ATGAM- equine thymocyte immune globulin injection, solution
Pharmacia and Upjohn Company
----------
Atgam®
(lymphocyte immune globulin,
anti-thymocyte globulin [equine] sterile solution)
WARNING
Only physicians experienced in immunosuppressive therapy in the treatment of renal transplant or aplastic anemia patients should use ATGAM.
Patients receiving ATGAM should be treated in facilities equipped and staffed with adequate laboratory and supportive medical resources.
DESCRIPTION
ATGAM Sterile Solution contains lymphocyte immune globulin, anti-thymocyte globulin [equine]. It is the purified, concentrated, and sterile gamma globulin, primarily monomeric IgG, from hyperimmune serum of horses immunized with human thymus lymphocytes. ATGAM is a transparent to slightly opalescent aqueous protein solution. It may appear colorless to faintly pink or brown and is nearly odorless. It may develop a slight granular or flaky deposit during storage. (For information about in-line filters, see Infusion Instructions in the DOSAGE AND ADMINISTRATION SECTION.)
Before release for clinical use, each lot of ATGAM is tested to assure its ability to inhibit rosette formation between human peripheral lymphocytes and sheep red blood cells in vitro. In each lot, antibody activity against human red blood cells and platelets is also measured and determined to be within acceptable limits. Only lots that test negative for antihuman serum protein antibody, antiglomerular basement membrane antibody and pyrogens are released.
Each milliliter of ATGAM contains 50 mg of horse gamma globulin stabilized in 0.3 molar glycine to a pH of approximately 6.8.
CLINICAL AND ANIMAL PHARMACOLOGY
ATGAM Sterile Solution is a lymphocyte-selective immunosuppressant as is demonstrated by its ability to reduce the number of circulating, thymus-dependent lymphocytes that form rosettes with sheep erythrocytes. This antilymphocytic effect is believed to reflect an alteration of the function of the T lymphocytes, which are responsible in part for cell-mediated immunity and are involved in humoral immunity. In addition to its antilymphocytic activity, ATGAM contains low concentrations of antibodies against other formed elements of the blood. In rhesus and cynomolgus monkeys, ATGAM reduces lymphocytes in the thymus-dependent areas of the spleen and lymph nodes. It also decreases the circulating sheep-erythrocyte-rosetting lymphocytes that can be detected, but ordinarily ATGAM does not cause severe lymphopenia.
In general, when ATGAM is given with other immunosuppressive therapy, such as antimetabolites and corticosteroids, the patient's own antibody response to horse gamma globulin is minimal. In a small clinical study, ATGAM administered with other immunosuppressive therapy and measured as horse IgG had a serum half-life of 5.7±3 days.
INDICATIONS AND USAGE
Renal Transplantation
ATGAM Sterile Solution is indicated for the management of allograft rejection in renal transplant patients. When administered with conventional therapy at the time of rejection, it increases the frequency of resolution of the acute rejection episode. The drug has also been administered as an adjunct to other immunosuppressive therapy to delay the onset of the first rejection episode. Data accumulated to date have not consistently demonstrated improvement in functional graft survival associated with therapy to delay the onset of the first rejection episode.
Aplastic Anemia
ATGAM is indicated for the treatment of moderate to severe aplastic anemia in patients who are unsuitable for bone marrow transplantation.
When administered with a regimen of supportive care, ATGAM may induce partial or complete hematologic remission. In a controlled trial, patients receiving ATGAM showed a statistically significantly higher improvement rate compared with standard supportive care at 3 months. Improvement was defined in terms of sustained increase in peripheral blood counts and reduced transfusion needs.
Clinical trials conducted at two centers evaluated the 1-year survival rate for patients with severe and moderate to severe aplastic anemia. Seventy-four of the 83 patients enrolled were evaluable based on response to treatment. The treatment groups studied consisted of 1) ATGAM and supportive care, 2) ATGAM administered following 3 months of supportive care alone, 3) ATGAM, mismatched marrow infusion, androgens, and supportive care, or 4) ATGAM, androgens, and supportive care. There were no statistically significant differences between the treatment groups. The 1-year survival rate for the pooled treatment groups was 69%. These survival results can be compared with a historical survival rate of about 25% for patients receiving standard supportive care alone.
The usefulness of ATGAM has not been demonstrated in patients with aplastic anemia who are suitable candidates for bone marrow transplantation or in patients with aplastic anemia secondary to neoplastic disease, storage disease, myelofibrosis, Fanconi's syndrome, or in patients known to have been exposed to myelotoxic agents or radiation.
To date, safety and efficacy have not been established in circumstances other than renal transplantation and aplastic anemia.
CONTRAINDICATIONS
Do not administer ATGAM Sterile Solution to a patient who has had a severe systemic reaction (e.g., anaphylactic reaction) during prior administration of ATGAM or any other equine gamma globulin preparation.
WARNINGS
Only physicians experienced in immunosuppressive therapy in the treatment of renal transplant or aplastic anemia patients should use ATGAM.
Patients receiving ATGAM should be treated in facilities equipped and staffed with adequate laboratory and supportive medical resources.
Precise methods of determining the potency of ATGAM have not been established, thus activity may potentially vary from lot to lot.
Discontinue treatment with ATGAM if any of the following occurs:
- Symptoms of anaphylaxis (See ADVERSE REACTIONS)
- Severe and unremitting thrombocytopenia in renal transplant patients
- Severe and unremitting leukopenia in renal transplant patients
Immune-Mediated Reactions
In rare instances, serious immune-mediated reactions have been reported with the use of ATGAM. Clinical signs associated with anaphylaxis, other infusion associated reactions, and serum sickness have been reported.
A systemic reaction such as a generalized rash, tachycardia, dyspnea, hypotension, or anaphylaxis precludes any additional administration of ATGAM.
Skin Testing
To identify those at greatest risk of systemic anaphylaxis, skin testing potential recipients is strongly recommended before commencing treatment. A conservative, conventional approach would first employ epicutaneous (prick) testing with undiluted ATGAM. If the subject does not show a wheal ten minutes after pricking, proceed to intradermal testing with 0.02 mL of a 1:1000 v/v (volume/volume) saline dilution of ATGAM with a separate saline control injection of similar volume. Read the result at 10 minutes: a wheal at the ATGAM site 3 or more mm larger in diameter than that at the saline control site (or a positive prick test) suggests clinical sensitivity and an increased possibility of a systemic allergic reaction should the drug be dosed intravenously.
The predictive value of this test has not been proven clinically. Allergic reactions such as anaphylaxis have occurred in patients whose skin test is negative. Also, skin testing done as described above will not predict for later development of serum sickness. In the presence of a locally positive skin test to ATGAM, serious consideration to alternative forms of therapy should be given. The risk to benefit ratio must be carefully weighed. If therapy with ATGAM is deemed appropriate following a locally positive skin test, treatment should be administered in a setting where intensive life support facilities are immediately available and a physician familiar with the treatment of potentially life threatening allergic reactions is in attendance.
Infection
Because this product is made using equine and human blood components, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
Immunizations
Live-virus vaccines may not replicate successfully and antibody response could be reduced when the vaccine is administered after immune globulin administration. Live-virus vaccines should ideally be administered six months after therapy with intramuscular immune globulin and six months after therapy with intravenous immune globulin.
PRECAUTIONS
General
Because ATGAM Sterile Solution is an immunosuppressive agent ordinarily given with corticosteroids and antimetabolites, monitor patients carefully for signs of leukopenia, thrombocytopenia, or concurrent infection. Several studies have suggested an increase in the incidence of cytomegalovirus infection in patients receiving ATGAM(1). In one study it has been found that it may be possible to reduce this risk by decreasing the dosage of other immunosuppressive agents administered concomitantly with ATGAM. If infection occurs, institute appropriate adjunctive therapy promptly. On the basis of the clinical circumstances, a physician should decide whether or not therapy with ATGAM will continue.
The safety and effectiveness of ATGAM have been demonstrated only in renal transplant patients who received concomitant immunosuppressive therapy and in patients with aplastic anemia.
Dilution of ATGAM in dextrose injection, USP, is not recommended, as low salt concentrations may result in precipitation. The use of highly acidic infusion solutions is also not recommended because of possible physical instability over time.
Driving and Using Machinery
No studies on the effect of ability to drive or use machines have been performed. Given the potential adverse reactions that may be experienced (e.g., dizziness, convulsion, confusional state, syncope), caution should be taken when driving or using machinery while on this medication.
Drug Interactions
When the dose of corticosteroids and other immunosuppressants is being reduced, some previously masked reactions to ATGAM may appear. Under these circumstances, observe patients especially carefully during therapy with ATGAM.
Pregnancy
Pregnancy category C
ATGAM was not teratogenic in rats or monkeys at a dose up to 20 mg/kg (See ANIMAL TOXICOLOGY). However, 20 mg/kg/day ATGAM for 16 days during organogenesis in cynomolgus monkeys was fetotoxic. No fetal or maternal toxicity was seen with 10 mg/kg/day ATGAM administered for 16 days during organogenesis. There are no adequate and well-controlled studies in pregnant women. It is also not known whether ATGAM can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity.
ATGAM should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
In animal studies, a single dose of ATGAM up to 40 mg/kg was not detected at the limit of quantification in the milk of lactating cynomolgus monkeys. It is not known whether ATGAM is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing neonates and infants from ATGAM, a decision should be made whether to discontinue nursing or to discontinue the drug taking into account the importance of the drug to the mother.
Geriatric Use
Clinical experience in a limited number of elderly patients (≥65 years of age) has not identified differences in responses between the elderly and younger patients. In general, the dose for an elderly patient should be selected with caution, usually starting at the low end of the dosage range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy in this age group.
ADVERSE REACTIONS
Renal Transplantation
The primary clinical experience with ATGAM Sterile Solution has been in renal allograft patients who were also receiving concurrent standard immunosuppressive therapy (azathioprine, corticosteroids). In controlled trials, investigators frequently reported the following adverse reactions: fever in 1 patient in 3; chills in 1 patient in 7; leukopenia in 1 patient in 7; thrombocytopenia in 1 patient in 9; and dermatologic reactions, such as rash, pruritus, urticaria, wheal, and flare, in 1 patient in 8. The following reactions were reported in more than 1% but less than 5% of the patients: arthralgia; chest or back pain, or both; clotted A/V fistula; diarrhea; dyspnea; headache; hypotension; nausea or vomiting, or both; night sweats; pain at the infusion site; peripheral thrombophlebitis; and stomatitis.
Reactions reported in less than 1% of the patients in the controlled trials were anaphylaxis, dizziness, weakness or faintness, edema, herpes simplex reactivation, hiccoughs or epigastric pain, hyperglycemia, hypertension, iliac vein obstruction, laryngospasm, localized infection, lymphadenopathy, malaise, myalgia, paresthesia, possible serum sickness, pulmonary edema, renal artery thrombosis, seizures, systemic infection, tachycardia, toxic epidermal necrosis, and wound dehiscence.
Aplastic Anemia
In premarketing clinical trials with ATGAM in the treatment of aplastic anemia, patients were also being concurrently managed with support therapy (transfusions, steroids, antibiotics, antihistamines).
In these trials most patients experienced fever and skin reactions. Other frequently reported adverse reactions were chills, 1 patient in 2; arthralgia, 1 patient in 2; headache, 1 patient in 6; myalgia, 1 patient in 10; nausea, 1 patient in 15; chest pain, 1 patient in 15 and phlebitis, 1 patient in 20.
The following reactions were reported by at least one patient and less than 5% of the total patients: diaphoresis, joint stiffness, periorbital edema, aches, edema, muscle ache, vomiting, agitation/lethargy, listlessness, light-headedness, seizures, diarrhea, bradycardia, myocarditis, cardiac irregularity, hepatosplenomegaly, possible encephalitis or post viral encephalopathy, hypotension, congestive heart failure, hypertension, burning soles/palms, foot sole pain, lymphadenopathy, post-cervical lymphadenopathy, tender lymph nodes, bilateral pleural effusion, respiratory distress, anaphylactic reaction, and proteinuria.
In some trials, clinical and laboratory findings of serum sickness were seen in a majority of patients.
Postmarketing Experience
During approximately 5 years of post approval marketing experience, the frequency of adverse reactions in voluntarily reported cases is as follows: fever 51%; chills 16%; thrombocytopenia 30%; leukopenia 14%; rashes 27%; systemic infection 13%. Events reported in 5% to 10% of reported cases include abnormal renal function tests; serum sickness-like symptoms; dyspnea/apnea; arthralgia; chest, back, or flank pain; diarrhea and nausea and/or vomiting. Events reported with a frequency of less than 5% include: hypertension, Herpes Simplex infection, pain, swelling or redness at infusion site, eosinophilia, headache, myalgias, or leg pains, hypotension, anaphylaxis, tachycardia, edema, localized infection, malaise, seizures, GI bleeding or perforation, deep vein thrombosis, sore mouth/throat, hyperglycemia, acute renal failure, abnormal liver function tests, confusion or disorientation, cough, neutropenia or granulocytopenia, anemia, thrombophlebitis, dizziness, epigastric or stomach pain, lymphadenopathy, pulmonary edema or congestive heart failure, abdominal pain, nosebleed, vasculitis, aplasia or pancytopenia, abnormal involuntary movement or tremor, rigidity, sweating, laryngospasm/edema, hemolysis or hemolytic anemia, viral hepatitis, faintness, enlarged or ruptured kidney, paresthesias, and renal artery thrombosis.
The recommended management for some of the adverse reactions that could occur with treatment with ATGAM follows:
- Anaphylaxis is uncommon but serious and may occur at any time during therapy with ATGAM. Stop infusion of ATGAM immediately; administer 0.3 mL aqueous epinephrine (1:1,000 solution) intramuscularly. Administer steroids; assist respiration; and provide other resuscitative measures. DO NOT resume therapy with ATGAM.
- Hemolysis can usually be detected only in the laboratory. Clinically significant hemolysis has been reported rarely. Appropriate treatment of hemolysis may include transfusion of erythrocytes; if necessary, administer intravenous mannitol, furosemide, sodium bicarbonate, and fluids. Severe and unremitting hemolysis may require discontinuation of therapy with ATGAM.
- Thrombocytopenia is usually transient in renal transplant patients; platelet counts generally return to adequate levels without discontinuing therapy with ATGAM. Platelet transfusions may be necessary in patients with aplastic anemia. (See PRECAUTIONS, WARNINGS, and DOSAGE AND ADMINISTRATION.)
- Respiratory distress may indicate an anaphylactoid reaction. Discontinue infusion of ATGAM. If distress persists, administer an antihistamine, epinephrine, corticosteroids, or some combination of the three.
- Pain in chest, flank, or back may indicate anaphylaxis or hemolysis. Treatment is that indicated above for those conditions.
- Hypotension may indicate anaphylaxis. Stop infusion of ATGAM and stabilize blood pressure with pressors if necessary.
- Chills and fever occur frequently in patients receiving ATGAM. ATGAM may release endogenous leukocyte pyrogens. Prophylactic and/or therapeutic administration of antihistamines, antipyretics, or corticosteroids generally controls this reaction.
- Chemical phlebitis can be caused by infusion of ATGAM through peripheral veins. This can often be avoided by administering the infusion solution into a high-flow vein. A subcutaneous arterialized vein produced by a Brescia fistula is also a useful administration site.
- Itching and erythema probably result from the effect of ATGAM on blood elements. Antihistamines generally control the symptoms.
- Serum sickness-like symptoms in aplastic anemia patients have been treated with oral or IV corticosteroids. Resolution of symptoms has generally been prompt and long-term sequelae have not been observed. Prophylactic administration of corticosteroids may decrease the frequency of this reaction.
OVERDOSAGE
The maximum tolerated dose of ATGAM Sterile Solution would be expected to vary from patient to patient due to the biological nature of the product. The largest single daily dose known to be administered to a patient (renal transplant recipient) was 7,000 mg administered at a concentration of approximately 10 mg/mL Sodium Chloride Injection, USP, seven times the recommended total dose and infusion concentration. In this patient, the administration of ATGAM was not associated with any signs of acute intoxication or late sequelae.
A maximum therapeutic dose has not been established therefore the definition of overdose for ATGAM has not been clearly defined. Some renal transplant patients have received up to 50 doses in 4 months, and others have received 28-day courses of 21 doses followed by as many as 3 more courses for the treatment of acute rejection. The incidence of toxicologic manifestations did not increase with any of these regimens; however, close monitoring of the patient is recommended.
DOSAGE AND ADMINISTRATION
Renal Allograft Recipients
Adult renal allograft patients have received ATGAM Sterile Solution at the dosage of 10 to 30 mg/kg of body weight daily. The few children studied received 5 to 25 mg/kg daily. ATGAM has been used to delay the onset of the first rejection episode(2–5) and at the time of the first rejection episode.(6–10) Most patients who received ATGAM for the treatment of acute rejection had not received it starting at the time of transplantation.
Usually, ATGAM is used concomitantly with azathioprine and corticosteroids, which are commonly used to suppress the immune response. Exercise caution during repeat courses of ATGAM; carefully observe patients for signs of allergic reactions.
Aplastic Anemia
The recommended dosage regimen is 10 to 20 mg/kg daily for 8 to 14 days. Additional alternate-day therapy up to a total of 21 doses can be administered.(11–13) Because thrombocytopenia can be associated with the administration of ATGAM, patients receiving it for the treatment of aplastic anemia may need prophylactic platelet transfusions to maintain platelets at clinically acceptable levels.
Preparation of Solution
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. However, because ATGAM is a gamma globulin product, it can be transparent to slightly opalescent, colorless to faintly pink or brown, and may develop a slight granular or flaky deposit during storage. ATGAM (diluted or undiluted) should not be shaken because excessive foaming and/or denaturation of the protein may occur.
Dilute ATGAM for intravenous infusion in an inverted bottle of sterile vehicle so the undiluted ATGAM does not contact the air inside. Add the total daily dose of ATGAM to the sterile vehicle (see Compatibility and Stability). The concentration should not exceed 4 mg of ATGAM per mL. The diluted solution should be gently rotated or swirled to effect thorough mixing.
Administration
The diluted ATGAM should be allowed to reach room temperature before infusion. ATGAM is appropriately administered into a vascular shunt, arterial venous fistula, or a high-flow central vein through an in-line filter with a pore size of 0.2 to 1.0 micron. The in-line filter should be used with all infusions of ATGAM to prevent the administration of any insoluble material that may develop in the product during storage. The use of high-flow veins will minimize the occurrence of phlebitis and thrombosis. Do not infuse a dose of ATGAM in less than 4 hours. Always keep appropriate resuscitation equipment at the patient's bedside while ATGAM is being administered. Observe the patient continuously for possible allergic reactions throughout the infusions (See ADVERSE REACTIONS).
Compatibility and Stability
ATGAM, once diluted, has been shown to be physically and chemically stable for up to 24 hours at concentrations of up to 4 mg per mL in the following diluents: 0.9% sodium chloride injection, 5% dextrose and 0.225% sodium chloride injection, and 5% dextrose and 0.45% sodium chloride injection.
Adding ATGAM to dextrose injection is not recommended, as low salt concentrations can cause precipitation. Highly acidic infusion solutions can also contribute to physical instability over time. It is recommended that diluted ATGAM be stored in a refrigerator if it is prepared prior to the time of infusion. Even if it is stored in a refrigerator, the total time in dilution should not exceed 24 hours (including infusion time).
HOW SUPPLIED
ATGAM Sterile Solution, containing 50 mg of horse gamma globulin/mL, is supplied as follows:
| 5 – 5 mL ampoules | NDC 0009-7224-02 |
ANIMAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Mutagenicity and carcinogenicity studies have not been conducted on ATGAM.
In fertility studies, ATGAM at doses 10, 20 and 40 mg/kg/day was administered to cynomolgus monkeys (Macaca fascicularis) for 14 days either before (male monkeys) or before and after (female monkeys) cohabitation with untreated mates. ATGAM treatment was not associated with male or female hormonal or copulation behavior changes. A decrease in fertility index in female monkeys receiving ATGAM was seen. Female toxicity, including death, was observed with ATGAM doses of ≥20 mg/kg/day. While the etiology of this toxicity is uncertain, it may be attributed to hemolytic anemia due to cross-reactivity of ATGAM to a monkey red blood antigen.
In embryo-fetal toxicity studies, ATGAM was administered to rats and cynomolgus monkeys for 11 and 16 days, respectively during organogenesis. In rats, hypoplastic cervical vertebrae, a finding consistent with delayed skeletal development, were observed in fetuses whose dams received ATGAM at doses of 100 mg/kg/day during organogenesis. In monkey reproduction studies, maternal toxicity (vaginal bleeding, decreased body weight and loss of appetite) was observed with ATGAM doses ≥ 20 mg/kg/day after 16 days of dosing. Fetal deaths occurred in dams treated with 20 mg/kg/day ATGAM earlier in organogenesis (days 20–35), but not when treatment was given at a later part of organogenesis (days 35–50). The maternal and fetal deaths were attributed to maternal anemia due to red blood cell antigen that humans do not share. Therefore, this toxicity is not considered relevant to human fetal development.
REFERENCES
- Rubin RH, Cosimi AB, Hirsch MS, Herrin JT: Effects of antithymocyte globulin on cytomegalovirus infection in renal transplant recipients. Transplantation. 1981; 31(2):143–145.
- Cosimi AB, Wortis HH, Delmonico FL, Russell PS: Randomized clinical trial of antithymocyte globulin in cadaver renal allograft recipients: importance of T cell monitoring. Surgery. 1976; 80:155–163.
- Wechter WJ, Brodie JA, Morrell RM, Rafi M, Schultz JR: Antithymocyte globulin (ATGAM) in renal allograft recipients. Transplantation. 1979; 28(4):294–302.
- Kountz SL, Butt KHM, Rao TKS, Zielinski CM, Rafi M, Schultz JR: Antithymocyte globulin (ATG) dosage and graft survival in renal transplantation. Transplant. Proc. 1977; 9:1023–1025.
- Butt KMH, Zielinski CM, Parsa I, Elberg AJ, Wechter WJ, Kountz SL: Trends in immunosuppression for kidney transplantation. Kidney Int. 1978; 13(Suppl 8): S95–S98.
- Filo RS, Smith EJ, Leapman SB: Reversal of acute renal allograft rejection with adjunctive ATG therapy. Transplant. Proc. 1981; 13(1): 482–490.
- Nowygrod R, Appel G, Hardy M: Use of ATG for reversal of acute allograft rejection. Transplant. Proc. 1981; 13(1): 469–472.
- Hardy MA, Nowygrod R, Elberg A, Appel G: Use of ATG in treatment of steroid-resistant rejection. Transplantation. 1980; 29:162–164.
- Shield CH, Cosimi AB, Tolkoff-Rubin N, Rubin R, Herrin J, Russell PS: Use of antithymocyte globulin for reversal of acute allograft rejection. Transplantation. 1979; 28(6): 461–464.
- Cosimi AB: The clinical value of antilymphocyte antibodies. Transplant. Proc. 1981; 13(1): 462–468.
- Cosimi AB, Peters C, Harmon D, Ellman L: Treatment of severe aplastic anemia with a prolonged course of antithymocyte globulin. Transplant. Proc. 1982; 14:761–764.
- Champlin R, Ho W, Gale R: Antithymocyte globulin treatment in patients with aplastic anemia. N Engl J Med. 1983; 308(3):113–118.
- Doney K, Dahlberg S, Monroe D et al: Therapy of severe aplastic anemia with anti-human thymocyte globulin and androgens: The effect of HLA-haploidentical marrow infusion. Blood. 1984; 63(2):342–348.
PRINCIPAL DISPLAY PANEL - 5 mL Ampule Label
5 mL
NDC 0009-7224-01
Atgam®
lymphocyte immune globulin,
anti-thymocyte globulin (equine)
250 mg protein
50 mg/mL
For I.V. use only
DOSAGE AND USE: See accompanying prescribing
information. U.S. License No. 1216.
ATTENTION—May contain particles; this is
normal. Use 0.2µ to 1.0µ in-line filter. See insert.
Distributed by Pharmacia & Upjohn Co
Division of Pfizer Inc, NY, NY 10017
Rx only
PAA042882
LOT / EXP

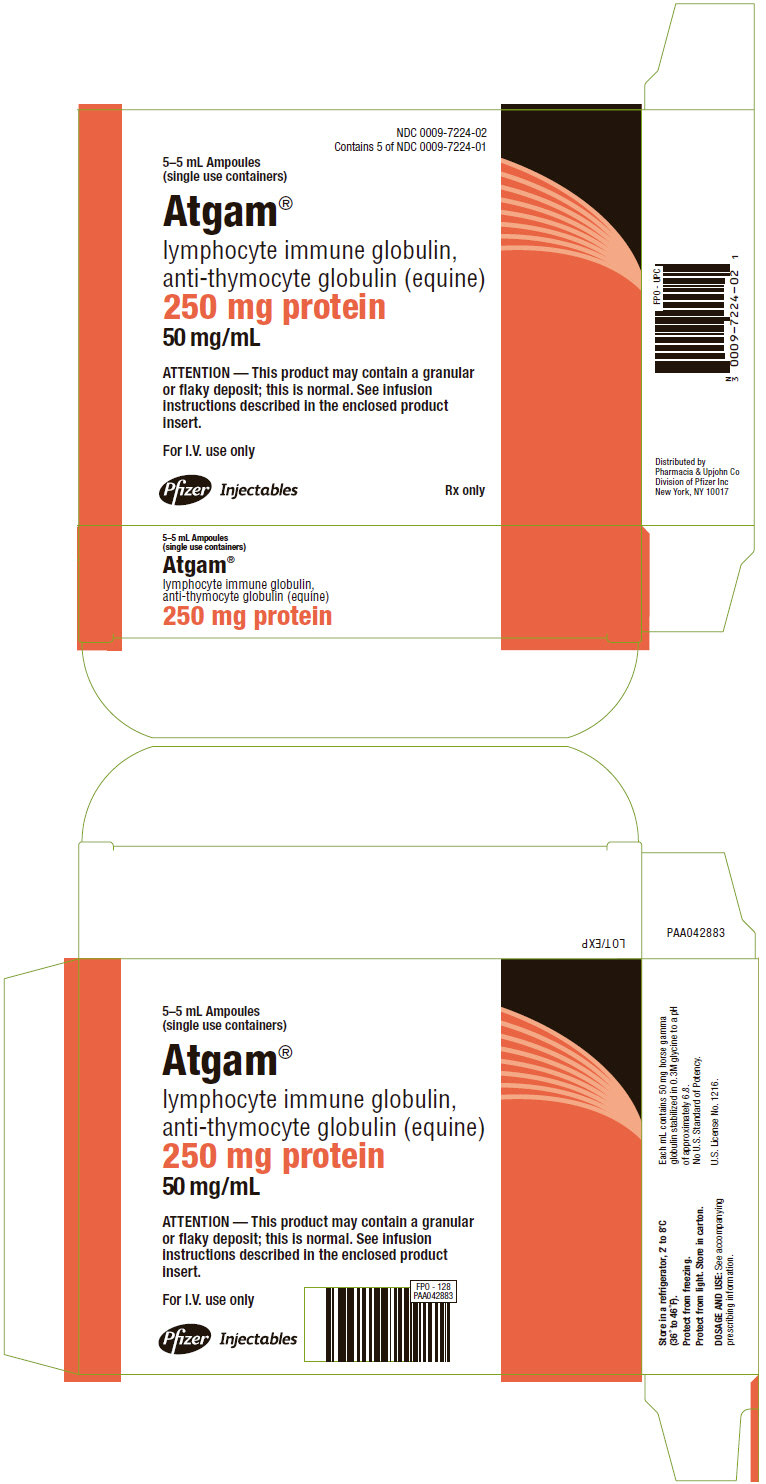
PRINCIPAL DISPLAY PANEL - 5–5 mL Ampule Carton
NDC 0009-7224-02
Contains 5 of NDC 0009-7224-01
5–5 mL Ampoules
(single use containers)
Atgam®
lymphocyte immune globulin,
anti-thymocyte globulin (equine)
250 mg protein
50 mg/mL
ATTENTION — This product may contain a granular
or flaky deposit; this is normal. See infusion
instructions described in the enclosed product
insert.
For I.V. use only
Pfizer Injectables
Rx only
| ATGAM
equine thymocyte immune globulin injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Pharmacia and Upjohn Company (829076566) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Pharmacia and Upjohn Company | 829076566 | ANALYSIS(0009-7224), API MANUFACTURE(0009-7224), LABEL(0009-7224), MANUFACTURE(0009-7224), PACK(0009-7224) | |
