HIZENTRA
-
human immunoglobulin g liquid
CSL Behring AG
----------
|
|||||||||||||||||||||
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Hizentra is an Immune Globulin Subcutaneous (Human) (IGSC), 20% Liquid indicated as replacement therapy for primary humoral immunodeficiency (PI). This includes, but is not limited to, the humoral immune defect in congenital agammaglobulinemia, common variable immunodeficiency, X-linked agammaglobulinemia, Wiskott-Aldrich syndrome, and severe combined immunodeficiencies.
2 DOSAGE AND ADMINISTRATION
For subcutaneous infusion only. Do not inject into a blood vessel.
2.1 Preparation and Handling
Hizentra is a clear and pale yellow to light brown solution. Do not use if the solution is cloudy or contains particulates.
- Prior to administration, visually inspect each vial of Hizentra for particulate matter or discoloration, whenever the solution and container permit.
- Do not freeze. Do not use any solution that has been frozen.
- Check the product expiration date on the vial label. Do not use beyond the expiration date.
- Do not mix Hizentra with other products.
- Do not shake the Hizentra vial.
- Use aseptic technique when preparing and administering Hizentra.
- The Hizentra vial is for single-use only. Discard all used administration supplies and any unused product immediately after each infusion in accordance with local requirements.
2.2 Dosage
The dose should be individualized based on the patient's clinical response to Hizentra therapy and serum immunoglobulin G (IgG) trough levels.
Begin treatment with Hizentra one week after the patient's last Immune Globulin Intravenous (Human) (IGIV) infusion. Prior to switching treatment from IGIV to Hizentra, obtain the patient's serum IgG trough level to guide subsequent dose adjustments (see below under Dose Adjustment).
Establish the initial weekly dose of Hizentra by converting the monthly IGIV dose into a weekly equivalent and increasing it using a dose adjustment factor. The goal is to achieve a systemic serum IgG exposure (area under the concentration-time curve [AUC]) not inferior to that of the previous IGIV treatment (see Pharmacokinetics [12.3]).
Initial Weekly Dose
To calculate the initial weekly dose of Hizentra, multiply the previous IGIV dose in grams by the dose adjustment factor of 1.53; then divide this by the number of weeks between doses during the patient's IGIV treatment (i.e., 3 or 4).
| Initial Hizentra dose = | 1.53 × previous IGIV dose (in grams) |
| Number of weeks between IGIV doses |
To convert the Hizentra dose (in grams) to milliliters (mL), multiply the calculated dose (in grams) by 5.
Dose Adjustment
Over time, the dose may need to be adjusted to achieve the desired clinical response and serum IgG trough level. To determine if a dose adjustment may be considered, measure the patient's serum IgG trough level 2 to 3 months after switching from IGIV to Hizentra. The target serum IgG trough level on weekly Hizentra treatment is projected to be 1.3 ± 0.2 times (i.e., between 1.1 and 1.5 times) the last IGIV trough level (see Pharmacokinetics [12.3]).
To adjust the dose based on trough levels, calculate the difference (in mg/dL) of the patient's serum IgG trough level from the target IgG trough level (1.3 times the last IGIV trough level). Then find this difference in Table 1 and the corresponding amount (in mL) by which to increase or decrease the weekly dose based on the patient's body weight. However, the patient's clinical response should be the primary consideration in dose adjustment.
| Difference From Target IgG Trough Level (mg/dL) | Body Weight (kg) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 10 | 15 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | 110 | 120 | |
| Dose Adjustment (mL per Week)* | |||||||||||||
|
|||||||||||||
| 50 | 0 | 1 | 1 | 1 | 2 | 2 | 3 | 3 | 4 | 4 | 5 | 5 | 6 |
| 100 | 1 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 8 | 9 | 10 | 11 |
| 150 | 1 | 2 | 3 | 4 | 6 | 7 | 8 | 10 | 11 | 13 | 14 | 15 | 17 |
| 200 | 2 | 3 | 4 | 6 | 8 | 9 | 11 | 13 | 15 | 17 | 19 | 21 | 23 |
| 250 | 2 | 4 | 5 | 7 | 9 | 12 | 14 | 16 | 19 | 21 | 23 | 26 | 28 |
| 300 | 3 | 4 | 6 | 8 | 11 | 14 | 17 | 20 | 23 | 25 | 28 | 31 | 34 |
| 350 | 3 | 5 | 7 | 10 | 13 | 16 | 20 | 23 | 26 | 30 | 33 | 36 | 39 |
| 400 | 4 | 6 | 8 | 11 | 15 | 19 | 23 | 26 | 30 | 34 | 38 | 41 | 45 |
| 450 | 4 | 6 | 8 | 13 | 17 | 21 | 25 | 30 | 34 | 38 | 42 | 46 | 51 |
| 500 | 5 | 7 | 9 | 14 | 19 | 23 | 28 | 33 | 38 | 42 | 47 | 52 | 56 |
For example, if a patient with a body weight of 70 kg has an actual IgG trough level of 900 mg/dL and the target trough level is 1000 mg/dL, this results in a difference of 100 mg/dL. Therefore, increase the weekly dose of Hizentra by 7 mL.
Monitor the patient's clinical response, and repeat the dose adjustment as needed.
Dosage requirements for patients switching to Hizentra from another IGSC product have not been studied. If a patient on Hizentra does not maintain an adequate clinical response or a serum IgG trough level equivalent to that of the previous IGSC treatment, the physician may want to adjust the dose. For such patients, Table 1 also provides guidance for dose adjustment if their desired IGSC trough level is known.
Measles Exposure
If a patient is at risk of measles exposure (i.e., due to an outbreak in the US or travel to endemic areas outside of the US), the weekly Hizentra dose should be a minimum of 200 mg/kg body weight for two consecutive weeks. If a patient has been exposed to measles, ensure this minimum dose is administered as soon as possible after exposure.
2.3 Administration
Hizentra is for subcutaneous infusion only. Do not inject into a blood vessel.
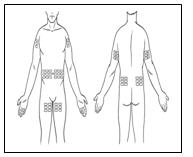
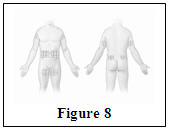
Hizentra is intended for weekly subcutaneous administration using an infusion pump. Infuse Hizentra in the abdomen, thigh, upper arm, and/or lateral hip.
- Injection sites – A Hizentra dose may be infused into multiple injection sites. However, do not use more than 4 sites simultaneously. Injection sites should be at least 2 inches apart. Change the actual site of injection with each weekly administration.
- Volume – For the first infusion of Hizentra, do not exceed a volume of 15 mL per injection site. The volume may be increased to 20 mL per site after the fourth infusion and to a maximum of 25 mL per site as tolerated.
- Rate – For the first infusion of Hizentra, the maximum recommended flow rate is 15 mL per hour per site. For subsequent infusions, the flow rate may be increased to a maximum of 25 mL per hour per site as tolerated. However, the maximum flow rate is not to exceed a total of 50 mL per hour for all sites combined at any time.
Follow the steps below and use aseptic technique to administer Hizentra.
|
|
|
|
|
|
|  |
| |
|  |
|  |
|  |
|  |
| |
| |
| |
After administration, immediately discard any unused product and all used disposable supplies in accordance with local requirements.
For self-administration, provide the patient with instructions and training for subcutaneous infusion in the home or other appropriate setting.
3 DOSAGE FORMS AND STRENGTHS
Hizentra is a 0.2 g/mL (20%) protein solution for subcutaneous injection.
4 CONTRAINDICATIONS
Hizentra is contraindicated in patients who have had an anaphylactic or severe systemic reaction to the administration of human immune globulin or to components of Hizentra, such as polysorbate 80.
Hizentra is contraindicated in patients with hyperprolinemia because it contains the stabilizer L-proline (see Description [11]).
Hizentra is contraindicated in IgA-deficient patients with antibodies against IgA and a history of hypersensitivity (see Description [11]).
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Severe hypersensitivity reactions may occur to human immune globulin or components of Hizentra, such as polysorbate 80. In case of hypersensitivity, discontinue the Hizentra infusion immediately and institute appropriate treatment.
Individuals with IgA deficiency can develop anti-IgA antibodies and anaphylactic reactions (including anaphylaxis and shock) after administration of blood components containing IgA. Patients with known antibodies to IgA may have a greater risk of developing potentially severe hypersensitivity and anaphylactic reactions with administration of Hizentra. Hizentra contains ≤50 mcg/mL IgA (see Description [11]).
5.2 Reactions Reported to Occur With IGIV Treatment
The following reactions have been reported to occur with IGIV treatment and may occur with IGSC treatment.
Renal Dysfunction/Failure
Renal dysfunction/failure, osmotic nephropathy, and death may occur with use of human immune globulin products. Ensure that patients are not volume depleted and assess renal function, including measurement of blood urea nitrogen (BUN) and serum creatinine, before the initial infusion of Hizentra and at appropriate intervals thereafter.
Periodic monitoring of renal function and urine output is particularly important in patients judged to have a potential increased risk of developing acute renal failure.1 If renal function deteriorates, consider discontinuing Hizentra. For patients judged to be at risk of developing renal dysfunction because of pre-existing renal insufficiency or predisposition to acute renal failure (such as those with diabetes mellitus or hypovolemia, those who are overweight or use concomitant nephrotoxic medicinal products, or those who are over 65 years of age), administer Hizentra at the minimum rate practicable.
Thrombotic Events
Thrombotic events may occur with use of human immune globulin products2-4. Patients at increased risk may include those with a history of atherosclerosis, multiple cardiovascular risk factors, advanced age, impaired cardiac output, hypercoagulable disorders, prolonged periods of immobilization, and/or known or suspected hyperviscosity. Because of the potentially increased risk of thrombosis, consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies. For patients judged to be at risk of developing thrombotic events, administer Hizentra at the minimum rate practicable.
Aseptic Meningitis Syndrome (AMS)
AMS may occur with use of human immune globulin products.5 The syndrome usually begins within several hours to 2 days following IGIV treatment. AMS is characterized by signs and symptoms including severe headache, nuchal rigidity, drowsiness, fever, photophobia, painful eye movements, nausea, and vomiting. Cerebrospinal fluid (CSF) studies frequently show pleocytosis up to several thousand cells per cubic millimeter, predominantly from the granulocytic series, with elevated protein levels up to several hundred mg/dL. AMS may occur more frequently in association with high doses (2 g/kg) and/or rapid infusion of IGIV.
Conduct a thorough neurological examination, including CSF studies, to rule out other causes of meningitis in patients exhibiting signs and symptoms of AMS. Discontinuation of IGIV treatment has resulted in remission of AMS within several days without sequelae.
Hemolysis
Hizentra can contain blood group antibodies that may act as hemolysins and induce in vivo coating of red blood cells (RBCs) with immunoglobulin, causing a positive direct antiglobulin (Coombs') test result and hemolysis.6-8 Delayed hemolytic anemia can develop subsequent to immune globulin therapy due to enhanced RBC sequestration, and acute hemolysis, consistent with intravascular hemolysis, has been reported.9
Monitor recipients of Hizentra for clinical signs and symptoms of hemolysis. If these are present after a Hizentra infusion, perform appropriate confirmatory laboratory testing. If transfusion is indicated for patients who develop hemolysis with clinically compromising anemia after receiving Hizentra, perform adequate cross-matching to avoid exacerbating on-going hemolysis.
Transfusion-Related Acute Lung Injury (TRALI)
Noncardiogenic pulmonary edema may occur in patients administered human immune globulin products.10 TRALI is characterized by severe respiratory distress, pulmonary edema, hypoxemia, normal left ventricular function, and fever. Typically, it occurs within 1 to 6 hours following transfusion. Patients with TRALI may be managed using oxygen therapy with adequate ventilatory support.
Monitor Hizentra recipients for pulmonary adverse reactions. If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil antibodies in both the product and patient's serum.
5.3 Transmissible Infectious Agents
Because Hizentra is made from human plasma, it may carry a risk of transmitting infectious agents (e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease [CJD] agent). The risk of infectious agent transmission has been reduced by screening plasma donors for prior exposure to certain viruses, testing for the presence of certain current virus infections, and including virus inactivation/removal steps in the manufacturing process for Hizentra.
Report all infections thought to be possibly transmitted by Hizentra to CSL Behring Pharmacovigilance at 1-866-915-6958.
5.4 Laboratory Tests
Various passively transferred antibodies in immunoglobulin preparations may lead to misinterpretation of the results of serological testing.
6 ADVERSE REACTIONS
The most common adverse reactions (ARs), observed in ≥5% of study subjects receiving Hizentra, were local reactions (i.e., swelling, redness, heat, pain, and itching at the injection site), headache, vomiting, pain, and fatigue.
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, AR rates observed in clinical studies of a product cannot be directly compared to rates in the clinical studies of another product and may not reflect the rates observed in clinical practice.
The safety of Hizentra was evaluated in a clinical study for 15 months in subjects with PI who had been treated previously with IGIV every 3 or 4 weeks. The safety analyses included 49 subjects in the intention-to-treat (ITT) population. The ITT population consisted of all subjects who received at least one dose of Hizentra (see Clinical Studies [14]).
Subjects were treated with Hizentra at weekly doses ranging from 66 to 331 mg/kg body weight during the wash-in/wash-out period and from 72 to 379 mg/kg during the efficacy period. The 49 subjects received a total of 2264 weekly infusions of Hizentra.
No deaths or serious ARs occurred during the study. Two subjects withdrew from the study due to ARs. One subject experienced a severe injection-site reaction one day after the third weekly infusion, and the other subject experienced moderate myositis. Both reactions were judged to be “at least possibly related” to the administration of Hizentra.
Table 2 summarizes the most frequent adverse events (AEs) (experienced by at least 4 subjects), irrespective of causality. Included are all AEs and those considered temporally associated with the Hizentra infusion, i.e., occurring during or within 72 hours after the end of an infusion. Local reactions were the most frequent AEs observed, with injection-site reactions (i.e., swelling, redness, heat, pain, and itching at the site of injection) comprising 98% of local reactions.
| Table 2: Incidence of Subjects With Adverse Events (AEs)* (Experienced by 4 or More Subjects) and Rate per Infusion, Irrespective of Causality (ITT Population) | ||||
|---|---|---|---|---|
| All AEs* | AEs* Occurring During or Within 72 Hours of Infusion | |||
| AE (≥4 Subjects) | Number (%) of Subjects (n=49) | Number (Rate†) of AEs (n=2264 Infusions) | Number (%) of Subjects (n=49) | Number (Rate†) of AEs (n=2264 Infusions) |
| Local reactions‡ | 49 (100) | 1340 (0.592) | 49 (100) | 1322 (0.584) |
| Other AEs: | ||||
| Headache | 13 (26.5) | 40 (0.018) | 12 (24.5) | 32 (0.014) |
| Cough | 8 (16.3) | 9 (0.004) | 5 (10.2) | 6 (0.003) |
| Diarrhea | 7 (14.3) | 8 (0.004) | 5 (10.2) | 6 (0.003) |
| Fatigue | 6 (12.2) | 6 (0.003) | 4 (8.2) | 4 (0.002) |
| Back pain | 5 (10.2) | 11 (0.005) | 4 (8.2) | 5 (0.002) |
| Nausea | 5 (10.2) | 5 (0.002) | 4 (8.2) | 4 (0.002) |
| Abdominal pain, upper | 5 (10.2) | 5 (0.002) | 3 (6.1) | 3 (0.001) |
| Rash | 5 (10.2) | 7 (0.003) | 2 (4.1) | 3 (0.001) |
| Pain in extremity | 4 (8.2) | 7 (0.003) | 4 (8.2) | 6 (0.003) |
| Migraine | 4 (8.2) | 5 (0.002) | 3 (6.1) | 4 (0.002) |
| Pain | 4 (8.2) | 5 (0.002) | 3 (6.1) | 4 (0.002) |
| Epistaxis | 4 (8.2) | 6 (0.003) | 2 (4.1) | 3 (0.001) |
| Pharyngolaryngeal pain | 4 (8.2) | 6 (0.003) | 2 (4.1) | 2 (<0.001) |
| Arthralgia | 4 (8.2) | 5 (0.002) | 2 (4.1) | 3 (0.001) |
The ratio of infusions with temporally associated AEs, including local reactions, to all infusions was 1338 to 2264 (59.1%; upper 95% confidence limit of 62.4%). Excluding local reactions, the corresponding ratio was 173 to 2264 (7.6%; upper 95% confidence limit of 8.9%).
Table 3 summarizes the most frequent ARs (i.e., those AEs considered by the investigators to be "at least possibly related" to Hizentra administration) experienced by at least 2 subjects.
| Adverse Reaction (≥2 Subjects) | Number (%) of Subjects (n=49) | Number (Rate*) of Adverse Reactions (n=2264 Infusions) |
|---|---|---|
| Local reactions† | 49 (100) | 1338 (0.591) |
| Other AEs: | ||
| Headache | 12 (24.5) | 36 (0.016) |
| Vomiting | 3 (6.1) | 3 (0.001) |
| Pain | 3 (6.1) | 4 (0.002) |
| Fatigue | 3 (6.1) | 3 (0.001) |
| Contusion | 2 (4.1) | 3 (0.001) |
| Back pain | 2 (4.1) | 3 (0.001) |
| Migraine | 2 (4.1) | 3 (0.001) |
| Diarrhea | 2 (4.1) | 2 (<0.001) |
| Abdominal pain, upper | 2 (4.1) | 2 (<0.001) |
| Nausea | 2 (4.1) | 2 (<0.001) |
| Rash | 2 (4.1) | 2 (<0.001) |
| Arthralgia | 2 (4.1) | 2 (<0.001) |
Table 4 summarizes injection-site reactions based on investigator assessments 15 to 45 minutes after the end of the 683 infusions administered during regularly scheduled visits (every 4 weeks).
| Injection-Site Reaction | Number† (Rate‡) of Reactions (n=683 Infusions§) |
|---|---|
|
|
| Edema/induration | 467 (0.68) |
| Erythema | 346 (0.50) |
| Local heat | 108 (0.16) |
| Local pain | 88 (0.13) |
| Itching | 64 (0.09) |
Most local reactions were either mild (93.4%) or moderate (6.3%) in intensity.
6.2 Postmarketing Experience
Because postmarketing reporting of adverse reactions is voluntary and from a population of uncertain size, it is not always possible to reliably estimate the frequency of these reactions or establish a causal relationship to product exposure.
The following adverse reactions have been identified and reported during the postmarketing use of IGIV products11:
- Infusion reactions: Hypersensitivity (e.g., anaphylaxis), headache, diarrhea, tachycardia, fever, fatigue, dizziness, malaise, chills, flushing, urticaria or other skin reactions, wheezing or other chest discomfort, nausea, vomiting, rigors, back pain, myalgia, arthralgia, and changes in blood pressure
- Renal: Acute renal dysfunction/failure, osmotic nephropathy
- Respiratory: Apnea, Acute Respiratory Distress Syndrome (ARDS), TRALI, cyanosis, hypoxemia, pulmonary edema, dyspnea, bronchospasm
- Cardiovascular: Cardiac arrest, thromboembolism, vascular collapse, hypotension
- Neurological: Coma, loss of consciousness, seizures, tremor, aseptic meningitis syndrome
- Integumentary: Stevens-Johnson syndrome, epidermolysis, erythema multiforme, dermatitis (e.g., bullous dermatitis)
- Hematologic: Pancytopenia, leukopenia, hemolysis, positive direct antiglobulin (Coombs') test
- Gastrointestinal: Hepatic dysfunction, abdominal pain
- General/Body as a Whole: Pyrexia, rigors
To report SUSPECTED ADVERSE REACTIONS, contact CSL Behring Pharmacovigilance at 1-866-915-6958 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 DRUG INTERACTIONS
7.1 Live Virus Vaccines
The passive transfer of antibodies with immunoglobulin administration may interfere with the response to live virus vaccines such as measles, mumps, rubella, and varicella (see Patient Counseling Information [17]).
7.2 Serological Testing
Various passively transferred antibodies in immunoglobulin preparations may lead to misinterpretation of the results of serological testing.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C. Animal reproduction studies have not been conducted with Hizentra. It is not known whether Hizentra can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Hizentra should be given to pregnant women only if clearly needed.
8.3 Nursing Mothers
Hizentra has not been evaluated in nursing mothers.
8.4 Pediatric Use
Hizentra was evaluated in 10 pediatric subjects (3 children and 7 adolescents) with PI. No pediatric-specific dose requirements were necessary to achieve the desired serum IgG levels.
Hizentra was not evaluated in neonates or infants.
8.5 Geriatric Use
Of the 49 subjects evaluated in the clinical study of Hizentra, 6 subjects were 65 years of age or older. No overall differences in safety or efficacy were observed between these subjects and younger subjects.
11 DESCRIPTION
Hizentra, Immune Globulin Subcutaneous (Human), 20% Liquid, is a ready-to-use, sterile 20% (0.2 g/mL) protein liquid preparation of polyvalent human immunoglobulin G (IgG) for subcutaneous administration. Hizentra is manufactured from large pools of human plasma by a combination of cold alcohol fractionation, octanoic acid fractionation, and anion exchange chromatography. The IgG proteins are not subjected to heating or to chemical or enzymatic modification. The Fc and Fab functions of the IgG molecule are retained. Fab functions tested include antigen binding capacities, and Fc functions tested include complement activation and Fc-receptor-mediated leukocyte activation (determined with complexed IgG).
Hizentra has a purity of ≥98% IgG and a pH of 4.6 to 5.2. Hizentra contains approximately 250 (range: 210 to 290 mmol/L) L-proline (a nonessential amino acid) as a stabilizer, 10 to 30 mg/L polysorbate 80, and trace amounts of sodium. Hizentra contains ≤50 mcg/mL IgA. Hizentra contains no carbohydrate stabilizers (e.g., sucrose, maltose) and no preservative.
Plasma units used in the manufacture of Hizentra are tested using FDA-licensed serological assays for hepatitis B surface antigen and antibodies to human immunodeficiency virus (HIV)-1/2 and hepatitis C virus (HCV) as well as FDA-licensed Nucleic Acid Testing (NAT) for HIV-1 and HCV. All plasma units have been found to be nonreactive (negative) in these tests. For hepatitis B virus (HBV), an investigational NAT procedure is used and the plasma units found to be negative; however, the significance of a negative result has not been established. In addition, the plasma has been tested for B19 virus (B19V) DNA by NAT. Only plasma that passes virus screening is used for production, and the limit for B19V in the fractionation pool is set not to exceed 104 IU of B19V DNA per mL.
The manufacturing process for Hizentra includes three steps to reduce the risk of virus transmission. Two of these are dedicated virus clearance steps: pH 4 incubation to inactivate enveloped viruses and virus filtration to remove, by size exclusion, both enveloped and non-enveloped viruses as small as approximately 20 nanometers. In addition, a depth filtration step contributes to the virus reduction capacity.12
These steps have been independently validated in a series of in vitro experiments for their capacity to inactivate and/or remove both enveloped and non-enveloped viruses. Table 5 shows the virus clearance during the manufacturing process for Hizentra, expressed as the mean log10 reduction factor (LRF).
| HIV-1 | PRV | BVDV | WNV | EMCV | MVM | |
|---|---|---|---|---|---|---|
| HIV-1, human immunodeficiency virus type 1, a model for HIV-1 and HIV-2; PRV, pseudorabies virus, a nonspecific model for large enveloped DNA viruses (e.g., herpes virus); BVDV, bovine viral diarrhea virus, a model for hepatitis C virus; WNV, West Nile virus; EMCV, encephalomyocarditis virus, a model for hepatitis A virus; MVM, minute virus of mice, a model for a small highly resistant non-enveloped DNA virus (e.g., parvovirus); LRF, log10 reduction factor; nt, not tested; na, not applicable. | ||||||
|
||||||
| Virus Property | ||||||
| Genome | RNA | DNA | RNA | RNA | RNA | DNA |
| Envelope | Yes | Yes | Yes | Yes | No | No |
| Size (nm) | 80-100 | 120-200 | 50-70 | 50-70 | 25-30 | 18-24 |
| Manufacturing Step | Mean LRF | |||||
| pH 4 incubation | ≥5.4 | ≥5.9 | 4.6 | ≥7.8 | nt | nt |
| Depth filtration | ≥5.3 | ≥6.3 | 2.1 | 3.0 | 4.2 | 2.3 |
| Virus filtration | ≥5.3 | ≥5.5 | ≥5.1 | ≥5.9 | ≥5.4 | ≥5.5 |
| Overall Reduction (Log10 Units) | ≥16.0 | ≥17.7 | ≥11.8 | ≥16.7 | ≥9.6 | ≥7.8 |
The manufacturing process was also investigated for its capacity to decrease the infectivity of an experimental agent of transmissible spongiform encephalopathy (TSE), considered a model for CJD and its variant (vCJD).12 Several of the production steps have been shown to decrease infectivity of an experimental TSE model agent. TSE reduction steps include octanoic acid fractionation (≥6.4 log10), depth filtration (2.6 log10), and virus filtration (≥5.8 log10). These studies provide reasonable assurance that low levels of vCJD/CJD agent infectivity, if present in the starting material, would be removed.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Hizentra supplies a broad spectrum of opsonizing and neutralizing IgG antibodies against a wide variety of bacterial and viral agents. The mechanism of action in PI has not been fully elucidated.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of Hizentra was evaluated in a PK substudy of subjects with PI participating in the 15-month efficacy and safety study (see Clinical Studies [14]). All PK subjects were treated previously with Privigen®, Immune Globulin Intravenous (Human), 10% Liquid and were switched to weekly subcutaneous treatment with Hizentra. After a 3-month wash-in/wash-out period, doses were adjusted individually with the goal of providing a systemic serum IgG exposure (area under the IgG serum concentration vs time curve; AUC) not inferior to that of the previous weekly-equivalent IGIV dose. Table 6 summarizes PK parameters for subjects in the substudy following treatment with Hizentra and IGIV.
| Hizentra | IGIV* (Privigen®) | |
|---|---|---|
| bw, body weight. | ||
| Number of subjects | 18 | 18 |
| Dose* | ||
| Mean | 228 mg/kg bw | 152 mg/kg bw |
| Range | 141-381 mg/kg bw | 86-254 mg/kg bw |
| IgG peak levels | ||
| Mean | 1616 mg/dL | 2564 mg/dL |
| Range | 1090-2825 mg/dL | 2046-3456 mg/dL |
| IgG trough levels | ||
| Mean | 1448 mg/dL | 1127 mg/dL |
| Range | 952-2623 mg/dL | 702-1810 mg/dL |
| AUC† | ||
| Mean | 10560 day x mg/dL | 10320 day x mg/dL |
| Range | 7210-18670 day x mg/dL | 8051-15530 day x mg/dL |
For the 19 subjects completing the wash-in/wash-out period, the average dose adjustment for Hizentra was 153% (range: 126% to 187%) of the previous weekly-equivalent IGIV dose. After 12 weeks of treatment with Hizentra at this individually adjusted dose, the final steady-state AUC determinations were made in 18 of the 19 subjects. The geometric mean ratio of the steady-state AUCs, standardized to a weekly treatment period, for Hizentra vs IGIV treatment was 1.002 (range: 0.77 to 1.20) with a 90% confidence limit of 0.951 to 1.055 for the 18 subjects.
The PK study included an additional assessment to determine the ratio of serum IgG trough levels with Hizentra (IGSC) compared to the previous trough levels with IGIV that were associated with matching AUCs. It was demonstrated that IgG trough levels during treatment with Hizentra were 1.3 times higher than the preceding trough levels during treatment with IGIV (Privigen®). This calculated IGSC:IGIV ratio of 1.3 (±15% of this value, or ±0.2) can be used to assess dosing with Hizentra by providing a steady-state target IgG trough level, which may be assumed to be within the range of 1.1 to 1.5 times the previous steady-state trough levels with IGIV. However, the patient's clinical response should be the primary consideration in dose adjustment (see Dosage and Administration [2.2]).
With Hizentra, peak serum levels are lower (1616 vs 2564 mg/dL) than those achieved with IGIV while trough levels are generally higher (1448 vs 1127 mg/dL). In contrast to IGIV administered every 3 to 4 weeks, weekly subcutaneous administration results in relatively stable steady-state serum IgG levels.13,14 After the subjects had reached steady-state with weekly administration of Hizentra, peak serum IgG levels were observed after a mean of 2.9 days (range: 0 to 7 days) in 18 subjects.
13 NONCLINICAL TOXICOLOGY
13.2 Animal Toxicology and/or Pharmacology
Long- and short-term memory loss was seen in juvenile rats in a study modeling hyperprolinemia. In this study, rats received daily subcutaneous injections with L-proline from day 6 to day 28 of life.15 The daily amounts of L-proline used in this study were more than 60 times higher than the L-proline dose that would result from the administration of 400 mg/kg body weight of Hizentra once weekly. In unpublished studies using the same animal model (i.e., rats) dosed with the same amount of L-proline with a dosing interval relevant to IGSC treatment (i.e., on 5 consecutive days on days 9 to 13, or once weekly on days 9, 16, and 23), no effects on learning and memory were observed. The clinical relevance of these studies is not known.
14 CLINICAL STUDIES
A prospective, open-label, multicenter, single-arm, clinical study conducted in the US evaluated the efficacy, tolerability, and safety of Hizentra in adult and pediatric subjects with PI. Subjects previously receiving monthly treatment with IGIV were switched to weekly subcutaneous administration of Hizentra for 15 months (a 3-month wash-in/wash-out period followed by a 12-month efficacy period). The efficacy analyses included 38 subjects in the modified intention-to-treat (MITT) population. The MITT population consisted of subjects who completed the wash-in/wash-out period and received at least one infusion of Hizentra during the efficacy period.
Although 5% of the administered doses could not be verified, the weekly doses of Hizentra ranged from 72 to 379 mg per kg body weight, which was 149% (range: 114% to 180%) of the previous IGIV dose. Subjects received a total of 2264 infusions of Hizentra.
In the study, the number of injection sites per infusion ranged from 1 to 12. In 73% of infusions; the number of injection sites was 4 or fewer. Up to 4 simultaneous injection sites were permitted using 2 pumps; however, more than 4 sites could be used consecutively during one infusion. The infusion flow rate did not exceed 50 mL per hour for all injection sites combined. During the efficacy period, the median duration of a weekly infusion ranged from 1.6 to 2.0 hours.
The study evaluated the annual rate of serious bacterial infections (SBIs), defined as bacterial pneumonia, bacteremia/septicemia, osteomyelitis/septic arthritis, bacterial meningitis, and visceral abscess. The study also evaluated the annual rate of any infections, the use of antibiotics for infection (prophylaxis or treatment), the days out of work/school/kindergarten/day care or unable to perform normal activities due to infections, and hospitalizations due to infections.
Table 7 summarizes the efficacy results for subjects in the efficacy phase (MITT population) of the study. No subjects experienced an SBI in this study.
| Number of subjects (efficacy phase) | 38 |
| Total number of subject days | 12,697 |
| Infections | |
| Annual rate of SBIs* | 0 SBIs per subject year† |
| Annual rate of any infections | 2.76 infections/subject year‡ |
| Antibiotic use for infection (prophylaxis or treatment) | |
| Number of subjects (%) | 27 (71.1) |
| Annual rate | 48.5 days/subject year |
| Total number of subject days | 12,605 |
| Days out of work/school/kindergarten/day care or unable to perform normal activities due to infections | |
| Number of days (%) | 71 (0.56) |
| Annual rate | 2.06 days/subject year |
| Hospitalizations due to infections | |
| Number of days (%) | 7 (0.06)§ |
| Annual rate | 0.2 days/subject year |
15 REFERENCES
- Cayco AV, Perazella MA, Hayslett JP. Renal insufficiency after intravenous immune globulin therapy: a report of two cases and an analysis of the literature. J Am Soc Nephrol 1997;8:1788-1793.
- Dalakas MC. High-dose intravenous immunoglobulin and serum viscosity: risk of precipitating thromboembolic events. Neurology 1994;44:223-226.
- Woodruff RK, Grigg AP, Firkin FC, Smith IL. Fatal thrombotic events during treatment of autoimmune thrombocytopenia with intravenous immunoglobulin in elderly patients. Lancet 1986;2:217-218.
- Wolberg AS, Kon RH, Monroe DM, Hoffman M. Coagulation factor XI is a contaminant in intravenous immunoglobulin preparations. Am J Hematol 2000;65:30-34.
- Gabor EP, Meningitis and skin reaction after intravenous immune globulin therapy. Ann Intern Med 1997;127:1130.
- Copelan EA, Strohm PL, Kennedy MS, Tutschka PJ. Hemolysis following intravenous immune globulin therapy. Transfusion 1986;26:410-412.
- Thomas MJ, Misbah SA, Chapel HM, Jones M, Elrington G, Newsom-Davis J. Hemolysis after high-dose intravenous Ig. Blood 1993;15:3789.
- Wilson JR, Bhoopalam N, Fisher M. Hemolytic anemia associated with intravenous immunoglobulin. Muscle Nerve 1997;20:1142-1145.
- Kessary-Shoham H, Levy Y, Shoenfeld Y, Lorber M, Gershon H. In vivo administration of intravenous immunoglobulin (IVIg) can lead to enhanced erythrocyte sequestration. J Autoimmun 1999;13:129-135.
- Rizk A, Gorson KC, Kenney L, Weinstein R. Transfusion-related acute lung injury after the infusion of IVIG. Transfusion 2001;41:264-268.
- Pierce LR, Jain N. Risks associated with the use of intravenous immunoglobulin. Trans Med Rev 2003;17:241-251.
- Stucki M, Boschetti N, Schäfer W, et al. Investigations of prion and virus safety of a new liquid IVIG product. Biologicals 2008;36:239-247.
- Smith GN, Griffiths B, Mollison D, Mollison PL. Uptake of IgG after intramuscular and subcutaneous injection. Lancet 1972;1:1208-1212.
- Waniewski I, Gardulf A, Hammarström L. Bioavailability of γ-globulin after subcutaneous infusions in patients with common variable immunodeficiency. J Clin Immunol 1994;14:90-97.
- Bavaresco CS, Streck EL, Netto CA, et al. Chronic hyperprolinemia provokes a memory deficit in the Morris Water Maze Task. Metabolic Brain Disease 2005;20:73-80.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Hizentra is supplied in a single-use, tamper-evident vial containing 0.2 grams of protein per mL of preservative-free liquid. Each vial label contains a peel-off strip with the vial size and product lot number for use in recording doses in a patient treatment record.
The components used in the packaging for Hizentra contain no latex.
The following dosage presentations are available:
| NDC Number | Fill Size (mL) | Grams Protein |
|---|---|---|
| 44206-451-01 | 5 mL | 1 |
| 44206-452-02 | 10 mL | 2 |
| 44206-454-04 | 20 mL | 4 |
16.2 Storage and Handling
When stored at room temperature (up to 25°C [77°F]), Hizentra is stable for the period indicated by the expiration date printed on the outer carton and vial label. DO NOT FREEZE. Do not use product that has been frozen. Do not shake. Keep Hizentra in its original carton to protect it from light.
17 PATIENT COUNSELING INFORMATION
-
Self-administration – If self-administration is appropriate, ensure that the patient receives instructions and training on subcutaneous administration in the home or other appropriate setting and has demonstrated the ability to perform subcutaneous infusions.
- Ensure patients understand the importance of adhering to the weekly administration schedule to maintain the steady levels of IgG in their blood.
- Instruct patients to keep their treatment diary/log book current by recording, after each infusion, the time, date, dose, and any reactions, and by removing the peel-off portion of the label (containing the lot number) from the product vial and placing it in the treatment diary/log book.
- Tell patients that mild to moderate local (injection-site) reactions (e.g., swelling and redness) are a common side effect of subcutaneous therapy, but to contact their healthcare professional if a local reaction persists for more than a few days.
- Inform patients of the importance of having an infusion needle long enough to reach the subcutaneous tissue and of changing the actual site of injection with each infusion.
- Inform patients to consider adjusting the injection-site location, volume per site, and rate of infusion based on how infusions are tolerated.
- Dose adjustments – Inform patients that they should be tested regularly to make sure they have the correct levels of Hizentra (IgG) in their blood. These tests may result in adjustments to the Hizentra dose.
- Hypersensitivity – Inform patients of the early signs of hypersensitivity reactions to Hizentra (including hives, generalized urticaria, tightness of the chest, wheezing, hypotension, and anaphylaxis), and advise them to notify their physician if they experience any of these symptoms.
- Interference With Vaccines – Inform patients that administration of IgG may interfere with the response to live virus vaccines (e.g., measles, mumps, rubella, and varicella) and to notify their immunizing physician of recent therapy with Hizentra.
-
Reactions reported to occur with IGIV treatment – Advise patients to be aware of and immediately report to their physician symptoms of the following potential reactions:
- Decreased urine output, sudden weight gain, fluid retention/edema, and/or shortness of breath, which may suggest kidney problems
- Shortness of breath, changes in mental status, chest pain, and other manifestations of thrombotic and embolic events
- Severe headache, neck stiffness, drowsiness, fever, sensitivity to light, painful eye movements, nausea, and vomiting, which may suggest aseptic meningitis syndrome
- Fatigue, increased heart rate, yellowing of the skin or eyes, and dark-colored urine, which may suggest hemolysis
- Severe breathing problems, lightheadedness, drops in blood pressure, and fever, which may suggest TRALI (a condition typically occurring within 1 to 6 hours following transfusion)
- Transmissible infectious agents – Inform patients that Hizentra is made from human plasma (part of the blood) and may contain infectious agents that can cause disease (e.g., viruses and, theoretically, the CJD agent). Explain that the risk that Hizentra may transmit an infectious agent has been reduced by screening the plasma donors, by testing the donated plasma for certain virus infections, and by inactivating and/or removing certain viruses during manufacturing.
The attached Hizentra "Information for Patients" contains more detailed instructions for patients who will be self-administering Hizentra.
Hizentra
Immune Globulin Subcutaneous (Human), 20% Liquid
Information for Patients
This patient package insert summarizes important information about Hizentra. Please read it carefully before using this medicine. This information does not take the place of talking with your healthcare professional, and it does not include all of the important information about Hizentra. If you have any questions after reading this, ask your healthcare professional.
What is the most important information I should know about Hizentra?
Hizentra is supposed to be infused under your skin only. DO NOT inject Hizentra into a blood vessel (vein or artery).
What is Hizentra?
Hizentra (Hi – ZEN – tra) is a prescription medicine used to treat primary immune deficiency (PI). Hizentra is made from human plasma. It contains antibodies, called immunoglobulin G (IgG), that healthy people have to fight germs (bacteria and viruses).
People with PI get a lot of infections. Hizentra helps lower the number of infections you will get.
Who should NOT take Hizentra?
Do not take Hizentra if you have too much proline in your blood (called "hyperprolinemia") or if you have had reactions to polysorbate 80.
Tell your doctor if you have had a serious reaction to other immune globulin medicines or if you have been told that you also have a deficiency of the immunoglobulin called IgA.
How should I take Hizentra?
You will take Hizentra through an infusion under your skin. You will use up to 4 needles that are put into different places of your body at one time. The needles are attached to a pump with an infusion tube. It usually takes about 60 minutes to do one infusion. You will need to have infusions once a week.
Instructions for using Hizentra are at the end of this patient package insert (see "How do I use Hizentra?"). Do not use Hizentra by yourself until you have been taught how by your doctor or healthcare professional.
What should I avoid while taking Hizentra?
Vaccines may not work well for you while you are taking Hizentra. Tell your doctor or healthcare professional that you are taking Hizentra before you get a vaccine.
Tell your doctor or healthcare professional if you are pregnant or plan to become pregnant, or if you are nursing.
What are possible side effects of Hizentra?
The most common side effects with Hizentra are:
- Redness, swelling, and itching at the injection site
- Headache/migraine
- Vomiting
- Pain (including pain in the back, joints, arms, legs)
- Fatigue
- Bruising
- Diarrhea
- Stomach ache
- Nausea
- Rash
Tell your doctor right away or go to the emergency room if you have hives, trouble breathing, wheezing, dizziness, or fainting. These could be signs of a bad allergic reaction.
Tell your doctor right away if you have any of the following symptoms. They could be signs of a serious problem.
- Reduced urination, sudden weight gain, or swelling in your legs. These could be signs of a kidney problem.
- Pain, swelling, warmth, redness, or a lump in your legs or arms. These could be signs of a blood clot.
- Bad headache with nausea, vomiting, stiff neck, fever, and sensitivity to light. These could be signs of a brain swelling called meningitis.
- Brown or red urine, fast heart rate, yellow skin or eyes. These could be signs of a blood problem.
- Chest pains or trouble breathing.
- Fever over 100ºF. This could be a sign of an infection.
Tell your doctor about any side effects that concern you. You can ask your doctor to give you more information that is available to healthcare professionals.
Infuse Hizentra only after you have been trained by your doctor or healthcare professional. Below are step-by-step instructions to help you remember how to use Hizentra. Ask your doctor or healthcare professional about any instructions you do not understand.
Instructions for use
Hizentra comes in single-use vials.
Keep Hizentra in the storage box at room temperature.
Step 1: Gather the Hizentra vials, the following disposable supplies (not provided with Hizentra), and other items (infusion pump, sharps container, treatment diary or logbook):
|
|
Step 2: Wash hands
|  |
| Step 3: Clean surface
Thoroughly clean a table or other flat surface using one of the alcohol wipes. | |
| Step 4: Check vials
Carefully look at the liquid in each vial of Hizentra (Figure 2). It should look clear and be pale yellow to light brown. Check for particles or color changes. Do not use the vial if:
|  |
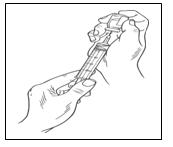
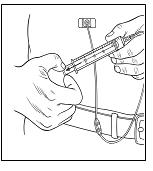
Step 5: Transfer Hizentra to syringe for infusion
|  |
|  |
|  |
| 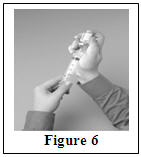 |
| Step 6: Prepare pump and infusion tubing
Prepare the infusion pump (following the manufacturer's instructions) and prime (fill) the infusion tubing. To prime the tubing, connect the syringe filled with Hizentra to the infusion tubing and gently push on the syringe plunger to fill the tubing with Hizentra (Figure 7). |  |
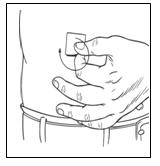
Step 7: Prepare injection site(s)
|   |
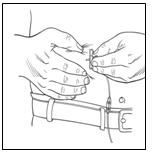
Step 8: Insert needle(s)
|  |
|  |
|  |
| Step 9: Start infusion
Follow the instructions on the infusion pump (Figure 13) to connect the infusion tubing and set the infusion rate. (Your doctor will tell you what rate to use for your Hizentra infusion.) Turn on the pump. |  |
| Step 10: Record treatment (Figure 14) Peel off the removable part of the label of the Hizentra vial. Put this label in your treatment diary or logbook with the date and time of the infusion. Also include the exact amount of Hizentra that you infused. |  |
Step 11: Clean up
|  |
Be sure to tell your doctor about any problems you have doing your infusions. Your doctor may ask to see your treatment diary or logbook, so be sure to take it with you each time you visit the doctor's office.
Call your doctor for medical advice about side effects. You can also report side effects to FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Manufactured by:
CSL Behring AG
Bern, Switzerland
US License No. 1766
Distributed by:
CSL Behring LLC
Kankakee, IL 60901 USA
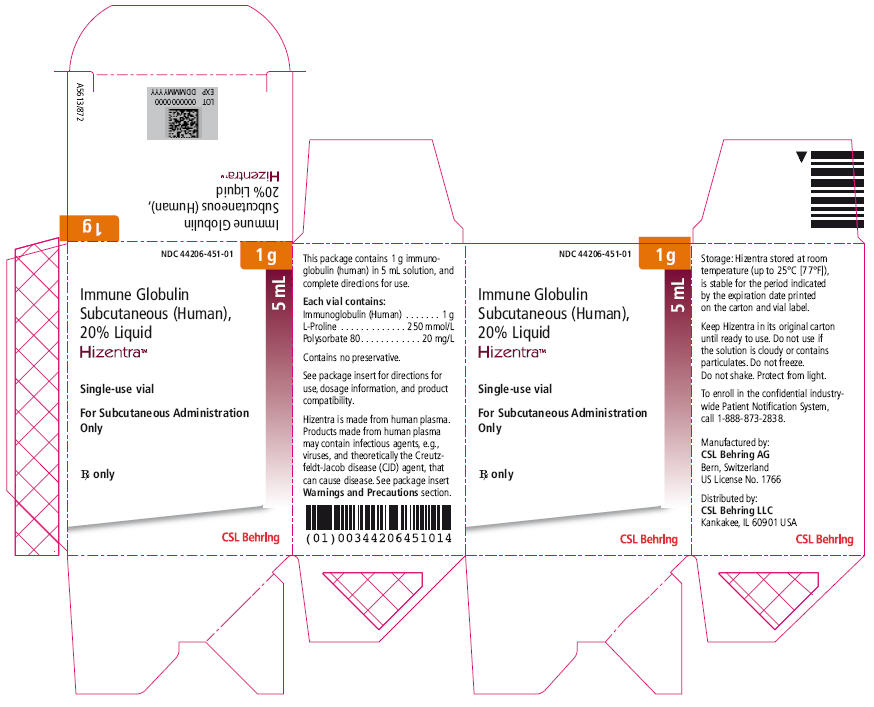
PRINCIPAL DISPLAY PANEL - 5 mL Vial Carton
NDC 44206-451-01
1 g
5 mL
Immune Globulin
Subcutaneous (Human),
20% Liquid
Hizentra™
Single-use vial
For Subcutaneous Administration
Only
Rx only
CSL Behring
PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton
NDC 44206-452-02
2 g
10 mL
Immune Globulin
Subcutaneous (Human),
20% Liquid
Hizentra™
Single-use vial
For Subcutaneous Administration
Only
Rx only
CSL Behring

PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton
NDC 44206-454-04
4 g
20 mL
Immune Globulin
Subcutaneous (Human),
20% Liquid
Hizentra™
Single-use vial
For Subcutaneous Administration
Only
Rx only
CSL Behring

| HIZENTRA
human immunoglobulin g liquid |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA125350 | 03/05/2010 | |
| HIZENTRA
human immunoglobulin g liquid |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA125350 | 03/05/2010 | |
| HIZENTRA
human immunoglobulin g liquid |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA125350 | 03/05/2010 | |
| Labeler - CSL Behring AG (481152762) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| CSL Behring AG | 481152762 | MANUFACTURE | |