KELO-COTE HC ANTI-ITCH SCAR TREATMENT- hydrocortisone acetate cream
Advanced Bio-Technologies, Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
KELO-COTE HC Anti-Itch Scar Treatment
Uses
For the temporary relief of itching associated with minor skin irritations, inflammation and rashes due to eczema, insect bites, soaps, detergents and cosmetics.
Stop use and ask a doctor if
- condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days
Keep out of reach of children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Directions
- ensure that affected area is clean and dry - apply evenly in a very thin layer to affected area not more than 3 to 4 times daily - children under 2 years of age : consult a doctor
Inactive ingredients
Cyclopentasiloxane, Dimethicone, Dimethicone Crosspolymer, Dimethyl Isosorbide, Pentylene Glycol, Propylene Glycol, Silicon Dioxide
Description
Kelo-cote HC with 1% hydrocortisone acetate for itchy scars Anti-Itch Scare Treatment In a recent survey, 42% of respondents reported that their scars were itchy.3 In fact, itch is the most common symptom of keloid scars.4 Kelo-Cote HC was specifically developed with 1% hydrocortisone acetate to provide relief from itch caused by skin irritation and inflammation while treating your scar. The foundation of this unique gel is Kelo-cote's patented silicone technology, which self-dries to form a flexible, breathable waterproof layer. Clinical studies have demonstrated that Kelo-cote helps flatten, soften, and reduce redness associated with scars, as well as help to relieve scar-related itch, pain, and discomfort. Kelo-cote may also help in the prevention of excessive and abnormal scar formations such as keloids. It takes many months for your scar to completely mature. The combination of Kelo-cote's patented, proven silicone technology and the itch and inflammation relief provided by the 1% hydrocortisone acetate helps to create an optimal environment for scar maturation and an aesthetically pleasing end result. The convenience of a single product makes it simple to use. - Kelo-cote HC is ideal for new and old scars that itch - To help prevent abnormal scars from developing, use as soon as the wound is closed or sutures are removed - This 10g tube is enough for 90 days effective treatment and itch relief for a scar size of 3-4 inches/7.5-10cm US Patent No. 5,741,509 International Patents Manufactured in the USA for: ABT Advanced Bio-Technologies, Inc. Suwanee, GA 30024 USA 1 Radwanski H et. al. Rev Bras Cir Plast 2010; 25(3): 428-33. 2 Sepehrmanesh M. et. al. Komp Dermatol 2006; 30-32. 3 Data on file. 4 Shaffer JJ et. al. J Am Acad Dermatol. 2002 Feb;46 (2 Suppl Understanding): S63-97. C 2011 Advanced Bio-Technologies, Inc. 7 92734 00416 3
Manufactured in the USA for: ABT Advanced Bio-Technologies, Inc. Suwanee, GA 30024 USA - US Patent #5,741,509 and International Patents. Kelo-cote HC with 1% hydrocortisone acetate for itchy scars ABT Advanced Bio-Technologies, Inc. NET WT 10g Gel
Kelo-cote HC with 1% hydrocortisone acetate for itchy scars Anti-Itch Scare Treatment Manufactured in the USA for: ABT Advanced Bio-Technologies, Inc. Suwanee, GA 30024 USA C 2011 Advanced Bio-Technologies, Inc.
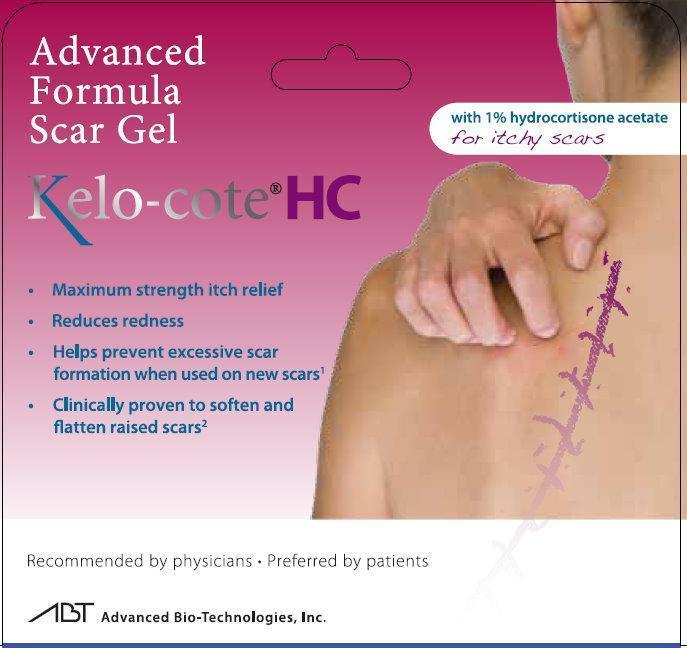
Advanced Formula Scar Gel Kelo-cote HC with 1% hydrocortisone acetate for itchy scars - Maximum strength itch relief - Reduces redness - Helps prevent excessive scar formation when used on new scars1 - Clinically proven to soften and flatten raised scars2 Recommended by physicians - Preferred by patients ABT Advanced Bio-Technologies, Inc.
| KELO-COTE HC ANTI-ITCH SCAR TREATMENT
hydrocortisone acetate cream |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Advanced Bio-Technologies, Inc (943947569) |
| Registrant - Advanced Bio-Technologies, Inc (943947569) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Formulated Solutions, LLC | 143266687 | manufacture(40101-001) | |