REGRANEX
-
becaplermin gel
Ortho-McNeil-Janssen Pharmaceuticals, Inc.
----------
REGRANEX® Gel 0.01%(becaplermin)
WARNING
An increased rate of mortality secondary to malignancy was observed in patients treated with 3 or more tubes of REGRANEX Gel in a post-marketing retrospective cohort study. REGRANEX Gel should only be used when the benefits can be expected to outweigh the risks. REGRANEX Gel should be used with caution in patients with known malignancy. (See CONTRAINDICATIONS and WARNINGS)
DESCRIPTION
REGRANEX® Gel contains becaplermin, a recombinant human platelet-derived growth factor (rhPDGF-BB) for topical administration. Becaplermin is produced by recombinant DNA technology by insertion of the gene for the B chain of platelet-derived growth factor (PDGF) into the yeast, Saccharomyces cerevisiae. Becaplermin has a molecular weight of approximately 25 KD and is a homodimer composed of two identical polypeptide chains that are bound together by disulfide bonds. Becaplermin Concentrate is produced by Novartis Vaccines and Diagnostics, Inc. and supplied to OMJ Pharmaceuticals under a shared manufacturing arrangement. REGRANEX Gel is a non-sterile, low bioburden, preserved, sodium carboxymethylcellulose-based (CMC) topical gel, containing the active ingredient becaplermin and the following inactive ingredients: sodium chloride, sodium acetate trihydrate, glacial acetic acid, water for injection, and methylparaben, propylparaben, and m-cresol as preservatives and l-lysine hydrochloride as a stabilizer. Each gram of REGRANEX Gel contains 100 µg of becaplermin.
CLINICAL PHARMACOLOGY
REGRANEX Gel has biological activity similar to that of endogenous platelet-derived growth factor, which includes promoting the chemotactic recruitment and proliferation of cells involved in wound repair and enhancing the formation of granulation tissue.
Pharmacokinetics
Ten patients with Stage III or IV (as defined in the International Association of Enterostomal Therapy (IAET) guide to chronic wound staging, J. Enterostomal Ther 15:4, 1988 and Decubitis 2:24, 1989) lower extremity diabetic ulcers received topical applications of becaplermin gel 0.01% at a dose range of 0.32–2.95 µg/kg (7µg/cm2) daily for 14 days. Six patients had non-quantifiable PDGF levels at baseline and throughout the study, two patients had PDGF levels at baseline which did not increase substantially, and two patients had PDGF levels that increased sporadically above their baseline values during the 14 day study period.
Systemic bioavailability of becaplermin was less than 3% in rats with full thickness wounds receiving single or multiple (5 days) topical applications of 127 µg/kg (20.1 µg/cm2 of wound area) of becaplermin gel.
Clinical Studies
The effects of REGRANEX Gel on the incidence of and time to complete healing in lower extremity diabetic ulcers were assessed in four randomized controlled studies. Of 922 patients studied, 478 received either REGRANEX Gel 0.003% or 0.01%. All study participants had lower extremity diabetic neuropathic ulcers that extended into the subcutaneous tissue or beyond (Stages III and IV of the IAET guide to chronic wound staging). Ninety-three percent of the patients enrolled in these four trials had foot ulcers. The remaining 7% of the patients had ankle or leg ulcers. The diabetic ulcers were of at least 8 weeks duration and had an adequate blood supply (defined as TcpO2 > 30 mm Hg). In the four trials, ninety-five percent of the ulcers measured in area up to 10 cm2, and the median ulcer size at baseline ranged from 1.4 cm2 to 3.5 cm2. All treatment groups received a program of good ulcer care consisting of initial complete sharp debridement, a non-weight-bearing regimen, systemic treatment for wound-related infection if present, moist saline dressings changed twice a day, and additional debridement as necessary. REGRANEX Gel 0.003% or 0.01% or placebo gel was applied once a day and covered with a saline moistened dressing. After approximately 12 hours, the gel was gently rinsed off and a saline moistened dressing was then applied for the remainder of the day. Patients were treated until complete healing, or for a period of up to 20 weeks. Patients were considered a treatment failure if their ulcer did not show an approximately 30% reduction in initial ulcer area after eight to ten weeks of REGRANEX Gel therapy.
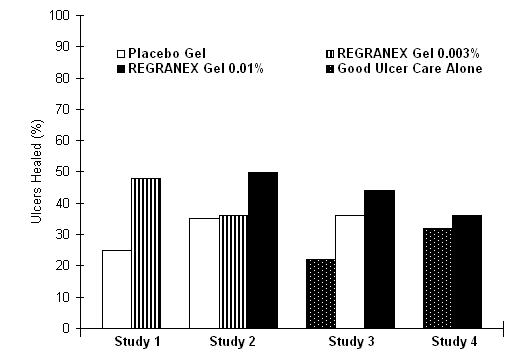
The primary endpoint, incidence of complete ulcer closure within 20 weeks, for all treatment arms is shown in Figure 1. In each study, REGRANEX Gel in conjunction with good ulcer care was compared to placebo gel plus good ulcer care or good ulcer care alone.
In Study 1, a multicenter, double-blind, placebo controlled trial of 118 patients, the incidence of complete ulcer closure for REGRANEX Gel 0.003% (n=61) was 48% versus 25% for placebo gel (n=57; p=0.02, logistic regression analysis).
In Study 2, a multicenter, double-blind, placebo controlled trial of 382 patients, the incidence of complete ulcer closure for REGRANEX Gel 0.01% (n=123), was 50% versus 36% for REGRANEX Gel 0.003% (n=132), and 35% for placebo gel (n=127). Only REGRANEX Gel 0.01% was significantly different from placebo gel (p=0.01, logistic regression analysis).
The primary goal of Study 3, a multicenter controlled trial of 172 patients, was to assess the safety of vehicle gel (placebo; n=70) compared to good ulcer care alone (n=68). The study included a small (n=34) REGRANEX Gel 0.01% arm. Incidences of complete ulcer closure were 44% for REGRANEX Gel, 36% for placebo gel and 22% for good ulcer care alone.
In Study 4, a multicenter, evaluator-blind, controlled trial of 250 patients, the incidences of complete ulcer closure in the REGRANEX Gel 0.01% arm (n=128) (36%) and good ulcer care alone (n=122) (32%) were not statistically different.
| Figure 1: Incidence of Complete Healing |
 |
In general, where REGRANEX Gel was associated with higher incidences of complete ulcer closure, differences in the incidence first became apparent after approximately 10 weeks and increased with continued treatment (Table 1).
| REGRANEX Gel 0.01% (%) | Placebo Gel (%) |
|
|---|---|---|
| Week 2 | 1 | 0 |
| Week 4 | 6 | 2 |
| Week 6 | 9 | 6 |
| Week 8 | 16 | 14 |
| Week 10 | 23 | 18 |
| Week 12 | 34 | 25 |
| Week 14 | 37 | 28 |
| Week 16 | 43 | 33 |
| Week 18 | 46 | 34 |
| Week 20 | 50 | 37 |
In a 3-month follow-up period where no standardized regimen of preventative care was utilized, the incidence of ulcer recurrence was approximately 30% in all treatment groups, demonstrating that the durability of ulcer closure was comparable in all treatment groups.
In a randomized, double-blind study of REGRANEX Gel (100 mcg/g once daily for 16 weeks) in patients with Stage III or IV pressure ulcers, the incidence of complete ulcer closure was 15% (28/189) in the becaplermin group and 12% (22/190) in the vehicle control group. This difference was not statistically significant.
In two small, randomized, double-blinded studies of REGRANEX Gel (100mcg/g once daily for 16 weeks) in patients with venous stasis ulcers, the combined incidence of complete ulcer closure was 46% (30/65) in the becaplermin group and 39% (26/67) in the vehicle control group. This difference was not statistically significant.
INDICATIONS AND USAGE
REGRANEX (becaplermin) Gel is indicated for the treatment of lower extremity diabetic neuropathic ulcers that extend into the subcutaneous tissue or beyond and have an adequate blood supply. When used as an adjunct to, and not a substitute for, good ulcer care practices including initial sharp debridement, pressure relief and infection control, REGRANEX Gel increases the incidence of complete healing of diabetic ulcers.
The efficacy of REGRANEX Gel has not been established for the treatment of pressure ulcers and venous stasis ulcers (see Clinical Studies) and has not been evaluated for the treatment of diabetic neuropathic ulcers that do not extend through the dermis into subcutaneous tissue (Stage I or II, IAET staging classification) or ischemic diabetic ulcers.
CONTRAINDICATIONS
REGRANEX (becaplermin) Gel is contraindicated in patients with:
- –
- known hypersensitivity to any component of this product (e.g., parabens);
- –
- known neoplasm(s) at the site(s) of application.
WARNINGS
REGRANEX Gel contains becaplermin, a recombinant human platelet-derived growth factor, which promotes cellular proliferation and angiogenesis. (See Clinical Pharmacology). The benefits and risks of becaplermin treatment should be carefully evaluated before prescribing. Becaplermin should be used with caution in patients with a known malignancy.
Malignancies distant from the site of application have occurred in becaplermin users in both a clinical study and in postmarketing use, and an increased rate of death from systemic malignancies was seen in patients who have received 3 or more tubes of REGRANEX Gel.
In a follow-up study, 491 (75%) of 651 subjects from two randomized, controlled trials of becaplermin gel 0.01% were followed for a median of approximately 20 months to identify malignancies diagnosed after the end of the trials. Eight of 291 subjects (3%) from the becaplermin group and two of 200 subjects (1%) from the vehicle/standard of care group were diagnosed with cancers during the follow-up period, a relative risk of 2.7, (95% confidence interval 0.6–12.8). The types of cancers varied and all were remote from the treatment site.
In a retrospective study of a medical claims database, cancer rates and overall cancer mortality were compared between 1,622 patients who used REGRANEX Gel and 2,809 matched comparators. Estimates of the incidence rates reported below may be under-reported due to limited follow-up for each individual.
- The incidence rate for all cancers was 10.2 per 1,000 person years for patients treated with REGRANEX Gel and 9.1 per 1,000 person years for the comparators. Adjusted for several possible confounders, the rate ratio was 1.2, (95% confidence interval 0.7–1.9). Types of cancers varied and were remote from the site of treatment.
- The incidence rate for mortality from all cancers was 1.6 per 1,000 person years for those who received REGRANEX Gel and 0.9 per 1,000 person years for the comparators. The adjusted rate ratio was 1.8 (95% confidence interval 0.7–4.9).
- The incidence rate for mortality from all cancers among patients who received 3 or more tubes of REGRANEX Gel was 3.9 per 1,000 person years and 0.9 per 1,000 person years in the comparators. The adjusted rate ratio for cancer mortality among those who received 3 or more tubes relative to those who received none was 5.2, (95% confidence interval 1.6–17.6). (See Boxed Warning)
REGRANEX Gel is a non-sterile, low bioburden preserved product. Therefore, it should not be used in wounds that close by primary intention.
PRECAUTIONS
For external use only.
If application site reactions occur, the possibility of sensitization or irritation caused by parabens or m-cresol should be considered.
The effects of becaplermin on exposed joints, tendons, ligaments, and bone have not been established in humans. In preclinical studies, rats injected at the metatarsals with 3 or 10 µg/site (approximately 60 or 200 µg/kg) of becaplermin every other day for 13 days displayed histological changes indicative of accelerated bone remodeling consisting of periosteal hyperplasia and subperiosteal bone resorption and exostosis. The soft tissue adjacent to the injection site had fibroplasia with accompanying mononuclear cell infiltration reflective of the ability of PDGF to stimulate connective tissue growth.
Information for Patients
Patients should be advised that:
- –
- hands should be washed thoroughly before applying REGRANEX Gel;
- –
- the tip of the tube should not come into contact with the ulcer or any other surface; the tube should be recapped tightly after each use;
- –
- a cotton swab, tongue depressor, or other application aid should be used to apply REGRANEX Gel;
- –
- REGRANEX Gel should only be applied once a day in a carefully measured quantity (see Dosage and Administration section). The measured quantity of gel should be spread evenly over the ulcerated area to yield a thin continuous layer of approximately 1/16 of an inch thickness. The measured length of the gel to be squeezed from the tube should be adjusted according to the size of the ulcer. The amount of REGRANEX Gel to be applied daily should be recalculated at weekly or biweekly intervals by the physician or wound care giver;
Step-by-step instructions for application of REGRANEX Gel are as follows:
- Squeeze the calculated length of gel on to a clean, firm, non-absorbable surface, e.g., wax paper.
- With a clean cotton swab, tongue depressor, or similar application aid, spread the measured REGRANEX Gel over the ulcer surface to obtain an even layer.
- Cover with a saline moistened gauze dressing.
- –
- after approximately 12 hours, the ulcer should be gently rinsed with saline or water to remove residual gel and covered with a saline-moistened gauze dressing (without REGRANEX Gel);
- –
- it is important to use REGRANEX Gel together with a good ulcer care program, including a strict non-weight-bearing program;
- –
- excess application of REGRANEX Gel has not been shown to be beneficial;
- –
- REGRANEX Gel should be stored in the refrigerator. Do not freeze REGRANEX Gel;
- –
- REGRANEX Gel should not be used after the expiration date on the bottom, crimped end of the tube.
Drug Interactions
It is not known if REGRANEX Gel interacts with other topical medications applied to the ulcer site. The use of REGRANEX Gel with other topical drugs has not been studied.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Becaplermin was not genotoxic in a battery of in vitro assays, (including those for bacterial and mammalian cell point mutation, chromosomal aberration, and DNA damage/repair). Becaplermin was also not mutagenic in an in vivo assay for the induction of micronuclei in mouse bone marrow cells.
Carcinogenesis and reproductive toxicity studies have not been conducted with REGRANEX Gel.
Pregnancy
Category C
Animal reproduction studies have not been conducted with REGRANEX Gel. It is also not known whether REGRANEX Gel can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. REGRANEX Gel should be given to pregnant women only if clearly needed.
Nursing Mothers
It is not known whether becaplermin is excreted in human milk. Because many drugs are secreted in human milk, caution should be exercised when REGRANEX Gel is administered to nursing women.
Geriatric Use
Among patients receiving any dose of REGRANEX Gel in clinical studies of diabetic lower extremity ulcers, 150 patients were 65 years of age and older. No overall differences in safety or effectiveness were observed between patients < 65 years of age and patients ≥ 65 years of age. The number of patients aged 75 and older were insufficient (n=34) to determine whether they respond differently from younger patients.
Pediatric Use
Safety and effectiveness of REGRANEX Gel in pediatric patients below the age of 16 years have not been established.
ADVERSE REACTIONS
Clinical Trial Data
The safety of REGRANEX Gel was evaluated in 1883 adult patients who participated in 17 clinical trials of REGRANEX and placebo and/or standard therapy (saline dressing). These 1883 patients had at least one topical administration of REGRANEX and provided safety data.
Adverse drug reactions reported in ≥1% of patients treated with REGRANEX in these trials are shown in Table 1. Incidences are similar between REGRANEX, placebo, and standard therapy (saline dressing), suggesting that these events may also be related to the underlying disease.
| System/Organ Class Adverse Reaction | REGRANEX | Placebo | Standard therapy |
|---|---|---|---|
| (n=1883) % | (n=1069) % | (n=190) % |
|
| Infections and Infestations | |||
| Infected skin ulcer | 12.3 | 11.9 | 18.9 |
| Cellulitis | 10.3 | 7.5 | 18.9 |
| Osteomyelitis | 7.2 | 5.4 | 14.2 |
Postmarketing Data
Adverse drug reactions first identified during postmarketing experience with REGRANEX are included in Table 2. The frequencies are provided according to the following convention:
| Very common | ≥1/10 |
| Common | ≥1/100 and <1/10 |
| Uncommon | ≥ 1/ 1,000 and <1/100 |
| Rare | ≥1/10,000 and <1/1,000 |
| Very rare | <1/10,000, including isolated reports. |
In Table 2, ADRs are presented by frequency category based on spontaneous reporting rates.
| Nervous System Disorders | |
| Rare | Burning sensation* |
| Skin and Subcutaneous Tissue Disorders | |
| Very rare | Rash |
| Erythema† | |
DOSAGE AND ADMINISTRATION
The amount of REGRANEX Gel to be applied will vary depending upon the size of the ulcer area. To calculate the length of gel to apply to the ulcer, measure the greatest length of the ulcer by the greatest width of the ulcer in either inches or centimeters. To calculate the length of gel in inches, use the formula shown below in Table 3, and to calculate the length of gel in centimeters, use the formula shown below in Table 4.
| INCHES | |
|---|---|
| Tube Size | Formula |
| 15 g tube | length × width × 0.6 |
| 2g tube | length × width × 1.3 |
Using the calculation, each square inch of ulcer surface will require approximately 2/3 inch length of gel squeezed from a 15g tube, or approximately 1 1/3inch length of the gel from a 2g tube. For example, if the ulcer measures 1 inch by 2 inches, then a 1 1/4 inch length of gel should be used for 15g tubes (1 × 2 × 0.6 = 1 1/4) and 2 3/4 inch gel length should be used for a 2g tube (1 × 2 × 1.3 = 2 3/4).
| CENTIMETERS | |
|---|---|
| Tube Size | Formula |
| 15 g tube | length × width ÷ 4 |
| 2g tube | length × width ÷ 2 |
Using the calculations for ulcer size in centimeters, each square centimeter of ulcer surface will require approximately a 0.25 centimeter length of gel squeezed from a 15g tube, or approximately a 0.5 centimeter length of gel from a 2g tube. For example, if the ulcer measures 4 cm by 2 cm, then a 2 centimeter length of gel should be used for a 15g tube [(4 × 2) ÷ 4 = 2] and a 4 centimeter length of gel should be used for a 2g tube [(4 X 2) ÷ 2 = 4].
The amount of REGRANEX Gel to be applied should be recalculated by the physician or wound caregiver at weekly or biweekly intervals depending on the rate of change in ulcer area. The weight of REGRANEX Gel from 15g tubes is 0.65g per inch length and 0.25g per centimeter length.
To apply REGRANEX Gel, the calculated length of gel should be squeezed on to a clean measuring surface, e.g., wax paper. The measured REGRANEX Gel is transferred from the clean measuring surface using an application aid and then spread over the entire ulcer area to yield a thin continuous layer of approximately 1/16 of an inch thickness. The site(s) of application should then be covered by a saline moistened dressing and left in place for approximately 12 hours. The dressing should then be removed and the ulcer rinsed with saline or water to remove residual gel and covered again with a second moist dressing (without REGRANEX Gel) for the remainder of the day. REGRANEX Gel should be applied once daily to the ulcer until complete healing has occurred. If the ulcer does not decrease in size by approximately 30% after 10 weeks of treatment or complete healing has not occurred in 20 weeks, continued treatment with REGRANEX Gel should be reassessed. The step-by-step instructions for applying REGRANEX Gel for home administration are described under "Information for Patients".
HOW SUPPLIED
REGRANEX (becaplermin) Gel, supplied as a clear, colorless to straw-colored preserved gel containing 100µg of becaplermin per gram of gel, is available in multi-use tubes in the following sizes:
| 2g tubes | NDC 0045-0810-02 |
| 15g tubes | NDC 0045-0810-15 |
REGRANEX Gel is for external use only.
Storage
Store refrigerated, 2–8°C (36–46°F). DO NOT FREEZE. DO NOT USE THE GEL AFTER THE EXPIRATION DATE AT THE BOTTOM OF THE TUBE.
U.S. Patent #5,457,093
Manufactured by:
OMJ Pharmaceuticals, Inc.
U.S. License No. 1196
San German, Puerto Rico 00683
Manufactured for:
Ortho-McNeil, Division of Ortho-McNeil-Janssen Pharmaceuticals, Inc.
Raritan, NJ 08869
Becaplermin Concentrate provided by: Novartis Vaccines and Diagnostics, Inc.,
U.S. License No. 1751, Emeryville, CA 94608
©OMJPI 2007
Revised: September 2009
Update 2 codes
PRINCIPAL DISPLAY PANEL - 15 g Tube
NDC 0045-0810-15
REGRANEX*
GEL
0.01%
(becaplermin)
Contains: becaplermin 0.01%, sodium
carboxymethylcellulose, sodium chloride,
sodium acetate trihydrate, glacial acetic acid,
L-lysine hydrochloride, and water for injection,
with preservatives: methylparaben 0.156%,
propylparaben 0.017%, and m-cresol 0.086%.
Rx only.
Dosage and Administration: See package insert.
STORE REFRIGERATED, 2° - 8°C (36° - 46°F).
DO NOT FREEZE.
Warning: Keep out of reach of children.
Important: Do not use if seal has been
punctured or is not visible.
To open: Use cap to puncture seal.
To close: Recap tightly after each use.
DIN 02239405 NET WT. 15g
For Topical Use Only
Multi-dose tube
See crimp end for lot number and
expiration date.
ORTHO-McNEIL
Distributed by:
OMP DIVISION
ORTHO-McNEIL
PHARMACEUTICAL, INC.
Raritan, New Jersey 08869
and JANSSEN-ORTHO INC.
Toronto, Canada M3C 1L9
Manufactured by:
OMJ Pharmaceuticals, Inc. 107-10-247-7
U.S. Lic. #1196
San German,
Puerto Rico 00683
© OMP 1998
Made in U.S.A.
*Trademark

PRINCIPAL DISPLAY PANEL - 15 g Tube Carton
NDC 0045-0810-15
REGRANEX®
GEL
0.01%
(becaplermin)
ORTHO-McNEIL
For Topical Use Only
NET WT. 15g
Multi-dose tube
Distributed by:
OMP DIVISION
ORTHO-McNEIL PHARMACEUTICAL, INC.
Raritan, New Jersey 08869
Manufactured by:
OMJ Pharmaceuticals, Inc.
U.S. Lic. #1196
San German, Puerto Rico 00683
| REGRANEX
becaplermin gel |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA103691 | 02/01/1998 | |
| Labeler - Ortho-McNeil-Janssen Pharmaceuticals, Inc. (010779978) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| OMJ Pharmaceuticals, Inc. | 835921206 | ANALYSIS, MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Novartis Vaccines and Diagnostics, Inc. | 046866463 | API MANUFACTURE | |