DIPHENHYDRAMINE HYDROCHLORIDE
-
diphenhydramine hydrochloride injection
McKesson Packaging Services Business Unit of McKesson Corporation
----------
Diphenhydramine Hydrochloride Injection, USPRx only
DESCRIPTION
Diphenhydramine Hydrochloride Injection is a sterile, nonpyrogenic solution for intravenous or deep intramuscular use as an antihistaminic agent. Each mL contains diphenhydramine hydrochloride 50 mg and benzethonium chloride 100 mcg in Water for Injection. pH 4.0-6.5; sodium hydroxide and/or hydrochloric acid added, if needed, for pH adjustment.
The chemical name of diphenhydramine hydrochloride is 2-(Diphenylmethoxy)-N,N-dimethylethylamine hydrochloride. The structural formula is as follows:
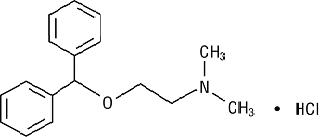
C17H21NO • HCl MW 291.82
Diphenhydramine hydrochloride occurs as a white crystalline powder and is freely soluble in water and alcohol.
CLINICAL PHARMACOLOGY
Diphenhydramine hydrochloride is an antihistamine with anticholinergic (drying) and sedative side effects. Antihistamines appear to compete with histamine for cell receptor sites on effector cells.
Diphenhydramine hydrochloride in the injectable form has a rapid onset of action. Diphenhydramine is widely distributed throughout the body, including the CNS. A portion of the drug is excreted unchanged in the urine, while the rest is metabolized via the liver. Detailed information on the pharmacokinetics of Diphenhydramine Hydrochloride Injection is not available.
INDICATIONS AND USAGE
Diphenhydramine Hydrochloride Injection is effective in adults and pediatric patients, other than premature infants and neonates, for the following conditions when the oral form is impractical:
Antihistaminic
For amelioration of allergic reactions to blood or plasma, in anaphylaxis as an adjunct to epinephrine and other standardmeasures after the acute symptoms have been controlled and for other uncomplicated allergic conditions of the immediate type when oral therapy is impossible or contraindicated.
Motion Sickness
For active treatment of motion sickness.
Antiparkinsonism
For use in parkinsonism, when oral therapy is impossible or contraindicated, as follows: parkinsonism in the elderly who are unable to tolerate more potent agents, mild cases of parkinsonism in other age groups and in other cases of parkinsonism in combination with centrally acting anticholinergic agents.
CONTRAINDICATIONS
Use in Neonates or Premature Infants
This drug should not be used in neonates or premature infants.
Use in Nursing Mothers
Because of the higher risk of antihistamines for infants generally, and for neonates and prematures in particular, antihistamine therapy is contraindicated in nursing mothers.
Use as a Local Anesthetic
Because of the risk of local necrosis, this drug should not be used as a local anesthetic.
Antihistamines are also Contraindicated in the Following Conditions
Hypersensitivity to diphenhydramine hydrochloride and other antihistamines of similar chemical structure.
WARNINGS
Antihistamines should be used with considerable caution in patients with narrow-angle glaucoma, stenosing peptic ulcer, pyloroduodenal obstruction, symptomatic prostatic hypertrophy or bladder-neck obstruction.
Local necrosis has been associated with the use of subcutaneous or intradermal use of intravenous diphenhydramine.
Use in Pediatric Patients
In pediatric patients, especially, antihistamines in overdosage may cause hallucinations, convulsions or death.
As in adults, antihistamines may diminish mental alertness in pediatric patients. In the young pediatric patient, particularly, they may produce excitation.
Use in the Elderly (approximately 60 years or older)
Antihistamines are more likely to cause dizziness, sedation and hypotension in elderly patients.
PRECAUTIONS
General
Diphenhydramine hydrochloride has an atropine-like action and, therefore, should be used with caution in patients with a history of bronchial asthma, increased intraocular pressure, hyperthyroidism, cardiovascular disease or hypertension. Use with caution in patients with lower respiratory disease, including asthma.
Information for Patients
Patients taking diphenhydramine hydrochloride should be advised that this drug may cause drowsiness and has an additive effect with alcohol.
Patients should be warned about engaging in activities requiring mental alertness, such as driving a car or operating appliances, machinery, etc.
Drug Interactions
Diphenhydramine hydrochloride has additive effects with alcohol and other CNS depressants (hypnotics, sedatives, tranquilizers, etc.)
MAO inhibitors prolong and intensify the anticholinergic (drying) effects of antihistamines.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to determine mutagenic and carcinogenic potential have not been performed.
Pregnancy
Teratogenic Effects—Pregnancy Category B
Reproduction studies have been performed in rats and rabbits at doses up to 5 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to diphenhydramine hydrochloride. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Pediatric Use
Diphenhydramine should not be used in neonates and premature infants (see CONTRAINDICATIONS).
Diphenhydramine may diminish mental alertness, or in the young pediatric patient, cause excitation. Overdosage may cause hallucinations, convulsions or death (see WARNINGS and OVERDOSAGE).
See also DOSAGE AND ADMINISTRATION section.
ADVERSE REACTIONS
The most frequent adverse reactions are italicized.
General
Urticaria; drug rash; anaphylactic shock; photosensitivity; excessive perspiration; chills; dryness of mouth, nose and throat.
Cardiovascular System
Hypotension, headache, palpitations, tachycardia, extrasystoles.
Hematologic System
Hemolytic anemia, thrombocytopenia, agranulocytosis.
Nervous System
Sedation, sleepiness, dizziness, disturbed coordination, fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesia, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, neuritis, convulsions.
Gastrointestinal System
Epigastric distress, anorexia, nausea, vomiting, diarrhea, constipation.
Genitourinary System
Urinary frequency, difficult urination, urinary retention, early menses.
Respiratory System
Thickening of bronchial secretions, tightness of chest and wheezing, nasal stuffiness.
OVERDOSAGE
Antihistamine overdosage reactions may vary from central nervous system depression to stimulation. Stimulation is particularly likely in pediatric patients. Atropine-like signs and symptoms, dry mouth; fixed, dilated pupils; flushing, and gastrointestinal symptoms may also occur.
Stimulants should not be used.
Vasopressors may be used to treat hypotension.
DOSAGE AND ADMINISTRATION
THIS PRODUCT IS FOR INTRAVENOUS OR INTRAMUSCULAR ADMINISTRATION ONLY.
Diphenhydramine Hydrochloride Injection is indicated when the oral form is impractical.
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND THE RESPONSE OF THE PATIENT.
Pediatric Patients, Other Than Premature Infants and Neonates
5 mg/kg/24 hours or 150 mg/m2/24 hours. Maximum daily dosage is 300 mg. Divide into four doses, administered intravenously at a rate generally not exceeding 25 mg/min, or deep intramuscularly.
Adults
10 to 50 mg intravenously at a rate generally not exceeding 25 mg/min, or deep intramuscularly; 100 mg if required; maximum daily dosage is 400 mg.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
HOW SUPPLIED
Diphenhydramine Hydrochloride Injection, USP 50 mg/mL in:
Boxes of 1x5 UD 05 NDC 63739-462-05
Storage
Protect from light. Keep covered in carton until time of use. Store at 20˚-25˚C (68˚-77˚F), excursions permitted to 15˚-30˚C (59˚-86˚F) [see USP Controlled Room Temperature].
Baxter and Dosette are trademarks of Baxter International Inc., or its subsidiaries.
For Product Inquiry 1 800 ANA DRUG (1-800-262-3784)
Manufactured by:
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Distributed by:
McKesson Packaging
Concord, NC 28027
MLT-43/1.0
IS-462-M75-01-A
PACKAGE DISPLAY PANEL

LABEL TEXT
NDC 63739-462-05
1 mL DOSETTE Vial
DiphenhydrAMINE
HCl Injection, USP
50 mg / mL
HIGH POTENCY
UD 05 Injections (1x5)
Rx Only
Each mL Contains:
diphenhydramine HCl 50mg and benzethonium chloride 100 mcg in Water for Injection.
PROTECT FROM LIGHT.
Store at 20°-25°C (68° -77°F), excursions permitted to 15°-30°C (59°-86°F)
[See USP Controlled Room Temperature].
Directions for use: See package insert.
Keep this and all medications out of the reach of children.
Mfg. By: Baxter Healthcare Corp. Deerfield, IL 60015
Dist. By McKesson Packaging Concord, NC 28027
LS-462-05-M75-01-A
| DIPHENHYDRAMINE HYDROCHLORIDE
diphenhydramine hydrochloride injection |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA080817 | 02/01/2010 | 04/14/2010 |
| Labeler - McKesson Packaging Services Business Unit of McKesson Corporation (140529962) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Baxter Healthcare Corporation | 005083209 | MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| McKesson Packaging Services Business Unit of McKesson Corporation | 140529962 | REPACK | |