TERBINEX
-
terbinafine hydrochloride tablet
JSJ Pharmaceuticals
----------
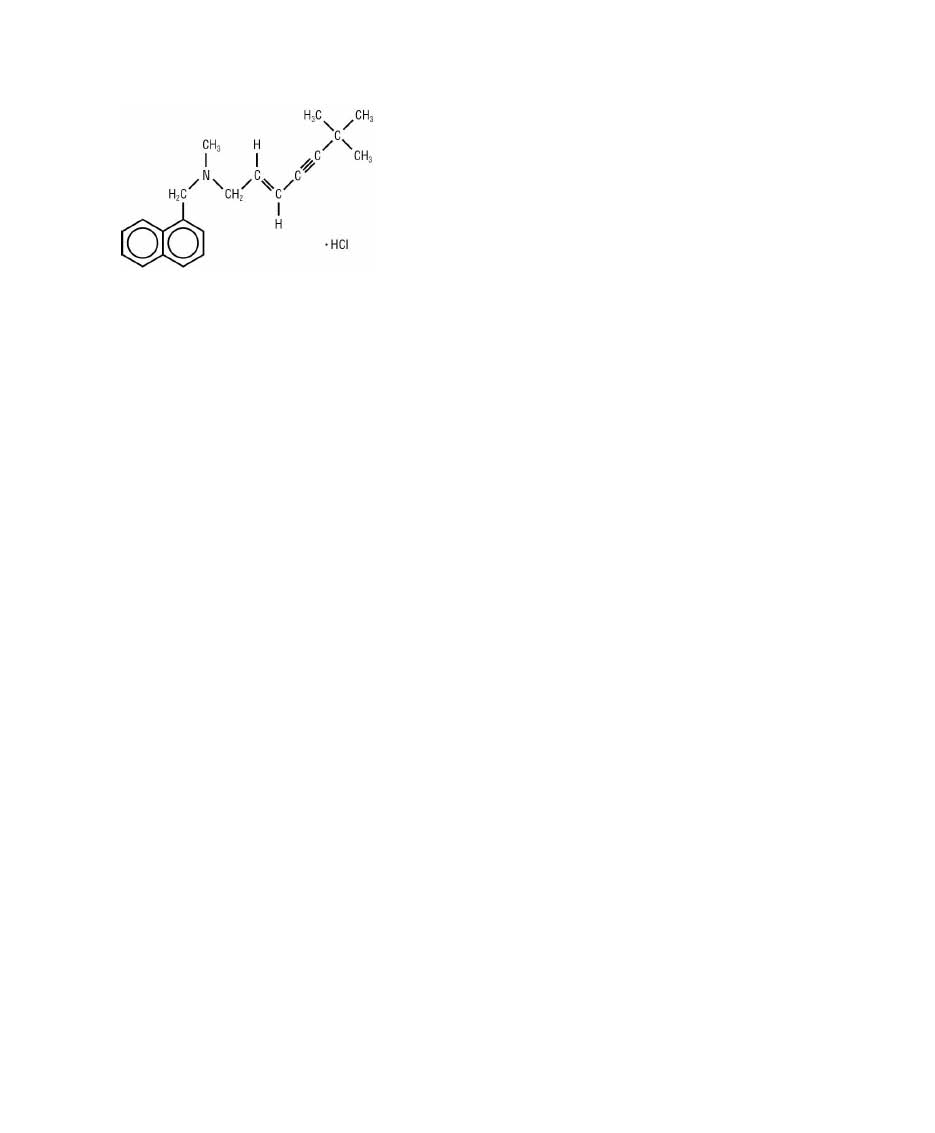
Terbinex (terbinafine Hydrochloride tablets equivalent to 250mg base)Terbinex™ Terbinafine Hydrochloride Tablets contain the synthetic allylamine antifungal compound terbinafine hydrochloride. Chemically, terbinafine
hydrochloride is (E)-N-(6,6-dimethyl-2-hepten-4-ynyl)-N-methyl-1-naphthalenemethanamine hydrochloride. The empirical formula C21H26ClN with a
molecular weight of 327.90, and the following structural formula:
Terbinafine hydrochloride is a white to off-white fine crystalline powder. It is freely soluble in methanol and methylene chloride, soluble in ethanol, and
slightly soluble in water.
Each tablet contains: Active Ingredients: terbinafine hydrochloride (equivalent to 250 mg base)
Inactive Ingredients: colloidal silicon dioxide, NF; hypromellose, USP; magnesium stearate, NF; microcrystalline cellulose, NF;
sodium starch glycolate, NF
Following oral administration, terbinafine is well absorbed (>70%) and the bioavailability of Terbinafine Hydrochloride Tablets as a result of first-pass
metabolism is approximately 40%. Peak plasma concentrations of 1 μg/mL appear within 2 h after a single 250 mg dose; the AUC (area under the curve) is
approximately 4.56 μg•h/mL. An increase in the AUC of terbinafine of less than 20% is observed when terbinafine hydrochloride is administered with food. No
clinically relevant age-dependent changes in steady-state plasma concentrations of terbinafine have been reported. In patients with renal impairment (creatinine
clearance ≤ 50mL/min) or hepatic cirrhosis, the clearance of terbinafine is decreased by approximately 50% compared to normal volunteers. No effect of
gender on the blood levels of terbinafine was detected in clinical trials. In plasma, terbinafine is >99% bound to plasma proteins and there are no specific
binding sites. At steady-state, in comparison to a single dose, the peak concentration of terbinafine is 25% higher and plasma AUC increases by a factor
of 2.5; the increase in plasma AUC is consistent with an effective half-life of ~36 hours. Terbinafine is distributed to the sebum and skin. A terminal half-life of
200-400 h may represent the slow elimination of terbinafine from tissues such as skin and adipose. Prior to excretion, terbinafine is extensively metabolized.
No metabolites have been identified that have antifungal activity similar to terbinafine. Approximately 70% of the administered dose is eliminated
in the urine.
Terbinafine hydrochloride is a synthetic allylamine derivative. Terbinafine hydrochloride is hypothesized to act by inhibiting squalene epoxidase, thus
blocking the biosynthesis of ergosterol, an essential component of fungal cell membranes. In vitro, mammalian squalene epoxidase is only inhibited at higher
(4000 fold) concentrations than is needed for inhibition of the dermatophyte enzyme. Depending on the concentration of the drug and the fungal species test
in vitro, terbinafine hydrochloride may be fungicidal. However, the clinical significance of in vitro data is unknown. Terbinafine has been shown to be active
against most strains of the following microorganisms both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
Trichophyton mentagrophytes
Trichophyton rubrum
The following in vitro data are available, but their clinical significance is unknown. In vitro, terbinafine exhibits satisfactory MIC’s against most strains of
the following microorganisms; however, the safety and efficacy of terbinafine in treating clinical infections due to these microorganisms have not been
established in adequate and well-controlled clinical trials:
Candida albicans
Epidermophyton floccosum
Scopulariopsis brevicaulis
The efficacy of Terbinafine Hydrochloride Tablets in the treatment of onychomycosis is illustrated by the response of patients with toenail and/or
fingernail infections who participated in three US/Canadian placebo-controlled clinical trials.
Results of the first toenail study, as assessed at week 48 (12 weeks of treatment with 36 weeks follow-up after completion of therapy), demonstrated
mycological cure, defined as simultaneous occurrence of negative KOH plus negative culture, in 70% of patients. Fifty-nine percent (59%) of patients
experienced effective treatment (mycological cure plus 0% nail involvement or >5mm of new unaffected nail growth); 38% of patients demonstrated
mycological cure plus clinical cure (0% nail involvement).
In a second toenail study of dermatophytic onychomycosis, in which nondermatophytes were also cultured, similar efficacy against the dermatophytes
was demonstrated. The pathogenic role of the non-dermatophytes cultured in the presence of dermatophytic onychomycosis has not been established. The
clinical significance of this association is unknown.
Results of the fingernail study, as assessed at week 24 (6 weeks of treatment with 18 weeks follow-up after completion of therapy), demonstrated
mycological cure in 79% of patients, effective treatment in 75% of the patients, and mycological cure plus clinical cure in 59% of the patients.
The mean time to overall success was approximately 10 months for the first toenail study and 4 months for the fingernail study. In the first toenail study, for
patients evaluated at least six months after achieving clinical cure and at least one year after completing terbinafine hydrochloride therapy, the clinical relapse
rate was approximately 15%.
Terbinafine Hydrochloride Tablets are indicated for the treatment of onychomycosis of the toenail or fingernail due to dermatophytes (tinea
unguium) (see DOSAGE AND ADMINISTRATION and CLINICAL STUDIES).
Prior to initiating treatment, appropriate nail specimens for laboratory testing (KOH preparation, fungal culture, or nail biopsy) should be obtained to confirm
the diagnosis of onychomycosis.
Terbinafine Hydrochloride Tablets are contraindicated in individuals with hypersensitivity to terbinafine or to any other ingredients of the formulation.
onychomycosis in individuals with and without pre-existing liver disease.
In the majority of liver cases reported in association with terbinafine hydrochloride use, the patients had serious underlying systemic conditions and
an uncertain causal association with terbinafine hydrochloride. The severity of hepatic events and/or their outcome may be worse in patients with active or
chronic liver disease (see PRECAUTIONS). Treatment with Terbinafine Hydrochloride Tablets should be discontinued if biochemical or clinical evidence
of liver injury develops (see PRECAUTIONS below).
There have been isolated reports of serious skin reactions (e.g., Stevens-Johnson Syndrome and toxic epidermal necrolysis). If progressive skin rash
occurs, treatment with terbinafine hydrochloride should be discontinued.
General: Terbinafine Hydrochloride Tablets are not recommended for patients with chronic or active liver disease.
Before prescribing Terbinafine Hydrochloride Tablets, pre-existing liver disease should be assessed. Hepatotoxicity may occur
in patients with and without pre-existing liver disease. Pretreatment serum transaminase (ALT and AST) tests are advised for all patients before taking
Terbinafine Hydrochloride Tablets. Patients prescribed Terbinafine Hydrochloride Tablets should be warned to report immediately to their physician any
symptoms of persistent nausea, anorexia, fatigue, vomiting, right upper abdominal pain or jaundice, dark urine or pale stools (see WARNINGS). Patients
with these symptoms should discontinue taking oral terbinafine, and the patient’s liver function should be immediately evaluated.
In patients with renal impairment (creatinine clearance ≤50 mL/ min), the use of terbinafine hydrochloride has not been adequately studied, and therefore, is not
recommended (see CLINICAL PHARMACOLOGY, Pharmacokinetics).
During postmarketing experience, precipitation and exacerbation of cutaneous and systemic lupus erythematosus have been reported infrequently in patients
taking terbinafine hydrochloride. Terbinafine hydrochloride therapy should be discontinued in patients with clinical signs and symptoms suggestive of lupus
erythematosus.
Changes in the ocular lens and retina have been reported following the use of Terbinafine Hydrochloride Tablets in controlled trials. The clinical significance of
these changes is unknown.
hydrochloride treated patients (1.7%) and 3/137 placebo-treated patients (2.2%) had decreases in ALC to below 1000/mm3 on two or more occasions.
The clinical significance of this observation is unknown. However, in patients with known or suspected immunodeficiency, physicians should consider
monitoring complete blood counts in individuals using Terbinafine Hydrochloride therapy for greater than six weeks.
Isolated cases of severe neutropenia have been reported. These were reversible upon discontinuation of terbinafine hydrochloride with or without supportive
therapy. If clinical signs and symptoms suggestive of secondary infection occur, a complete blood count should be obtained. If the neutrophil count is ≤1,000
cells/mm3, terbinafine hydrochloride should be discontinued and supportive management started.
the following drug classes; tricyclic antidepressants, selective serotonin reuptake inhibitors, beta-blockers, antiarrhythmics class 1C (e.g. flecainide and
propafenone) and monoamine oxidase inhibitors Type B. Coadministration of Terbinafine Hydrochloride Tablets should be done with careful monitoring and
may require a reduction in dose of the 2D6-metabolized drug. In a study to assess the effects of terbinafine on desipramine in healthy volunteers
characterized as normal metabolizers, the administration of terbinafine resulted in a 2-fold increase in Cmax and a 5-fold increase in AUC. In this study, these
effects were shown to persist at the last observation at 4 weeks after discontinuation of terbinafine hydrochloride.
In vitro studies with human liver microsomes showed that terbinafine does not inhibit the metabolism of tolbutamide, ethinylestradiol, ethoxycoumarin, and
cyclosporine.
In vivo drug-drug interaction studies conducted in healthy volunteer subjects showed that terbinafine does not affect the clearance of antipyrine or digoxin.
Terbinafine decreases the clearance of caffeine by 19%. Terbinafine increases the clearance of cyclosporine by 15%.
There have been spontaneous reports of increase or decrease in prothrombin times in patients concomitantly taking oral terbinafine and warfarin, however, a
causal relationship between Terbinafine Hydrochloride Tablets and these changes has not been established.
Terbinafine clearance is increased 100% by rifampin, a CYP450 enzyme inducer, and decreased 33% by cimetidine, a CYP450 enzyme inhibitor. Terbinafine
clearance is unaffected by cyclosporine.
There is no information available from adequate drug-drug interaction studies with the following classes of drugs: oral contraceptives, hormone replacement
therapies, hypoglycemics, theophyllines, phenytoins, thiazide diuretics, and calcium channel blockers.
In a 28-month oral carcinogenicity study in rats, an increase in the incidence of liver tumors was observed in males at the highest dose tested, 69 mg/kg/day [2x
the Maximum Recommended Human Dose (MRHD) based on AUC comparisons of the parent terbinafine]; however, even though dose limiting toxicity was not
achieved at the highest tested dose, higher doses were not tested.
The results of a variety of in vitro (mutations in E. coli and S. typhimurium, DNA repair in rat hepatocytes, mutagenicity in Chinese hamster fibroblasts,
chromosome aberration and sister chromatid exchanges in Chinese hamster lung cells), and in vivo (chromosome aberration in Chinese hamsters,
micronucleus test in mice) genotoxicity tests gave no evidence of a mutagenic or clastogenic potential. Oral reproduction studies in rats at doses up to
300 mg/kg/day (approximately 12x the MRHD based on body surface area comparisons, BSA) did not reveal any specific effects on fertility or other
reproductive parameters. Intravaginal application of terbinafine hydrochloride at 150 mg/day in pregnant rabbits did not increase the incidence of abortions or
premature deliveries nor affect fetal parameters. Pregnancy Category B: Oral reproduction studies have been performed in rabbits and rats at doses
up to 300 mg/kg/day (12x to 23x the MRHD, in rabbits and rats, respectively, based on BSA) and have revealed no evidence of impaired
fertility or harm to the fetus due to terbinafine. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction
studies are not always predictive of human response, and because treatment of onychomycosis can be postponed until after pregnancy is completed, it is
recommended that terbinafine hydrochloride not be initiated during pregnancy. After oral administration, terbinafine is present in breast milk of nursing mothers. The ratio of terbinafine in milk to plasma is 7:1. Treatment with
terbinafine hydrochloride is not recommended in nursing mothers. The safety and efficacy of Terbinafine Hydrochloride Tablets have not been established in pediatric patients.
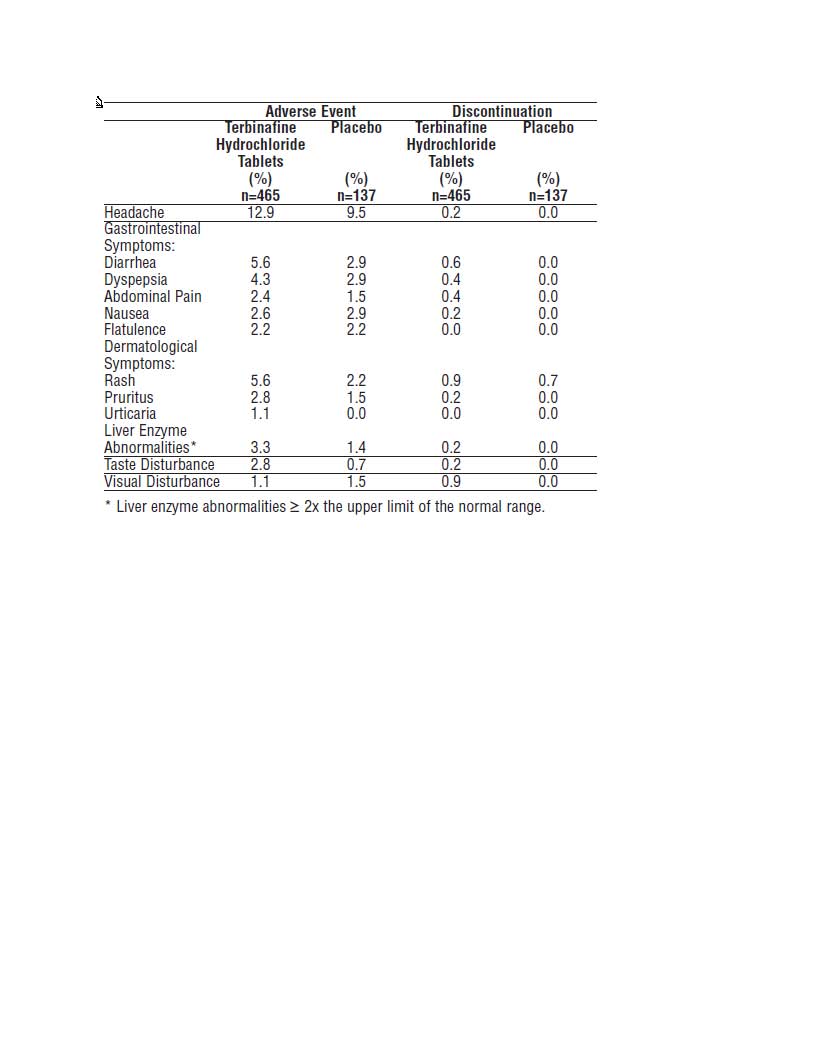
The most frequently reported adverse events observed in the three US/Canadian placebo-controlled trials are listed in the table below. The adverse events
reported encompass gastrointestinal symptoms (including diarrhea, dyspepsia, and abdominal pain), liver test abnormalities, rashes, urticaria, pruritus, and
taste disturbances. In general, the adverse events were mild, transient, and did not lead to discontinuation from study participation.
Adverse events, based on worldwide experience with Terbinafine Hydrochloride Tablets use, include: idiosyncratic and symptomatic hepatic injury and more
rarely, cases of liver failure, some leading to death or liver transplant, (see WARNINGS and PRECAUTIONS), serious skin reactions (see WARNINGS),
severe neutropenia (see PRECAUTIONS), thrombocytopenia, angioedema and allergic reactions (including anaphylaxis). Psoriasiform eruptions or
exacerbation of psoriasis, acute generalized exanthematous pustulosis and precipitation and exacerbation of cutaneous and systemic lupus erythematosus
have been reported in patients taking Terbinafine Hydrochloride Tablets. Terbinafine Hydrochloride Tablets may cause taste disturbance (including taste
loss) which usually recovers within several weeks after discontinuation of the drug. There have been isolated reports of prolonged (greater than one year)
taste disturbance. Taste disturbances associated with oral terbinafine have been reported to be severe enough to result in decreased food intake leading to
significant and unwanted weight loss.
Other adverse reactions which have been reported include malaise, fatigue, vomiting, arthralgia, myalgia, and hair loss.
Clinical adverse effects reported spontaneously since the drug was marketed include altered prothrombin time (prolongation and reduction) in patients
concomitantly treated with warfarin and Terbinafine Hydrochloride Tablets and agranulocytosis (rare).
Clinical experience regarding overdose with Terbinafine Hydrochloride Tablets is limited. Doses up to 5 grams (20 times the therapeutic daily dose) have
been taken without inducing serious adverse reactions. The symptoms of overdose included nausea, vomiting, abdominal pain, dizziness, rash, frequent
urination,and headache.
Terbinafine Hydrochloride Tablets, one 250 mg tablet, should be taken once daily for 6 weeks by patients with fingernail onychomycosis.
Terbinafine hydrochloride, one 250 mg tablet, should be taken once daily for 12 weeks by patients with toenail onychomycosis.
The optimal clinical effect is seen some months after mycological cure and cessation of treatment. This is related to the
period required for outgrowth of healthy nail.
Terbinex™ Terbinafine Hydrochloride Tablets
Supplied as white, round, flat faced beveled edge tablets debossed with IG on one side and 209 on the other.
Bottle of 42 tablets packaged with 12 mL bottle of Eco Formula NDC 68712-037-01
Store tablets at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]; in a tight container.
Protect from light.
Manufactured by:
INVAGEN Pharmaceuticals, Inc.
Hauppauge, NY 11788
Manufactured for:
JSJ Pharmaceuticals
Charleston SC 29401
800-499-4468
www.jsjpharm.com
hepatocytes suggest that peroxisome proliferation in the liver is a rat-specific finding. However, other effects, including
increased liver weights and APTT, occurred in dogs and monkeys at doses giving Css trough levels of the parent terbinafine 2-3x those seen in humans at
the MRHD. Higher doses were not tested.
| TERBINEX
terbinafine hydrochloride tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA077533 | 01/31/2009 | |
| Labeler - JSJ Pharmaceuticals (615074866) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| InvaGen | 165104469 | manufacture | |