hour pH |
4.9* |
4.1 |
|
Coefficient of variation |
16% |
27% |
In a second study, the effect on intragastric pH of NEXIUM 40 mg administered once daily over a five day period was similar to the first study, (% time with pH>4 was 68% or 16.3 hours).
Serum Gastrin Effects:
The effect of NEXIUM on serum gastrin concentrations was evaluated in approximately 2,700 patients in clinical trials up to 8 weeks and in over 1,300 patients for up to 6-12 months. The mean fasting gastrin level increased in a dose-related manner. This increase reached a plateau within two to three months of therapy and returned to baseline levels within four weeks after discontinuation of therapy.
Enterochromaffin-like (ECL) Cell Effects:
In 24-month carcinogenicity studies of omeprazole in rats, a dose-related significant occurrence of gastric ECL cell carcinoid tumors and ECL cell hyperplasia was observed in both male and female animals (see PRECAUTIONS, Carcinogenesis, Mutagenesis, Impairment of Fertility). Carcinoid tumors have also been observed in rats subjected to fundectomy or long-term treatment with other proton pump inhibitors or high doses of H2-receptor antagonists.
Human gastric biopsy specimens have been obtained from more than 3,000 patients treated with omeprazole in long-term clinical trials. The incidence of ECL cell hyperplasia in these studies increased with time; however, no case of ECL cell carcinoids, dysplasia, or neoplasia has been found in these patients.
In over 1,000 patients treated with NEXIUM (10, 20 or 40 mg/day) up to 6-12 months, the prevalence of ECL cell hyperplasia increased with time and dose. No patient developed ECL cell carcinoids, dysplasia, or neoplasia in the gastric mucosa.
Endocrine Effects:
NEXIUM had no effect on thyroid function when given in oral doses of 20 or 40 mg for 4 weeks. Other effects of NEXIUM on the endocrine system were assessed using omeprazole studies. Omeprazole given in oral doses of 30 or 40 mg for 2 to 4 weeks had no effect on carbohydrate metabolism, circulating levels of parathyroid hormone, cortisol, estradiol, testosterone, prolactin, cholecystokinin or secretin.
Microbiology
Esomeprazole magnesium, amoxicillin and clarithromycin triple therapy has been shown to be active against most strains of Helicobacter pylori (H. pylori)in vitro and in clinical infections as described in the Clinical Studies and INDICATIONS AND USAGE sections.
Helicobacter
Helicobacter pylori:
Susceptibility testing of H. pylori isolates was performed for amoxicillin and clarithromycin using agar dilution methodology, and minimum inhibitory concentrations (MICs) were determined.
Pretreatment Resistance:
Clarithromycin pretreatment resistance rate (MIC ≥ 1 μg/mL) to H. pylori was 15% (66/445) at baseline in all treatment groups combined. A total of > 99% (394/395) of patients had H. pylori isolates which were considered to be susceptible (MIC ≤ 0.25 μg/mL) to amoxicillin at baseline. One patient had a baseline H. pylori isolate with an amoxicillin MIC = 0.5 μg/mL.
Clarithromycin Susceptibility Test Results and Clinical/Bacteriologic Outcomes:
The baseline H. pylori clarithromycin susceptibility results and the H. pylori eradication results at the Day 38 visit are shown in the table below:
|
Clarithromycin Pretreatment Results |
H. pylori negative (Eradicated) |
H. pylori positive (Not Eradicated) Post-treatment susceptibility results |
||||
|
S† |
I† |
R† |
No MIC |
|||
|
Susceptible† |
182 |
162 |
4 |
0 |
2 |
14 |
|
Intermediate† |
1 |
1 |
0 |
0 |
0 |
0 |
|
Resistant† |
29 |
13 |
1 |
0 |
13 |
2 |
Patients not eradicated of H. pylori following esomeprazole magnesium/amoxicillin/clarithromycin triple therapy will likely have clarithromycin resistant H. pylori isolates. Therefore, clarithromycin susceptibility testing should be done, when possible. Patients with clarithromycin resistant H. pylori should not be re-treated with a clarithromycin-containing regimen.
Amoxicillin Susceptibility Test Results and Clinical/Bacteriological Outcomes:
In the esomeprazole magnesium/amoxicillin/clarithromycin clinical trials, 83% (176/212) of the patients in the esomeprazole magnesium/amoxicillin/clarithromycin treatment group who had pretreatment amoxicillin susceptible MICs (≤ 0.25 μg/mL) were eradicated of H. pylori, and 17% (36/212) were not eradicated of H. pylori. Of the 36 patients who were not eradicated of H. pylori on triple therapy, 16 had no post-treatment susceptibility test results and 20 had post-treatment H. pylori isolates with amoxicillin susceptible MICs. Fifteen of the patients who were not eradicated of H. pylori on triple therapy also had post-treatment H. pylori isolates with clarithromycin resistant MICs. There were no patients with H. pylori isolates who developed treatment emergent resistance to amoxicillin.
Susceptibility Test for Helicobacter pylori:
The reference methodology for susceptibility testing of H. pylori is agar dilution MICs. One to three microliters of an inoculum equivalent to a No.2 McFarland standard (1 x 107 - 1 x 108 CFU/mL for H. pylori) are inoculated directly onto freshly prepared antimicrobial containing Mueller-Hinton agar plates with 5% aged defibrinated sheep blood (> 2 weeks old). The agar dilution plates are incubated at 35°C in a microaerobic environment produced by a gas generating system suitable for Campylobacter. After 3 days of incubation, the MICs are recorded as the lowest concentration of antimicrobial agent required to inhibit growth of the organism. The clarithromycin and amoxicillin MIC values should be interpreted according to the following criteria:
|
Clarithromycin MIC(μg/mL)* |
Interpretation |
|
≤ 0.25 |
Susceptible (S) |
|
0.5 |
Intermediate (I) |
|
≥1.0 |
Resistant (R) |
|
Amoxicillin MIC (μg/mL)* † |
Interpretation |
|
≤ 0.25 |
Susceptible (S) |
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard clarithromycin and amoxicillin powders should provide the following MIC values:
|
||
|
Microorganism |
Antimicrobial Agent |
MIC (μg/mL)* |
|
H. pylori ATCC 43504 |
Clarithromycin |
0.016 − 0.12 (μg/mL) |
|
H. pylori ATCC 43504 |
Amoxicillin |
0.016 − 0.12 (μg/mL) |
Clinical Studies
Healing of Erosive Esophagitis:
The healing rates of NEXIUM 40 mg, NEXIUM 20 mg, and omeprazole 20 mg (the approved dose for this indication) were evaluated in patients with endoscopically diagnosed erosive esophagitis in four multicenter, double-blind, randomized studies. The healing rates at weeks 4 and 8 were evaluated and are shown in the table below:
|
Study |
No. of Patients |
Treatment Groups |
Week 4 |
Week 8 |
Significance Level* |
|
1 |
588 |
NEXIUM 20 mg |
68.7% |
90.6% |
N.S.† |
|
588 |
Omeprazole 20 mg |
69.5% |
88.3% |
||
|
2 |
654 |
NEXIUM 40 mg |
75.9% |
94.1% |
p < 0.001 |
|
656 |
NEXIUM 20 mg |
70.5% |
89.9% |
p < 0.05 |
|
|
650 |
Omeprazole 20 mg |
64.7% |
86.9% |
||
|
3 |
576 |
NEXIUM 40 mg |
71.5% |
92.2% |
N.S.† |
|
572 |
Omeprazole 20 mg |
68.6% |
89.8% |
||
|
4 |
1216 |
NEXIUM 40 mg |
81.7% |
93.7% |
p < 0.001 |
|
1209 |
Omeprazole 20 mg |
68.7% |
84.2% |
||
In these same studies of patients with erosive esophagitis, sustained heartburn resolution and time to sustained heartburn resolution were evaluated and are shown in the table below:
|
Cumulative Percent† with Sustained Resolution |
|||||
|
Study |
No. of Patients |
Treatment Groups |
Day 14 |
Day 28 |
Significance Level‡ |
|
1 |
573 |
NEXIUM 20 mg |
64.3% |
72.7% |
N.S.† |
|
555 |
Omeprazole 20 mg |
64.1% |
70.9% |
||
|
2 |
621 |
NEXIUM 40 mg |
64.8% |
74.2% |
p <0.001 |
|
620 |
NEXIUM 20 mg |
62.9% |
70.1% |
N.S.† |
|
|
626 |
Omeprazole 20 mg |
56.5% |
66.6% |
||
|
3 |
568 |
NEXIUM 40 mg |
65.4% |
73.9% |
N.S.† |
|
551 |
Omeprazole 20 mg |
65.5% |
73.1% |
||
|
4 |
1187 |
NEXIUM 40 mg |
67.6% |
75.1% |
P <0.001 |
|
1188 |
Omeprazole 20 mg |
62.5% |
70.8% |
||
In these four studies, the range of median days to the start of sustained resolution (defined as 7 consecutive days with no heartburn) was 5 days for NEXIUM 40 mg, 7-8 days for NEXIUM 20 mg and 7-9 days for omeprazole 20 mg.
There are no comparisons of 40 mg of NEXIUM with 40 mg of omeprazole in clinical trials assessing either healing or symptomatic relief of erosive esophagitis.
Long-Term Maintenance of Healing of Erosive Esophagitis:
Two multicenter, randomized, double-blind placebo-controlled 4-arm trials were conducted in patients with endoscopically confirmed, healed erosive esophagitis to evaluate NEXIUM 40 mg (n=174), 20 mg (n=180), 10 mg (n= 168) or placebo (n=171) once daily over six months of treatment.
No additional clinical benefit was seen with NEXIUM 40 mg over NEXIUM 20 mg.
The percentage of patients that maintained healing of erosive esophagitis at the various time points are shown in the figures below:
Maintenance of Healing Rates by Month (Study 177)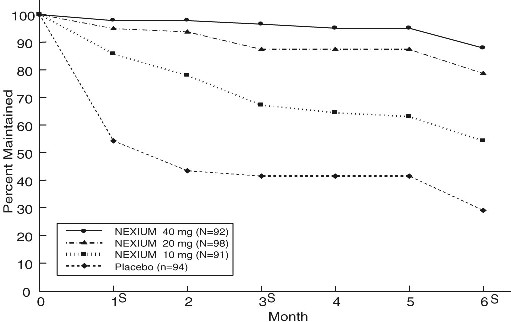 s= scheduled visit
s= scheduled visit
Maintenance of Healing Rates by Month (Study 178)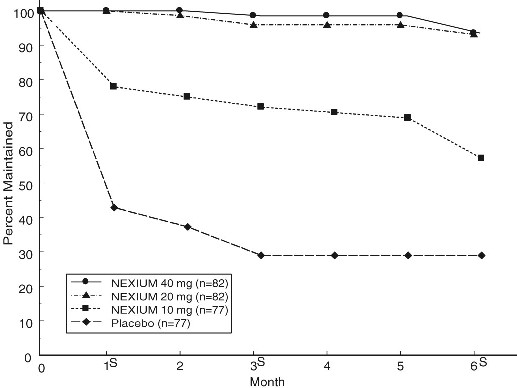 s=scheduled visit
s=scheduled visit
Patients remained in remission significantly longer and the number of recurrences of erosive esophagitis was significantly less in patients treated with NEXIUM compared to placebo.
In both studies, the proportion of patients on NEXIUM who remained in remission and were free of heartburn and other GERD symptoms was well differentiated from placebo.
In a third multicenter open label study of 808 patients treated for 12 months with NEXIUM 40 mg, the percentage of patients that maintained healing of erosive esophagitis was 93.7% for six months and 89.4% for one year.
Symptomatic Gastroesophageal Reflux Disease (GERD):
Two multicenter, randomized, double-blind, placebo-controlled studies were conducted in a total of 717 patients comparing four weeks of treatment with NEXIUM 20 mg or 40 mg once daily versus placebo for resolution of GERD symptoms. Patients had ≥ 6-month history of heartburn episodes, no erosive esophagitis by endoscopy, and heartburn on at least four of the seven days immediately preceding randomization.
The percentage of patients that were symptom-free of heartburn was significantly higher in the NEXIUM groups compared to placebo at all follow-up visits (Weeks 1, 2, and 4).
No additional clinical benefit was seen with NEXIUM 40 mg over NEXIUM 20 mg.
The percent of patients symptom-free of heartburn by day are shown in the figures below:
Percent of Patients Symptom-Free of Heartburn by Day (Study 225)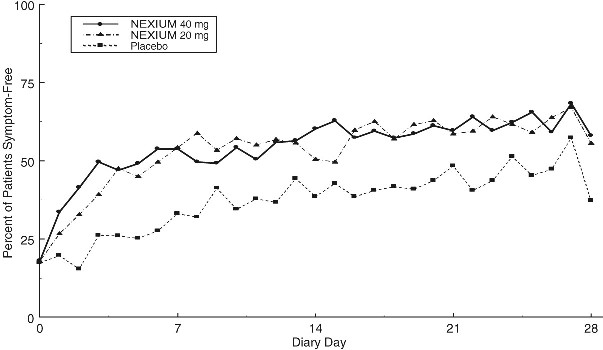
Percent of Patients Symptom-Free of Heartburn by Day (Study 226)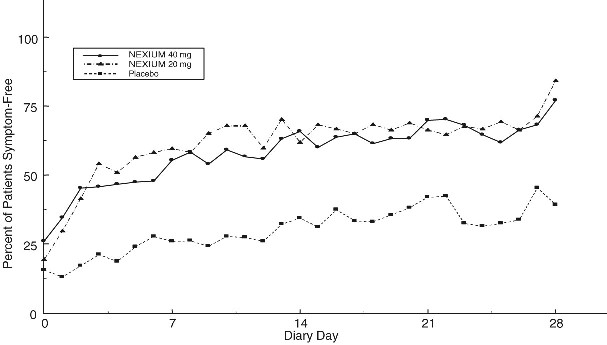
In three European symptomatic GERD trials, NEXIUM 20 mg and 40 mg and omeprazole 20 mg were evaluated. No significant treatment related differences were seen.
Risk Reduction of NSAID-Associated Gastric Ulcer:
Two multicenter, double-blind, placebo-controlled studies were conducted in patients at risk of developing gastric and/or duodenal ulcers associated with continuous use of non-selective and COX-2 selective NSAIDs. A total of 1429 patients were randomized across the 2 studies. Patients ranged in age from 19 to 89 (median age 66.0 years) with 70.7% female, 29.3% male, 82.9% Caucasian, 5.5% Black, 3.7% Asian, and 8.0% Others. At baseline, the patients in these studies were endoscopically confirmed not to have ulcers but were determined to be at risk for ulcer occurrence due to their age (≥60 years) and/or history of a documented gastric or duodenal ulcer within the past 5 years. Patients receiving NSAIDs and treated with NEXIUM 20 mg or 40 mg once-a-day experienced significant reduction in gastric ulcer occurrences relative to placebo treatment at 26 weeks. No additional benefit was seen with NEXIUM 40 mg over NEXIUM 20 mg. These studies did not demonstrate significant reduction in the development of NSAID-associated duodenal ulcer due to the low incidence.
|
Study |
No. of Patients |
Treatment Group |
% of Patients Remaining Gastric Ulcer Free* |
|
|||
|
1 |
191 |
NEXIUM 20 mg |
95.4 |
|
194 |
NEXIUM 40 mg |
96.7 |
|
|
184 |
Placebo |
88.2 |
|
|
2 |
267 |
NEXIUM 20 mg |
94.7 |
|
271 |
NEXIUM 40 mg |
95.3 |
|
|
257 |
Placebo |
83.3 |
|
Helicobacter pylori (H. pylori) Eradication in Patients with Duodenal Ulcer Disease:
Triple Therapy (NEXIUM/amoxicillin/clarithromycin): Two multicenter, randomized, double-blind studies were conducted using a 10 day treatment regimen. The first study (191) compared NEXIUM 40 mg once daily in combination with amoxicillin 1000 mg twice daily and clarithromycin 500 mg twice daily to NEXIUM 40 mg once daily plus clarithromycin 500 mg twice daily. The second study (193) compared NEXIUM 40 mg once daily in combination with amoxicillin 1000 mg twice daily and clarithromycin 500 mg twice daily to NEXIUM 40 mg once daily. H. pylori eradication rates, defined as at least two negative tests and no positive tests from CLOtest®, histology and/or culture, at 4 weeks post-therapy were significantly higher in the NEXIUM plus amoxicillin and clarithromycin group than in the NEXIUM plus clarithromycin or NEXIUM alone group. The results are shown in the following table:
|
|||
|
% of Patients Cured [95% Confidence Interval] (Number of patients) |
|||
|
Study |
Treatment Group |
Pre-Protocol* |
Intent-to-Treat† |
|
191 |
NEXIUM plus amoxicillin and clarithromycin |
84%‡ [78, 89] (n=196) |
77%‡ [71, 82] (n=233) |
|
NEXIUM plus clarithromycin |
55% [48, 62] (n=187 |
52% [45, 59] (n=215) |
|
|
193 |
NEXIUM plus amoxicillin and clarithromycin |
85%§ [74, 93] (n=67) |
78%§ [67, 87] (n=74) |
|
NEXIUM |
5% [0, 23] (n=22) |
4% [0, 21] (n=24) |
|
The percentage of patients with a healed baseline duodenal ulcer by 4 weeks after the 10 day treatment regimen in the NEXIUM plus amoxicillin and clarithromycin group was 75% (n=156) and 57% (n=60) respectively, in the 191 and 193 studies (per-protocol analysis).
INDICATIONS AND USAGE
Treatment of Gastroesophageal Reflux Disease (GERD)
Healing of Erosive Esophagitis:
NEXIUM is indicated for the short-term treatment (4 to 8 weeks) in the healing and symptomatic resolution of diagnostically confirmed erosive esophagitis. For those patients who have not healed after 4-8 weeks of treatment, an additional 4-8-week course of NEXIUM may be considered.
Maintenance of Healing of Erosive Esophagitis:
NEXIUM is indicated to maintain symptom resolution and healing of erosive esophagitis. Controlled studies do not extend beyond 6 months.
Symptomatic Gastroesophageal Reflux Disease:
NEXIUM is indicated for treatment of heartburn and other symptoms associated with GERD.
Risk Reduction of NSAID-Associated Gastric Ulcer
NEXIUM is indicated for the reduction in the occurrence of gastric ulcers associated with continuous NSAID therapy in patients at risk for developing gastric ulcers. Patients are considered to be at risk due to their age (> 60) and/or documented history of gastric ulcers. Controlled studies do not extend beyond 6 months.
H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence
Triple Therapy (NEXIUMplus amoxicillin and clarithromycin): NEXIUM, in combination with amoxicillin and clarithromycin, is indicated for the treatment of patients with H. pylori infection and duodenal ulcer disease (active or history of within the past 5 years) to eradicate H. pylori. Eradication of H. pylori has been shown to reduce the risk of duodenal ulcer recurrence. (See Clinical Studies and DOSAGE AND ADMINISTRATION)
In patients who fail therapy, susceptibility testing should be done. If resistance to clarithromycin is demonstrated or susceptibility testing is not possible, alternative antimicrobial therapy should be instituted. (See CLINICAL PHARMACOLOGY, Microbiology and the clarithromycin package insert, CLINICAL PHARMACOLOGY,Microbiology.)
CONTRAINDICATIONS
NEXIUM is contraindicated in patients with known hypersensitivity to any component of the formulation or to substituted benzimidazoles.
Clarithromycin is contraindicated in patients with a known hypersensitivity to any macrolide antibiotic.
Concomitant administration of clarithromycin with pimozide is contraindicated. There have been post-marketing reports of drug interactions when clarithromycin and/or erythromycin are co-administered with pimozide resulting in cardiac arrhythmias (QT prolongation, ventricular tachycardia, ventricular fibrillation, and torsade de pointes) most likely due to inhibition of hepatic metabolism of pimozide by erythromycin and clarithromycin. Fatalities have been reported. (Please refer to full prescribing information for clarithromycin.)
Amoxicillin is contraindicated in patients with a known hypersensitivity to any penicillin. (Please refer to full prescribing information for amoxicillin.)
WARNINGS
CLARITHROMYCIN SHOULD NOT BE USED IN PREGNANT WOMEN EXCEPT IN CLINICAL CIRCUMSTANCES WHERE NO ALTERNATIVE THERAPY IS APPROPRIATE. IF PREGNANCY OCCURS WHILE TAKING CLARITHROMYCIN, THE PATIENT SHOULD BE APPRISED OF THE POTENTIAL HAZARD TO THE FETUS. (See WARNINGSin prescribing information for clarithromycin.)
Amoxicillin: Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients on penicillin therapy. These reactions are more apt to occur in individuals with a history of penicillin hypersensitivity and/or a history of sensitivity to multiple allergens.
There have been well documented reports of individuals with a history of penicillin hypersensitivity reactions who have experienced severe hypersensitivity reactions when treated with a cephalosporin. Before initiating therapy with any penicillin, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, and other allergens. If an allergic reaction occurs, amoxicillin should be discontinued and the appropriate therapy instituted.
SERIOUS ANAPHYLACTIC REACTIONS REQUIRE IMMEDIATE EMERGENCY TREATMENT WITH EPINEPHRINE. OXYGEN, INTRAVENOUS STEROIDS, AND AIRWAY MANAGEMENT, INCLUDING INTUBATION, SHOULD ALSO BE ADMINISTERED AS INDICATED.
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including clarithromycin and amoxicillin, and may range in severity from mild to life threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that a toxin produced by Clostridium difficile is a primary cause of “antibiotic-associated colitis”.
After the diagnosis of pseudomembranous colitis has been established, therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to discontinuation of the drug alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation, and treatment with an antibacterial drug clinically effective against Clostridium difficile colitis.
PRECAUTIONS
General
Symptomatic response to therapy with NEXIUM does not preclude the presence of gastric malignancy.
Atrophic gastritis has been noted occasionally in gastric corpus biopsies from patients treated long-term with omeprazole, of which NEXIUM is an enantiomer.
Information for Patients
Patients should be informed of the following:
NEXIUM Delayed-Release Capsules should be taken at least one hour before meals.
For patients who have difficulty swallowing capsules, one tablespoon of applesauce can be added to an empty bowl and the NEXIUM Delayed-Release Capsule can be opened, and the pellets inside the capsule carefully emptied onto the applesauce. The pellets should be mixed with the applesauce and then swallowed immediately. The applesauce used should not be hot and should be soft enough to be swallowed without chewing. The pellets should not be chewed or crushed. The pellet/applesauce mixture should not be stored for future use.
Antacids may be used while taking NEXIUM.
Drug Interactions
Esomeprazole is extensively metabolized in the liver by CYP2C19 and CYP3A4.
In vitro and in vivo studies have shown that esomeprazole is not likely to inhibit CYPs 1A2, 2A6, 2C9, 2D6, 2E1 and 3A4. No clinically relevant interactions with drugs metabolized by these CYP enzymes would be expected. Drug interaction studies have shown that esomeprazole does not have any clinically significant interactions with phenytoin, warfarin, quinidine, clarithromycin or amoxicillin. Post-marketing reports of changes in prothrombin measures have been received among patients on concomitant warfarin and esomeprazole therapy. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Patients treated with proton pump inhibitors and warfarin concomitantly may need to be monitored for increases in INR and prothrombin time.
Esomeprazole may potentially interfere with CYP2C19, the major esomeprazole metabolizing enzyme. Coadministration of esomeprazole 30 mg and diazepam, a CYP2C19 substrate, resulted in a 45% decrease in clearance of diazepam. Increased plasma levels of diazepam were observed 12 hours after dosing and onwards. However, at that time, the plasma levels of diazepam were below the therapeutic interval, and thus this interaction is unlikely to be of clinical relevance.
Coadministration of oral contraceptives, diazepam, phenytoin, or quinidine did not seem to change the pharmacokinetic profile of esomeprazole.
Concomitant administration of esomeprazole may reduce the plasma levels of atazanavir.
Studies evaluating concomitant administration of esomeprazole and either naproxen (non-selective NSAID) or rofecoxib (COX-2 selective NSAID) did not identify any clinically relevant changes in the pharmacokinetic profiles of esomeprazole or these NSAIDs.
Esomeprazole inhibits gastric acid secretion. Therefore, esomeprazole may interfere with the absorption of drugs where gastric pH is an important determinant of bioavailability (eg, ketoconazole, iron salts and digoxin).
Combination Therapy with Clarithromycin:
Co-administration of esomeprazole, clarithromycin, and amoxicillin has resulted in increases in the plasma levels of esomeprazole and 14-hydroxyclarithromycin. (See CLINICAL PHARMACOLOGY, Pharmacokinetics: Combination Therapy with Antimicrobials.)
Concomitant administration of clarithromycin with pimozide is contraindicated. (See clarithromycin package insert.)
Carcinogenesis, Mutagenesis, Impairment of Fertility:
The carcinogenic potential of esomeprazole was assessed using omeprazole studies. In two 24-month oral carcinogenicity studies in rats, omeprazole at daily doses of 1.7, 3.4, 13.8, 44.0 and 140.8 mg/kg/day (about 0.7 to 57 times the human dose of 20 mg/day expressed on a body surface area basis) produced gastric ECL cell carcinoids in a dose-related manner in both male and female rats; the incidence of this effect was markedly higher in female rats, which had higher blood levels of omeprazole. Gastric carcinoids seldom occur in the untreated rat. In addition, ECL cell hyperplasia was present in all treated groups of both sexes. In one of these studies, female rats were treated with 13.8 mg omeprazole/kg/day (about 5.6 times the human dose on a body surface area basis) for 1 year, then followed for an additional year without the drug. No carcinoids were seen in these rats. An increased incidence of treatment-related ECL cell hyperplasia was observed at the end of 1 year (94% treated vs 10% controls). By the second year the difference between treated and control rats was much smaller (46% vs 26%) but still showed more hyperplasia in the treated group. Gastric adenocarcinoma was seen in one rat (2%). No similar tumor was seen in male or female rats treated for 2 years. For this strain of rat no similar tumor has been noted historically, but a finding involving only one tumor is difficult to interpret. A 78-week mouse carcinogenicity study of omeprazole did not show increased tumor occurrence, but the study was not conclusive.
Esomeprazole was negative in the Ames mutation test, in the in vivo rat bone marrow cell chromosome aberration test, and the in vivo mouse micronucleus test. Esomeprazole, however, was positive in the in vitro human lymphocyte chromosome aberration test. Omeprazole was positive in the in vitro human lymphocyte chromosome aberration test, the in vivo mouse bone marrow cell chromosome aberration test, and the in vivo mouse micronucleus test.
The potential effects of esomeprazole on fertility and reproductive performance were assessed using omeprazole studies. Omeprazole at oral doses up to 138 mg/kg/day in rats (about 56 times the human dose on a body surface area basis) was found to have no effect on reproductive performance of parental animals.
Pregnancy
Teratogenic Effects. Pregnancy Category B:
Teratology studies have been performed in rats at oral doses up to 280 mg/kg/day (about 57 times the human dose on a body surface area basis) and in rabbits at oral doses up to 86 mg/kg/day (about 35 times the human dose on a body surface area basis) and have revealed no evidence of impaired fertility or harm to the fetus due to esomeprazole. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Teratology studies conducted with omeprazole in rats at oral doses up to 138 mg/kg/day (about 56 times the human dose on a body surface area basis) and in rabbits at doses up to 69 mg/kg/day (about 56 times the human dose on a body surface area basis) did not disclose any evidence for a teratogenic potential of omeprazole. In rabbits, omeprazole in a dose range of 6.9 to 69.1 mg/kg/day (about 5.5 to 56 times the human dose on a body surface area basis) produced dose-related increases in embryo-lethality, fetal resorptions, and pregnancy disruptions. In rats, dose-related embryo/fetal toxicity and postnatal developmental toxicity were observed in offspring resulting from parents treated with omeprazole at 13.8 to 138.0 mg/kg/day (about 5.6 to 56 times the human doses on a body surface area basis). There are no adequate and well-controlled studies in pregnant women. Sporadic reports have been received of congenital abnormalities occurring in infants born to women who have received omeprazole during pregnancy.
Amoxicillin
Pregnancy Category B. See full prescribing information for amoxicillin before using in pregnant women.
Clarithromycin
Pregnancy Category C. See WARNINGS (above) and full prescribing information for clarithromycin before using in pregnant women.
Nursing Mothers
The excretion of esomeprazole in milk has not been studied. However, omeprazole concentrations have been measured in breast milk of a woman following oral administration of 20 mg. Because esomeprazole is likely to be excreted in human milk, because of the potential for serious adverse reactions in nursing infants from esomeprazole, and because of the potential for tumorigenicity shown for omeprazole in rat carcinogenicity studies, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatric Use
Of the total number of patients who received NEXIUM in clinical trials, 1459 were 65 to 74 years of age and 354 patients were ≥ 75 years of age.
No overall differences in safety and efficacy were observed between the elderly and younger individuals, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
ADVERSE REACTIONS
The safety of NEXIUM was evaluated in over 15,000 patients (aged 18-84 years) in clinical trials worldwide including over 8,500 patients in the United States and over 6,500 patients in Europe and Canada. Over 2,900 patients were treated in long-term studies for up to 6-12 months. In general, NEXIUM was well tolerated in both short and long-term clinical trials.
The safety in the treatment of healing of erosive esophagitis was assessed in four randomized comparative clinical trials, which included 1,240 patients on NEXIUM 20 mg, 2,434 patients on NEXIUM 40 mg, and 3,008 patients on omeprazole 20 mg daily. The most frequently occurring adverse events (≥1%) in all three groups was headache (5.5, 5.0, and 3.8, respectively) and diarrhea (no difference among the three groups). Nausea, flatulence, abdominal pain, constipation, and dry mouth occurred at similar rates among patients taking NEXIUM or omeprazole.
Additional adverse events that were reported as possibly or probably related to NEXIUM with an incidence < 1% are listed below by body system:
Body as a Whole: abdomen enlarged, allergic reaction, asthenia, back pain, chest pain, chest pain substernal, facial edema, peripheral edema, hot flushes, fatigue, fever, flu-like disorder, generalized edema, leg edema, malaise, pain, rigors; Cardiovascular: flushing, hypertension, tachycardia; Endocrine: goiter; Gastrointestinal: bowel irregularity, constipation aggravated, dyspepsia, dysphagia, dysplasia GI, epigastric pain, eructation, esophageal disorder, frequent stools, gastroenteritis, GI hemorrhage, GI symptoms not otherwise specified, hiccup, melena, mouth disorder, pharynx disorder, rectal disorder, serum gastrin increased, tongue disorder, tongue edema, ulcerative stomatitis, vomiting; Hearing: earache, tinnitus; Hematologic: anemia, anemia hypochromic, cervical lymphoadenopathy, epistaxis, leukocytosis, leukopenia, thrombocytopenia; Hepatic: bilirubinemia, hepatic function abnormal, SGOT increased, SGPT increased; Metabolic/Nutritional: glycosuria, hyperuricemia, hyponatremia, increased alkaline phosphatase, thirst, vitamin B12 deficiency, weight increase, weight decrease; Musculoskeletal: arthralgia, arthritis aggravated, arthropathy, cramps, fibromyalgia syndrome, hernia, polymyalgia rheumatica; Nervous System/Psychiatric: anorexia, apathy, appetite increased, confusion, depression aggravated, dizziness, hypertonia, nervousness, hypoesthesia, impotence, insomnia, migraine, migraine aggravated, paresthesia, sleep disorder, somnolence, tremor, vertigo, visual field defect; Reproductive: dysmenorrhea, menstrual disorder, vaginitis; Respiratory: asthma aggravated, coughing, dyspnea, larynx edema, pharyngitis, rhinitis, sinusitis; Skin and Appendages: acne, angioedema, dermatitis, pruritus, pruritus ani, rash, rash erythematous, rash maculo-papular, skin inflammation, sweating increased, urticaria; Special Senses: otitis media, parosmia, taste loss, taste perversion; Urogenital: abnormal urine, albuminuria, cystitis, dysuria, fungal infection, hematuria, micturition frequency, moniliasis, genital moniliasis, polyuria; Visual: conjunctivitis, vision abnormal.
Endoscopic findings that were reported as adverse events include: duodenitis, esophagitis, esophageal stricture, esophageal ulceration, esophageal varices, gastric ulcer, gastritis, hernia, benign polyps or nodules, Barrett’s esophagus, and mucosal discoloration.
The incidence of treatment-related adverse events during 6-month maintenance treatment was similar to placebo. There were no differences in types of related adverse events seen during maintenance treatment up to 12 months compared to short-term treatment.
Two placebo-controlled studies were conducted in 710 patients for the treatment of symptomatic gastroesophageal reflux disease. The most common adverse events that were reported as possibly or probably related to NEXIUM were diarrhea (4.3%), headache (3.8%), and abdominal pain (3.8%).
Postmarketing Reports - There have been spontaneous reports of adverse events with postmarketing use of esomeprazole. These reports occurred rarely and are listed below by body system: Blood And Lymphatic System Disorders: agranulocytosis, pancytopenia; Eye Disorders: blurred vision; Gastrointestinal Disorders: pancreatitis; Hepatobiliary Disorders: hepatitis with or without jaundice; Immune System Disorders: anaphylactic reaction/shock; Musculoskeletal And Connective Tissue Disorders: myalgia; Psychiatric Disorders: depression; Skin and Subcutaneous Tissue Disorders: alopecia, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis (TEN, some fatal).
Other adverse events not observed with NEXIUM, but occurring with omeprazole can be found in the omeprazole package insert, ADVERSE REACTIONS section.
Combination Treatment with Amoxicillin and Clarithromycin
In clinical trials using combination therapy with NEXIUM plus amoxicillin and clarithromycin, no adverse events peculiar to these drug combinations were observed. Adverse events that occurred have been limited to those that had been observed with either NEXIUM, amoxicillin, or clarithromycin alone.
The most frequently reported drug-related adverse events for patients who received triple therapy for 10 days were diarrhea (9.2%), taste perversion (6.6%), and abdominal pain (3.7%). No treatment-emergent adverse events were observed at higher rates with triple therapy than were observed with NEXIUM alone.
For more information on adverse events with amoxicillin or clarithromycin, refer to their package inserts, ADVERSE REACTIONS sections.
Laboratory Events
The following potentially clinically significant laboratory changes in clinical trials, irrespective of relationship to NEXIUM, were reported in ≤ 1% of patients: increased creatinine, uric acid, total bilirubin, alkaline phosphatase, ALT, AST, hemoglobin, white blood cell count, platelets, serum gastrin, potassium, sodium, thyroxine and thyroid stimulating hormone (see CLINICAL PHARMACOLOGY, Endocrine Effects for further information on thyroid effects). Decreases were seen in hemoglobin, white blood cell count, platelets, potassium, sodium, and thyroxine.
In clinical trials using combination therapy with NEXIUM plus amoxicillin and clarithromycin, no additional increased laboratory abnormalities particular to these drug combinations were observed.
For more information on laboratory changes with amoxicillin or clarithromycin, refer to their package inserts, ADVERSE REACTIONS section.
OVERDOSAGE
A single oral dose of esomeprazole at 510 mg/kg (about 103 times the human dose on a body surface area basis), was lethal to rats. The major signs of acute toxicity were reduced motor activity, changes in respiratory frequency, tremor, ataxia, and intermittent clonic convulsions.
There have been some reports of overdosage with esomeprazole. Reports have been received of overdosage with omeprazole in humans. Doses ranged up to 2,400 mg (120 times the usual recommended clinical dose). Manifestations were variable, but included confusion, drowsiness, blurred vision, tachycardia, nausea, diaphoresis, flushing, headache, dry mouth, and other adverse reactions similar to those seen in normal clinical experience (see omeprazole package insert - ADVERSE REACTIONS). No specific antidote for esomeprazole is known. Since esomeprazole is extensively protein bound, it is not expected to be removed by dialysis. In the event of overdosage, treatment should be symptomatic and supportive.
As with the management of any overdose, the possibility of multiple drug ingestion should be considered. For current information on treatment of any drug overdose, a certified Regional Poison Control Center should be contacted. Telephone numbers are listed in the Physicians’ Desk Reference (PDR) or local telephone book.
DOSAGE AND ADMINISTRATION
The recommended adult dosages are outlined in the table below. NEXIUM Delayed-Release Capsules should be swallowed whole and taken at least one hour before eating.
For patients who have difficulty swallowing capsules, one tablespoon of applesauce can be added to an empty bowl and the NEXIUM Delayed-Release Capsule can be opened, and the pellets inside the capsule carefully emptied onto the applesauce. The pellets should be mixed with the applesauce and then swallowed immediately. The applesauce used should not be hot and should be soft enough to be swallowed without chewing. The pellets should not be chewed or crushed. The pellet/applesauce mixture should not be stored for future use.
For patients who have a nasogastric tube in place, NEXIUM Delayed-Release Capsules can be opened and the intact granules emptied into a 60 mL syringe and mixed with 50 mL of water. Replace the plunger and shake the syringe vigorously for 15 seconds. Hold the syringe with the tip up and check for granules remaining in the tip. Attach the syringe to a nasogastric tube and deliver the contents of the syringe through the nasogastric tube into the stomach. After administering the granules, the nasogastric tube should be flushed with additional water. Do not administer the pellets if they have dissolved or disintegrated.
The suspension must be used immediately after preparation.
|
Indication |
Dose |
Frequency |
|
||
|
Gastroesophageal Reflux Disease (GERD) |
||
|
Healing of Erosive Esophagitis |
20 mg or 40 mg |
Once Daily for 4 to 8 Weeks* |
|
Maintenance of Healing of Erosive Esophagitis |
20 mg |
Once Daily† |
|
Symptomatic Gastroesophageal Reflux Disaese |
20 mg |
Once Daily for 4 Weeks‡ |
|
Risk Reduction of NSAID- Associated Gastric Ulcer |
20 mg or 40 mg |
Once Daily for up to 6 months† |
|
H. pylori Eradication to Reduce the Risk of Duodenal Ulcer Recurrence |
||
|
Triple therapy: |
||
|
NEXIUM |
40 mg |
Once Daily for 10 Days |
|
Amoxicillin |
1000 mg |
Twice Daily for 10 Days |
|
Clarithromycin |
500 mg |
Twice Daily for 10 Days |
Please refer to amoxicillin and clarithromycin full prescribing information for CONTRAINDICATIONS, WARNINGS and dosing in elderly and renally-impaired patients.
Special Populations
Geriatric
No dosage adjustment is necessary. (See CLINICAL PHARMACOLOGY, Pharmacokinetics)
Renal Insufficiency
No dosage adjustment is necessary. (See CLINICAL PHARMACOLOGY, Pharmacokinetics)
Hepatic Insufficiency
No dosage adjustment is necessary in patients with mild to moderate liver impairment (Child Pugh Classes A and B). For patients with severe liver impairment (Child Pugh Class C), a dose of 20 mg of NEXIUM should not be exceeded (See CLINICAL PHARMACOLOGY, Pharmacokinetics)
Gender
No dosage adjustment is necessary. (See CLINICAL PHARMACOLOGY, Pharmacokinetics)
HOW SUPPLIED
NEXIUM Delayed-Release Capsules, 20 mg, are opaque, hard gelatin, amethyst colored capsules with two radial bars in yellow on the cap and NEXIUM 20 mg in yellow on the body. They are supplied as follows:
NDC 0186-5020-31 unit of use bottles of 30
NDC 0186-5022-28 unit dose packages of 100
NDC 0186-5020-54 bottles of 90
NDC 0186-5020-82 bottles of 1000
NEXIUM Delayed-Release Capsules, 40 mg, are opaque, hard gelatin, amethyst colored capsules with three radial bars in yellow on the cap and NEXIUM 40 mg in yellow on the body. They are supplied as follows:
NDC 0186-5040-31 unit of use bottles of 30
NDC 0186-5042-28 unit dose packages of 100
NDC 0186-5040-54 bottles of 90
NDC 0186-5040-82 bottles of 1000
Storage
Store at 25°C (77°F); excursions permitted to 15 - 30°C (59 - 86°F). [See USP Controlled Room Temperature]. Keep container tightly closed. Dispense in a tight container if the product package is subdivided.
REFERENCES
1. National Committee for Clinical Laboratory Standards. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. Fifth Edition: Approved Standard NCCLS Document M7-A5, Vol. 20, no. 2, NCCLS, Wayne, PA, January 2000.
NEXIUM and the color purple as applied to the capsule are registered trademarks of the AstraZeneca group of companies.
©AstraZeneca 2004, 2005
Distributed by:
AstraZeneca LP
Wilmington, DE 19850
Product of France
9346611
31026-01
Rev. 08/05
| NEXIUM (Esomeprazole magnesium) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NEXIUM (Esomeprazole magnesium) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NEXIUM (Esomeprazole magnesium) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NEXIUM (Esomeprazole magnesium) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Revised: 05/2006AstraZeneca LP