tazorac (Tazarotene) gel
[Allergan, Inc.]
FOR DERMATOLOGIC USE ONLY
NOT FOR OPHTHALMIC, ORAL OR INTRAVAGINAL USE
DESCRIPTION:
TAZORAC® Gel is a translucent, aqueous gel and contains the compound tazarotene, a member of the acetylenic class of retinoids. It is for topical dermatologic use only. The active ingredient is represented by the following structural formula:
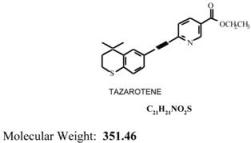
Chemical Name:
Ethyl 6-[2-(4,4-dimethylthiochroman-6-yl) ethynyl] nicotinate
Contains:
-
Active: Tazarotene................................... 0.05% or 0.1% (w/w)
Preservative: Benzyl alcohol.................................. 1.0% (w/w)
Inactives: Ascorbic acid, butylated hydroxyanisole, butylated hydroxytoluene, carbomer 934P, edetate disodium, hexylene glycol, poloxamer 407, polyethylene glycol 400, polysorbate 40, purified water, and tromethamine.
CLINICAL PHARMACOLOGY:
Tazarotene is a retinoid prodrug which is converted to its active form, the cognate carboxylic acid of tazarotene (AGN 190299), by rapid deesterification in animals and man. AGN 190299 (“tazarotenic acid”) binds to all three members of the retinoic acid receptor (RAR) family: RARα, RARβ, and RARγ but shows relative selectivity for RARβ, and RARγ and may modify gene expression. The clinical significance of these findings is unknown.
Psoriasis: The mechanism of tazarotene action in psoriasis is not defined. Topical tazarotene blocks induction of mouse epidermal ornithine decarboxylase (ODC) activity, which is associated with cell proliferation and hyperplasia. In cell culture and in vitro models of skin, tazarotene suppresses expression of MRP8, a marker of inflammation present in the epidermis of psoriasis patients at high levels. In human keratinocyte cultures, it inhibits cornified envelope formation, whose build-up is an element of the psoriatic scale. Tazarotene also induces the expression of a gene which may be a growth suppressor in human keratinocytes and which may inhibit epidermal hyperproliferation in treated plaques. However, the clinical significance of these findings is unknown.
Acne: The mechanism of tazarotene action in acne vulgaris is not defined. However, the basis of tazarotene's therapeutic effect in acne may be due to its anti-hyperproliferative, normalizing-of-differentiation and anti-inflammatory effects. Tazarotene inhibited corneocyte accumulation in rhino mouse skin and cross-linked envelope formation in cultured human keratinocytes. The clinical significance of these findings is unknown.
Pharmacokinetics:
Following topical application, tazarotene undergoes esterase hydrolysis to form its active metabolite, tazarotenic acid. Little parent compound could be detected in the plasma. Tazarotenic acid was highly bound to plasma proteins (>99%). Tazarotene and tazarotenic acid were metabolized to sulfoxides, sulfones and other polar metabolites which were eliminated through urinary and fecal pathways. The half-life of tazarotenic acid was approximately 18 hours, following topical application of tazarotene to normal, acne or psoriatic skin.
The human in vivo studies described below were conducted with tazarotene gel applied topically at approximately 2 mg/cm2 and left on the skin for 10 to 12 hours. Both the peak plasma concentration (Cmax) and area under the plasma concentration time curve (AUC) refer to the active metabolite only.
Two single, topical dose studies were conducted using 14C-tazarotene gel. Systemic absorption, as determined from radioactivity in the excreta, was less than 1% of the applied dose (without occlusion) in six psoriatic patients and approximately 5% of the applied dose (under occlusion) in six healthy subjects. One non-radiolabeled single-dose study comparing the 0.05% gel to the 0.1% gel in healthy subjects indicated that the Cmax and AUC were 40% higher for the 0.1% gel.
After 7 days of topical dosing with measured doses of tazarotene 0.1% gel on 20% of the total body surface without occlusion in 24 healthy subjects, the Cmax for tazarotenic acid was 0.72 ± 0.58 ng/mL (mean ± SD) occurring 9 hours after the last dose, and the AUC0-24hr for tazarotenic acid was 10.1 ± 7.2 ng·hr/mL. Systemic absorption was 0.91 ± 0.67% of the applied dose.
In a 14-day study in five psoriatic patients, measured doses of tazarotene 0.1% gel were applied daily by nursing staff to involved skin without occlusion (8 to 18% of total body surface area; mean ± SD: 13 ± 5%). The Cmax for tazarotenic acid was 12.0 ± 7.6 ng/mL occurring 6 hours after the final dose, and the AUC0-24hr for tazarotenic acid was 105 ± 55 ng·hr/mL. Systemic absorption was 14.8 ± 7.6% of the applied dose. Extrapolation of these results to represent dosing on 20% of total body surface yielded estimates for tazarotenic acid with Cmax of 18.9 ± 10.6 ng/mL and AUC0-24hr of 172 ± 88 ng·hr/mL.
An in vitro percutaneous absorption study, using radiolabeled drug and freshly excised human skin or human cadaver skin, indicated that approximately 4 to 5% of the applied dose was in the stratum corneum (tazarotene: tazarotenic acid= 5:1) and 2 to 4% was in the viable epidermis-dermis layer (tazarotene: tazarotenic acid= 2:1) 24 hours after topical application of the gel.
Clinical Studies:
Psoriasis:
In two large vehicle-controlled clinical studies, tazarotene 0.05% and 0.1% gels applied once daily for 12 weeks were significantly more effective than vehicle in reducing the severity of the clinical signs of stable plaque psoriasis covering up to 20% of body surface area. In one of the studies, patients were followed up for an additional 12 weeks following cessation of therapy with TAZORAC® Gel. Mean baseline scores and changes from baseline (reductions) after treatment in these two studies are shown in the following Table:
| TAZORAC® 0.05% Gel | TAZORAC® 0.1% Gel | Vehicle Gel | |||||||||||
| Trunk/Arm/ Leg lesions | Knee/Elbow lesions | Trunk/Arm/ Leg lesions | Knee/Elbow lesions | Trunk/Arm/ Leg lesions | Knee/Elbow lesions |
||||||||
| N=108 | N=111 | N=108 | N=111 | N=108 | N=112 | N=108 | N=112 | N=108 | N=113 | N=108 | N=113 | ||
|
Plaque elevation, scaling and erythema scored on a 0-4 scale with 0=none, 1=mild, 2=moderate, 3=severe and 4=very severe. |
|||||||||||||
|
*B=Mean Baseline Severity: C-12=Mean Change from Baseline at end of 12 weeks of therapy: |
|||||||||||||
|
C-24=Mean Change from Baseline at week 24 (12 weeks after the end of therapy). |
|||||||||||||
| Plaque elevation | B* C-12* C-24* | 2.5
-1.4 -1.2 | 2.6
-1.3 | 2.6
-1.3 -1.1 | 2.6
-1.1 | 2.5
-1.4 -1.1 | 2.6
-1.4 | 2.6
-1.5 -1.0 | 2.6
-1.3 | 2.4
-0.8 -0.9 | 2.6
-0.7 | 2.6
-0.7 -0.7 | 2.6
-0.6 |
| Scaling | B* C-12* C-24* | 2.4
-1.1 -0.9 | 2.5
-1.1 | 2.5
-1.1 -0.8 | 2.6
-0.9 | 2.4
-1.3 -1.0 | 2.6
-1.3 | 2.5
-1.2 -0.8 | 2.7
-1.2 | 2.4
-0.7 -0.8 | 2.6
-0.7 | 2.5
-0.6 -0.7 | 2.7
-0.6 |
| Erythema | B* C-12* C-24* | 2.4
-1.0 -1.1 | 2.7
-0.8 | 2.2
-0.9 -0.7 | 2.5
-0.8 | 2.4
-1.0 -0.9 | 2.8
-1.1 | 2.3
-1.0 -0.8 | 2.5
-0.8 | 2.3
-0.6 -0.7 | 2.7
-0.5 | 2.2
-0.5 -0.6 | 2.5
-0.5 |
Global improvement over baseline at the end of 12 weeks of treatment in these two studies is shown in the following Table:
| TAZORAC® 0.05% Gel | TAZORAC® 0.1% Gel | Vehicle Gel | ||||
| N=81 | N=93 | N=79 | N=69 | N=84 | N=91 | |
| 100% improvement | 2 (2%) | 1(1%) | 0 | 0 | 1 (1%) | 0 |
| ≥75% improvement | 23 (28%) | 17 (18%) | 30 (38%) | 17 (25%) | 10 (12%) | 9 (10%) |
| ≥50% improvement | 42 (52%) | 39 (42%) | 51 (65%) | 36 (52%) | 28 (33%) | 21 (23%) |
| 1-49% improvement | 21 (26%) | 32 (34%) | 18 (23%) | 23 (33%) | 27 (32%) | 32 (35%) |
| No change or worse | 18 (22%) | 22 (24%) | 10 (13%) | 10 (14%) | 29 (35%) | 38 (42%) |
The 0.1% gel was more effective than the 0.05% gel, but the 0.05% gel was associated with less local irritation than the 0.1% gel (see ADVERSE REACTIONS section).
Acne:
In two large vehicle-controlled studies, tazarotene 0.1% gel applied once daily was significantly more effective than vehicle in the treatment of facial acne vulgaris of mild to moderate severity. Percent reductions in lesion counts after treatment for 12 weeks in these two studies are shown in the following Table:
| TAZORAC® 0.1% Gel | Vehicle Gel | |||
| N=150 | N=149 | N=148 | N=149 | |
| Noninflammatory lesions | 55% | 43% | 35% | 27% |
| Inflammatory lesions | 42% | 47% | 30% | 28% |
| Total lesions | 52% | 45% | 33% | 27% |
Global improvement over baseline at the end of 12 weeks of treatment in these two studies is shown in the following Table:
| TAZORAC® 0.1% Gel | Vehicle Gel | |||
| N=105 | N=117 | N=117 | N=110 | |
| 100% improvement | 1 (1%) | 0 | 0 | 0 |
| ≥75% improvement | 40 (38%) | 21 (18%) | 23 (20%) | 11 (10%) |
| ≥50% improvement | 71 (68%) | 56 (48%) | 47 (40%) | 32 (29%) |
| 1-49% improvement | 23 (22%) | 49 (42%) | 48 (41%) | 46 (42%) |
| No change or worse | 11 (10%) | 12 (10%) | 22 (19%) | 32 (29%) |
INDICATIONS AND USAGE:
TAZORAC® (tazarotene) Gel 0.05% and 0.1% are indicated for the topical treatment of patients with stable plaque psoriasis of up to 20% body surface area involvement.
TAZORAC® (tazarotene) Gel 0.1% is also indicated for the topical treatment of patients with facial acne vulgaris of mild to moderate severity.
The efficacy of TAZORAC® Gel in the treatment of acne previously treated with other retinoids or resistant to oral antibiotics has not been established.
CONTRAINDICATIONS:
Retinoids may cause fetal harm when administered to a pregnant woman.
In rats, tazarotene 0.05% gel, administered topically during gestation days 6 through 17 at 0.25 mg/kg/day (1.5 mg/m2/day) resulted in reduced fetal body weights and reduced skeletal ossification. Rabbits dosed topically with 0.25 mg/kg/day (2.75 mg/m2 total body surface area/day) tazarotene gel during gestation days 6 through 18 were noted with single incidences of known retinoid malformations, including spina bifida, hydrocephaly, and heart anomalies. Systemic daily-exposure (AUCde) to tazarotenic acid at topical doses of 0.25 mg/kg/day tazarotene in a gel formulation in rats and rabbits represented 0.62 and 6.7 times, respectively, the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 0.78 and 8.4 times, respectively, the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15% (targeted) body surface area.
As with other retinoids, when tazarotene was given orally to experimental animals, developmental delays were seen in rats, and teratogenic effects and post-implantation loss were observed in rats and rabbits at AUCde values that were 0.55 and 13.2 times, respectively, the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 0.68 and 16.4 times, respectively, the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15% (targeted) body surface area.
In a study of the effect of oral tazarotene on fertility and early embryonic development in rats, decreased number of implantation sites, decreased litter size, decreased numbers of live fetuses, and decreased fetal body weights, all classic developmental effects of retinoids, were observed when female rats were administered 2 mg/kg/day from 15 days before mating through gestation day 7. A low incidence of retinoid-related malformations at that dose was reported to be related to treatment. This dose produced an AUCde that was 1.7 times the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area) and 2.1 times the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15% (targeted) body surface area.
SYSTEMIC EXPOSURE TO TAZAROTENIC ACID IS DEPENDENT UPON THE EXTENT OF THE BODY SURFACE AREA TREATED. IN PATIENTS TREATED TOPICALLY OVER SUFFICIENT BODY SURFACE AREA, EXPOSURE COULD BE IN THE SAME ORDER OF MAGNITUDE AS IN THESE ORALLY TREATED ANIMALS. ALTHOUGH THERE MAY BE LESS SYSTEMIC EXPOSURE IN THE TREATMENT OF ACNE OF THE FACE ALONE DUE TO LESS SURFACE AREA FOR APPLICATION, TAZAROTENE IS A TERATOGENIC SUBSTANCE, AND IT IS NOT KNOWN WHAT LEVEL OF EXPOSURE IS REQUIRED FOR TERATOGENICITY IN HUMANS (SEE CLINICAL PHARMACOLOGY: PHARMACOKINETICS).
There were thirteen reported pregnancies in patients who participated in clinical trials for topical tazarotene. Nine of the patients were found to have been treated with topical tazarotene, and the other four had been treated with vehicle. One of the patients who was treated with tazarotene cream elected to terminate the pregnancy for non-medical reasons unrelated to treatment. The other eight pregnant women who were inadvertently exposed to topical tazarotene during clinical trials subsequently delivered apparently healthy babies. As the exact timing and extent of exposure in relation to the gestation times are not certain, the significance of these findings is unknown.
TAZORAC® Gel is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, treatment should be discontinued and the patient apprised of the potential hazard to the fetus. Women of child-bearing potential should be warned of the potential risk and use adequate birth-control measures when TAZORAC® Gel is used. The possibility that a woman of childbearing potential is pregnant at the time of institution of therapy should be considered. A negative result for pregnancy test having a sensitivity down to at least 50 mIU/mL for human chorionic gonadotropin (hCG) should be obtained within 2 weeks prior to TAZORAC® Gel therapy, which should begin during a normal menstrual period (see also PRECAUTIONS: Pregnancy: Teratogenic Effects).
TAZORAC® Gel is contraindicated in individuals who have shown hypersensitivity to any of its components.
WARNINGS:
Pregnancy Category X. See CONTRAINDICATIONS section. Women of child-bearing potential should be warned of the potential risk and use adequate birth-control measures when TAZORAC® Gel is used. The possibility that a woman of childbearing potential is pregnant at the time of institution of therapy should be considered. A negative result for pregnancy test having a sensitivity down to at least 50 mIU/mL for hCG should be obtained within 2 weeks prior to TAZORAC® Gel therapy, which should begin during a normal menstrual period.
PRECAUTIONS:
General:
TAZORAC® Gel should be applied only to the affected areas. For external use only. Avoid contact with eyes, eyelids, and mouth. If contact with eyes occurs, rinse thoroughly with water. The safety of use of TAZORAC® Gel over more than 20% of body surface area has not been established in psoriasis or acne.
Retinoids should not be used on eczematous skin, as they may cause severe irritation.
Because of heightened burning susceptibility, exposure to sunlight (including sunlamps) should be avoided unless deemed medically necessary, and in such cases, exposure should be minimized during the use of TAZORAC® Gel. Patients must be warned to use sunscreens (minimum SPF of 15) and protective clothing when using TAZORAC® Gel. Patients with sunburn should be advised not to use TAZORAC® Gel until fully recovered. Patients who may have considerable sun exposure due to their occupation and those patients with inherent sensitivity to sunlight should exercise particular caution when using TAZORAC® Gel and ensure that the precautions outlined in the Information for Patients subsection are observed.
TAZORAC® Gel should be administered with caution if the patient is also taking drugs known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the increased possibility of augmented photosensitivity.
Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, the medication should either be discontinued until the integrity of the skin is restored, or the dosing should be reduced to an interval the patient can tolerate. However, efficacy at reduced frequency of application has not been established. Alternatively, patients with psoriasis who are being treated with the 0.1% concentration can be switched to the lower concentration.
Weather extremes, such as wind or cold, may be more irritating to patients using TAZORAC® Gel.
Information for Patients:
See attached Patient Package Insert.
Drug Interactions:
Concomitant dermatologic medications and cosmetics that have a strong drying effect should be avoided. It is also advisable to "rest" a patient's skin until the effects of such preparations subside before use of TAZORAC® Gel is begun.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
A long-term study of tazarotene following oral administration of 0.025, 0.050, and 0.125 mg/kg/day to rats showed no indications of increased carcinogenic risks. Based on pharmacokinetic data from a shorter-term study in rats, the highest dose of 0.125 mg/kg/day was anticipated to give systemic exposure (AUCde) in the rat equivalent to 0.32 times the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 0.38 times the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15% (targeted) body surface area.
In evaluation of photo co-carcinogenicity, median time to onset of tumors was decreased, and the number of tumors increased in hairless mice following chronic topical dosing with intercurrent exposure to ultraviolet radiation at tazarotene concentrations of 0.001%, 0.005%, and 0.01% in a gel formulation for up to 40 weeks.
A long-term topical application study of up to 0.1% tazarotene in a gel formulation in mice terminated at 88 weeks showed that dose levels of 0.05, 0.125, 0.25, and 1.0 mg/kg/day (reduced to 0.5 mg/kg/day for males after 41 weeks due to severe dermal irritation) revealed no apparent carcinogenic effects when compared to vehicle control animals; untreated control animals were not completely evaluated. Systemic exposure (AUC0-12h) at the highest dose was 2.0 times the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 2.5 times the maximum AUC0-24h in acne patients treated with 2 mg/cm 2 of tazarotene gel 0.1% over 15 % (targeted) body surface area.
Tazarotene was found to be non-mutagenic in the Ames assay using Salmonella and E. coli and did not produce structural chromosomal aberrations in a human lymphocyte assay. Tazarotene was also non-mutagenic in the CHO/HGPRT mammalian cell forward gene mutation assay and was non-clastogenic in the in vivo mouse micronucleus test.
No impairment of fertility occurred in rats when male animals were treated for 70 days prior to mating and female animals were treated for 14 days prior to mating and continuing through gestation and lactation with topical doses of tazarotene gel up to 0.125 mg/kg/day. Based on data from another study, the systemic drug exposure (AUCde) in the rat would be equivalent to 0.31 times the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 0.38 times the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15 % (targeted) body surface area.
No impairment of mating performance or fertility was observed in male rats treated for 70 days prior to mating with oral doses of up to 1.0 mg/kg/day tazarotene, which produced an AUCde that was 0.95 times the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 1.2 times the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15 % (targeted) body surface area.
No effect on parameters of mating performance or fertility was observed in female rats treated for 15 days prior to mating and continuing through day 7 of gestation with oral doses of tazarotene up to 2.0 mg/kg/day. However, there was a significant decrease in the number of estrous stages and an increase in developmental effects at 2.0 mg/kg/day (see CONTRAINDICATIONS). This dose produced an AUC0-24h which was 1.7 times that observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 2.1 times the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15% (targeted) body surface area.
Reproductive capabilities of F1 animals, including F2 survival and development, were not affected by topical administration of tazarotene gel to female F0 parental rats from gestation day 16 through lactation day 20 at the maximum tolerated dose of 0.125 mg/kg/day. Based on data from another study, the systemic drug exposure (AUCde) in the rat would be equivalent to 0.31 times the AUC0-24h observed in psoriatic patients treated with 2 mg/cm2 of tazarotene gel 0.1% (extrapolated for topical application over 20% body surface area), and 0.38 times the maximum AUC0-24h in acne patients treated with 2 mg/cm2 of tazarotene gel 0.1% over 15% (targeted) body surface area.
Pregnancy:
Teratogenic Effects: Pregnancy Category X: See CONTRAINDICATIONS section. Women of child-bearing potential should use adequate birth-control measures when TAZORAC® Gel is used. The possibility that a woman of childbearing potential is pregnant at the time of institution of therapy should be considered. A negative result for pregnancy test having a sensitivity down to at least 50 mIU/mL for hCG should be obtained within 2 weeks prior to TAZORAC® Gel therapy, which should begin during a normal menstrual period. There are no adequate, well-controlled studies in pregnant women. Although there may be less systemic exposure in the treatment of acne of the face alone due to less surface area for application, tazarotene is a teratogenic substance , and it is not known what level of exposure is required for teratogenicity in humans (see CLINICAL PHARMACOLOGY: Pharmacokinetics).
Nursing mothers:
After single topical doses of 14C-tazarotene to the skin of lactating rats, radioactivity was detected in milk, suggesting that there would be transfer of drug-related material to the offspring via milk. It is not known whether this drug is excreted in human milk. Caution should be exercised when tazarotene is administered to a nursing woman.
Pediatric Use:
The safety and efficacy of tazarotene have not been established in pediatric patients under the age of 12 years.
Geriatric Use:
Of the total number of subjects in clinical studies of tazarotene gels, 0.05% and 0.1% for plaque psoriasis, 163 were over the age of 65. Subjects over 65 years of age experienced more adverse events and lower treatment success rates after 12 weeks of use of TAZORAC® Gel compared with those 65 years of age and younger. Currently there is no other reliable clinical experience on the differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals can not be ruled out. Tazarotene gel for the treatment of acne has not been clinically evaluated in persons over the age of 65.
ADVERSE REACTIONS:
In human dermal safety studies, tazarotene 0.05% and 0.1% gels did not induce allergic contact sensitization, phototoxicity or photoallergy.
Psoriasis:
The most frequent adverse events reported with TAZORAC® Gel 0.05% and 0.1% were limited to the skin. Those occurring in 10 to 30% of patients, in descending order, included pruritus, burning/stinging, erythema, worsening of psoriasis, irritation, and skin pain. Events occurring in 1 to 10% of patients included rash, desquamation, irritant contact dermatitis, skin inflammation, fissuring, bleeding and dry skin. Increases in “psoriasis worsening” and “sun-induced erythema” were noted in some patients over the 4th to 12th months as compared to the first three months of a 1 year study. In general, the incidence of adverse events with TAZORAC® Gel 0.05% was 2 to 5% lower than that seen with TAZORAC® Gel 0.1%.
Acne:
The most frequent adverse events reported with TAZORAC® Gel 0.1% in the treatment of acne occurring in 10 to 30% of patients, in descending order, included desquamation, burning/stinging, dry skin, erythema and pruritus. Events occurring in 1 to 10% of patients included irritation, skin pain, fissuring, localized edema and skin discoloration.
OVERDOSAGE:
Excessive topical use of TAZORAC® Gel may lead to marked redness, peeling, or discomfort (see PRECAUTIONS: General).
TAZORAC® Gels 0.05% and 0.1% are not for oral use. Oral ingestion of the drug may lead to the same adverse effects as those associated with excessive oral intake of Vitamin A (hypervitaminosis A) or other retinoids. If oral ingestion occurs, the patient should be monitored, and appropriate supportive measures should be administered as necessary.
DOSAGE AND ADMINISTRATION:
General:
Application may cause excessive irritation in the skin of certain sensitive individuals. In cases where it has been necessary to temporarily discontinue therapy, or the dosing has been reduced to a lower concentration (in patients with psoriasis) or to an interval the patient can tolerate, therapy can be resumed, or the drug concentration or frequency of application can be increased as the patient becomes able to tolerate the treatment. Frequency of application should be closely monitored by careful observation of the clinical therapeutic response and skin tolerance. Efficacy has not been established for less than once daily dosing frequencies.
For psoriasis: It is recommended that treatment start with TAZORAC® 0.05% Gel, with strength increased to 0.1% if tolerated and medically indicated. Apply TAZORAC® Gel once a day, in the evening, to psoriatic lesions, using enough (2 mg/cm2) to cover only the lesion with a thin film to no more than 20% of body surface area. If a bath or shower is taken prior to application, the skin should be dry before applying the gel. If emollients are used, they should be applied at least an hour before application of TAZORAC® Gel. Because unaffected skin may be more susceptible to irritation, application of tazarotene to these areas should be carefully avoided. TAZORAC® Gel was investigated for up to 12 months during clinical trials for psoriasis.
For acne: Cleanse the face gently. After the skin is dry, apply a thin film of TAZORAC® Gel 0.1% (2 mg/cm2) once a day, in the evening, to the skin where acne lesions appear. Use enough to cover the entire affected area. TAZORAC® Gel was investigated for up to 12 weeks during clinical trials for acne.
HOW SUPPLIED:
TAZORAC® (tazarotene) Gel is available in concentrations of 0.05% and 0.1%. It is available in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white propylene screw cap, in 30 gm and 100 gm sizes.
| TAZORAC® Gel 0.05% | TAZORAC® Gel 0.1% | |
| 30 gm | NDC 0023-8335-03 | NDC 0023-0042-03 |
| 100 gm | NDC 0023-8335-10 | NDC 0023-0042-10 |
NOTE: TAZORAC® (tazarotene) Gel should be stored at 25°C (77°F): excursion permitted to 15-30°C (59-86°F).
RX ONLY
® Marks owned by Allergan, Inc.
U.S. Patent Numbers 5,089,509; 5,914,334 and 6,258,830
ALLERGAN
Irvine, California 92612, USA
©2004 Allergan, Inc.
Printed in USA
8606X, 8607XA
January 2004
71722US10P
Pharmacist: Please cut or tear at dotted line and provide this patient package insert to your customer
TAZORAC®
(tazarotene) Gel 0.05%
(tazarotene) Gel 0.1%
INFORMATION FOR PATIENTS
Please read this leaflet carefully before you start to use your medicine. Read the information you get every time you get more medicine. There may be new information about the drug. This leaflet does not take the place of talks with your doctor. If you have any questions, or are not sure about anything, ask your doctor or pharmacist.
USES
TAZORAC® Gel 0.05% is used in the treatment of stable plaque psoriasis covering up to 20% of body surface area.
TAZORAC® Gel 0.1% is used in the treatment of stable plaque psoriasis covering up to 20% of body surface area and in the treatment of mild to moderately severe facial acne.
BEFORE YOU USE THIS MEDICINE
You should be aware that:
- TAZORAC® Gel should not be used if you are pregnant, attempting to become pregnant or at high risk of pregnancy. Consult your physician for adequate birth control measures if you are a female of child-bearing potential. If you are able to become pregnant, take a pregnancy test within 2 weeks prior to beginning to use TAZORAC® Gel to be sure you are not pregnant. If you have menstrual periods, begin taking TAZORAC® Gel during a normal menstrual period. These actions help assure that you are not pregnant when you begin use.
- TAZORAC® Gel should be used with caution if you are also using other topical agents with a strong skin drying effect, products with high concentrations of alcohol, astringents, spices, the peel of lime, medicated soaps or shampoos, permanent wave solutions, electrolysis, hair depilatories or waxes, or other preparations or processes that might dry or irritate the skin, unless otherwise instructed by your health care practitioner.
- TAZORAC® Gel should not be used if you have sunburn, eczema or other chronic skin condition(s). TAZORAC® Gel may cause severe irritation if applied to eczematous skin. If you have sunburn, you should wait until full recovery before using TAZORAC® Gel.
- TAZORAC® Gel should not be used if you are inherently sensitive to sunlight.
- TAZORAC® Gel should not be used if you are taking other drugs that increase your sensitivity to sunlight (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides). Inform your physician if you are taking any other medications.
- You should use protective clothing and sunscreens with minimum SPF of 15 during the day when being treated with TAZORAC® Gel . You should avoid direct sun exposure as much as possible and avoid sunlamps totally while being treated with TAZORAC® Gel, unless advised otherwise by your doctor.
- If you have considerable sun exposure due to occupation, particular caution as described above should be exercised when using TAZORAC® Gel.
- Weather extremes, such as wind or cold, may be more irritating to your skin while you are using TAZORAC® Gel.
BEFORE YOU USE THIS MEDICINE
Tell your doctor:
- if you are pregnant or are considering becoming pregnant.
- if you are breast-feeding. We do not know if tazarotene can pass through the milk to the baby. The potential harm to the baby is unknown.
- if you are allergic to any ingredients in this medicine.
- if you are already using other products that make your skin dry.
- if you have a skin condition called eczema.
- if you will be subject to excessive sun exposure.
- if you are taking vitamin A supplements.
HOW TO USE THIS PRODUCT:
- Read the directions on your prescription label carefully. Ask your doctor or pharmacist to explain anything that you do not understand.
- If you become pregnant while using TAZORAC® Gel, you should immediately discontinue its use and contact your doctor.
- If you use a cream or lotion to soften or lubricate your skin, apply TAZORAC® Gel after ensuring that there is no more cream or lotion on the skin.
- With use of TAZORAC® Gel, some people notice a feeling of itching, burning or stinging. If irritation is excessive, consult your health care provider, who may adjust your medication temporarily to a more comfortable level. Effectiveness of this medication when used less often than once daily has not been proven.
- Do not cover treated areas with dressings or bandages.
- Never use more TAZORAC® Gel than instructed and never use it more often than instructed, as application of larger amounts of medication than recommended will not lead to more rapid or better results, and marked redness, peeling or discomfort may occur.
- Wash your hands after applying the medication unless you are treating your hands for psoriasis. If the gel accidentally gets on areas you do not need to treat, wash it off.
- Keep TAZORAC® Gel away from your eyes, eyelids, and mouth. If it gets in contact with your eyes, wash them with large amounts of cool water, and contact a doctor if eye irritation persists.
MISSED DOSES:
- If you forget or miss a dose of TAZORAC® Gel, do not try to "make it up." Return to your normal application schedule as soon as you can.
INSTRUCTIONS SPECIFIC TO TREATMENT OF PSORIASIS:
- If you bathe or shower before using TAZORAC® Gel, be sure the skin is dry before application. Apply a thin film of the gel to your psoriasis lesions once a day before going to bed.
- Carefully avoid application to apparently uninvolved skin. TAZORAC® Gel may be more irritating to non-lesional skin.
- If you need to treat your hands, avoid contact with your eyes.
- Usually your psoriasis plaques and scales will begin to improve in about one to four weeks, but the redness may take longer to improve. Continue to use TAZORAC® Gel as directed by your doctor.
- Contact your doctor if your psoriasis becomes worse.
INSTRUCTIONS SPECIFIC TO TREATMENT OF ACNE:
- Gently clean and dry your face before using TAZORAC® Gel. Apply TAZORAC® Gel once a day, before going to bed, to entire areas of the face where you have acne lesions. Use enough gel to cover the entire affected area with a thin film.
- Follow your doctor's directions for other routine skin care and the use of make-up. Talk to your doctor about the use of sunscreens and cosmetics, especially those that dry your skin.
- Usually, your acne will begin to improve in about 4 weeks. Continue to use TAZORAC® Gel for up to 12 weeks as directed by your doctor.
- Contact your doctor if your acne becomes worse.
POSSIBLE SIDE EFFECTS OF TAZORAC®GEL
For patients with psoriasis, the most common side effects of TAZORAC® Gel 0.05% and 0.1% are itching, burning/stinging, red skin, worsening of psoriasis, irritation, and skin pain.
For patients with acne, the most common side effects of TAZORAC® Gel 0.1% are peeling, burning/stinging, dry skin, red skin, and itching.
Tell your doctor if these side effects become problems. Your doctor may wish to adjust your dose of TAZORAC® Gel. However, effectiveness of TAZORAC® Gel when used less often than once a day has not been proven.
While you take TAZORAC® Gel, weather extremes, such as wind or cold, may irritate your skin more than usual.
WARNINGS:
TAZORAC® Gel should not be used if you are allergic to any of its ingredients. TAZORAC® Gel contains tazarotene as the active ingredient, and the following inactive ingredients: ascorbic acid, benzyl alcohol, butylated hydroxyanisole, butylated hydroxytoluene, carbomer 934P, edetate disodium, hexylene glycol, poloxamer 407, polyethylene glycol 400, polysorbate 40, purified water, and tromethamine.
TAZORAC® Gel should not be used if you are pregnant, attempting to become pregnant or at high risk of pregnancy. Women of child-bearing potential should use adequate birth-control measures when TAZORAC® Gel is used.
If TAZORAC® Gel is swallowed by accident, contact your doctor or a poison control center.
Do not use TAZORAC® Gel after the expiration date found on the bottom seal of the tube.
This medicine is for your use only. It can only be prescribed by a doctor. Never give it to anyone else. It may harm them even if their skin problem appears to be the same as yours.
Retinoids should not be used on eczematous skin, as they may cause severe irritation. Do not use TAZORAC® Gel until your doctor has confirmed that your eczema has fully recovered.
Because of increased burning susceptibility, exposure to sunlight (including sunlamps) should be avoided or minimized during the use of TAZORAC® Gel, unless prescribed differently by your doctor.
You should use sunscreens (minimum SPF of 15) and protective clothing when using TAZORAC® Gel. Be certain that you use these precautions if you expect to experience considerable sun exposure or if you are sensitive to sunlight.
If you have a sunburn, do not use TAZORAC® Gel until you have fully recovered.
Do not use TAZORAC® Gel if you are also taking drugs known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides), unless you have discussed taking both drugs with your doctor, because of the increased possibility of a more severe reaction.
The safety of use over more than 20% of body surface area has not been established in psoriasis or acne.
INSTRUCTIONS FOR USE AND HANDLING: Keep tube tightly closed when not in use. Store it in a safe place where children cannot reach it. TAZORAC® Gel should be stored at 25°C (77°F): excursion permitted to 15-30 °C (59-86°F).
GENERAL ADVICE ABOUT PRESCRIPTION MEDICINES:
This medicine is for your use only. Never give it to other people. It may harm them even if their skin problem appears to be the same as yours. Do not use TAZORAC® Gel for a condition for which it was not prescribed. Do not use TAZORAC® Gel after the expiration date on the bottom seal of the tube.
WHERE CAN I GET MORE INFORMATION ABOUT TAZORAC®GEL?
You can contact Allergan by calling 800-433-8871. You can ask your doctor or pharmacist for the information about TAZORAC® Gel that is written for health professionals.
IF YOU HAVE QUESTIONS ABOUT PSORIASIS: Information is available from:
The National Psoriasis Foundation:
6600 SW 92nd Avenue, Suite 300, Portland, OR 97223-7195.
Telephone: (800) 723-9166, or on the World Wide Web at http://www.psoriasis.org.
® Marks owned by Allergan, Inc.
U.S. Patent Numbers 5,089,509; 5,914,334 and 6,258,830
ALLERGAN
Irvine, California 92612, USA
©2004 Allergan, Inc.
Printed in USA
8606X, 8607XA
January 2004
71722US10P
| TAZORAC (Tazarotene) | |||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
| TAZORAC (Tazarotene) | |||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
Revised: 05/2006