FLOLAN
-
epoprostenol sodium injection, powder, lyophilized, for solution
DILUENT
-
water solution
SmithKline Beecham Corporation
----------
FLOLAN®(epoprostenol sodium)
for Injection
DESCRIPTION
FLOLAN (epoprostenol sodium) for Injection is a sterile sodium salt formulated for intravenous (IV) administration. Each vial of FLOLAN contains epoprostenol sodium equivalent to either 0.5 mg (500,000 ng) or 1.5 mg (1,500,000 ng) epoprostenol, 3.76 mg glycine, 2.93 mg sodium chloride, and 50 mg mannitol. Sodium hydroxide may have been added to adjust pH.
Epoprostenol (PGI2, PGX, prostacyclin), a metabolite of arachidonic acid, is a naturally occurring prostaglandin with potent vasodilatory activity and inhibitory activity of platelet aggregation.
Epoprostenol is (5Z,9α,11α,13E,15S)-6,9-epoxy-11,15-dihydroxyprosta-5,13-dien-1-oic acid.
Epoprostenol sodium has a molecular weight of 374.45 and a molecular formula of C20H31NaO5. The structural formula is:
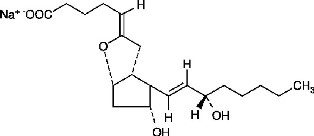
FLOLAN is a white to off-white powder that must be reconstituted with STERILE DILUENT for FLOLAN. STERILE DILUENT for FLOLAN is supplied in glass vials containing 50 mL of 94 mg glycine, 73.3 mg sodium chloride, sodium hydroxide (added to adjust pH), and Water for Injection, USP.
The reconstituted solution of FLOLAN has a pH of 10.2 to 10.8 and is increasingly unstable at a lower pH.
CLINICAL PHARMACOLOGY
General
Epoprostenol has 2 major pharmacological actions: (1) direct vasodilation of pulmonary and systemic arterial vascular beds, and (2) inhibition of platelet aggregation. In animals, the vasodilatory effects reduce right- and left-ventricular afterload and increase cardiac output and stroke volume. The effect of epoprostenol on heart rate in animals varies with dose. At low doses, there is vagally mediated bradycardia, but at higher doses, epoprostenol causes reflex tachycardia in response to direct vasodilation and hypotension. No major effects on cardiac conduction have been observed. Additional pharmacologic effects of epoprostenol in animals include bronchodilation, inhibition of gastric acid secretion, and decreased gastric emptying.
Pharmacokinetics
Epoprostenol is rapidly hydrolyzed at neutral pH in blood and is also subject to enzymatic degradation. Animal studies using tritium-labeled epoprostenol have indicated a high clearance (93 mL/kg/min), small volume of distribution (357 mL/kg), and a short half-life (2.7 minutes). During infusions in animals, steady-state plasma concentrations of tritium-labeled epoprostenol were reached within 15 minutes and were proportional to infusion rates.
No available chemical assay is sufficiently sensitive and specific to assess the in vivo human pharmacokinetics of epoprostenol. The in vitro half-life of epoprostenol in human blood at 37°C and pH 7.4 is approximately 6 minutes; therefore, the in vivo half-life of epoprostenol in humans is expected to be no greater than 6 minutes. The in vitro pharmacologic half-life of epoprostenol in human plasma, based on inhibition of platelet aggregation, was similar for males (n = 954) and females (n = 1,024).
Tritium-labeled epoprostenol has been administered to humans in order to identify the metabolic products of epoprostenol. Epoprostenol is metabolized to 2 primary metabolites: 6-keto-PGF1α (formed by spontaneous degradation) and 6,15-diketo-13,14-dihydro-PGF1α (enzymatically formed), both of which have pharmacological activity orders of magnitude less than epoprostenol in animal test systems. The recovery of radioactivity in urine and feces over a 1-week period was 82% and 4% of the administered dose, respectively. Fourteen additional minor metabolites have been isolated from urine, indicating that epoprostenol is extensively metabolized in humans.
CLINICAL TRIALS IN PULMONARY HYPERTENSION
Acute Hemodynamic Effects
Acute intravenous infusions of FLOLAN for up to 15 minutes in patients with secondary and primary pulmonary hypertension produce dose-related increases in cardiac index (CI) and stroke volume (SV) and dose-related decreases in pulmonary vascular resistance (PVR), total pulmonary resistance (TPR), and mean systemic arterial pressure (SAPm). The effects of FLOLAN on mean pulmonary artery pressure (PAPm) were variable and minor.
Chronic Infusion in Primary Pulmonary Hypertension (PPH): Hemodynamic Effects
Chronic continuous infusions of FLOLAN in patients with PPH were studied in 2 prospective, open, randomized trials of 8 and 12 weeks’ duration comparing FLOLAN plus conventional therapy to conventional therapy alone. Dosage of FLOLAN was determined as described in DOSAGE AND ADMINISTRATION and averaged 9.2 ng/kg/min at study’s end. Conventional therapy varied among patients and included some or all of the following: anticoagulants in essentially all patients; oral vasodilators, diuretics, and digoxin in one half to two thirds of patients; and supplemental oxygen in about half the patients. Except for 2 New York Heart Association (NYHA) functional Class II patients, all patients were either functional Class III or Class IV. As results were similar in the 2 studies, the pooled results are described. Chronic hemodynamic effects were generally similar to acute effects. Increases in CI, SV, and arterial oxygen saturation and decreases in PAPm, mean right atrial pressure (RAPm), TPR, and systemic vascular resistance (SVR) were observed in patients who received FLOLAN chronically compared to those who did not. Table 1 illustrates the treatment-related hemodynamic changes in these patients after 8 or 12 weeks of treatment.
|
Baseline |
Mean Change from Baseline at End of Treatment Period* |
|||
|
Hemodynamic Parameter |
FLOLAN (N = 52) |
Standard Therapy (N = 54) |
FLOLAN (N = 48) |
Standard Therapy (N = 41) |
|
CI (L/min/m2) |
2.0 |
2.0 |
0.3† |
-0.1 |
|
PAPm (mm Hg) |
60 |
60 |
-5† |
1 |
|
PVR (Wood U) |
16 |
17 |
-4† |
1 |
|
SAPm (mm Hg) |
89 |
91 |
-4 |
-3 |
|
SV (mL/beat) |
44 |
43 |
6† |
-1 |
|
TPR (Wood U) |
20 |
21 |
-5† |
1 |
* At 8 weeks: FLOLAN N = 10, conventional therapy N = 11 (N is the number of patients with hemodynamic data).
At 12 weeks: FLOLAN N = 38, conventional therapy N = 30 (N is the number of patients with hemodynamic data).
† Denotes statistically significant difference between FLOLAN and conventional therapy groups.
CI = cardiac index, PAPm = mean pulmonary arterial pressure, PVR = pulmonary vascular resistance, SAPm = mean systemic arterial pressure, SV = stroke volume, TPR = total pulmonary resistance.
These hemodynamic improvements appeared to persist when FLOLAN was administered for at least 36 months in an open, nonrandomized study.
Clinical Effects
Statistically significant improvement was observed in exercise capacity, as measured by the 6-minute walk test in patients receiving continuous intravenous FLOLAN plus conventional therapy (N = 52) for 8 or 12 weeks compared to those receiving conventional therapy alone (N = 54). Improvements were apparent as early as the first week of therapy. Increases in exercise capacity were accompanied by statistically significant improvement in dyspnea and fatigue, as measured by the Chronic Heart Failure Questionnaire and the Dyspnea Fatigue Index.
Survival was improved in NYHA functional Class III and Class IV PPH patients treated with FLOLAN for 12 weeks in a multicenter, open, randomized, parallel study. At the end of the treatment period, 8 of 40 (20%) patients receiving conventional therapy alone died, whereas none of the 41 patients receiving FLOLAN died (p = 0.003).
Chronic Infusion in Pulmonary Hypertension Associated with the Scleroderma Spectrum of Diseases (PH/SSD)
Hemodynamic Effects
Chronic continuous infusions of FLOLAN in patients with PH/SSD were studied in a prospective, open, randomized trial of 12 weeks’ duration comparing FLOLAN plus conventional therapy (N = 56) to conventional therapy alone (N = 55). Except for 5 NYHA functional Class II patients, all patients were either functional Class III or Class IV. Dosage of FLOLAN was determined as described in DOSAGE AND ADMINISTRATION and averaged 11.2 ng/kg/min at study’s end. Conventional therapy varied among patients and included some or all of the following: anticoagulants in essentially all patients, supplemental oxygen and diuretics in two thirds of the patients, oral vasodilators in 40% of the patients, and digoxin in a third of the patients. A statistically significant increase in CI, and statistically significant decreases in PAPm, RAPm, PVR, and SAPm after 12 weeks of treatment were observed in patients who received FLOLAN chronically compared to those who did not. Table 2 illustrates the treatment-related hemodynamic changes in these patients after 12 weeks of treatment.
|
Baseline |
Mean Change from Baseline at 12 Weeks |
|||
|
Hemodynamic Parameter |
FLOLAN (N = 56) |
Conventional Therapy (N = 55) |
FLOLAN (N = 50) |
Conventional Therapy (N = 48) |
|
CI (L/min/m2) |
1.9 |
2.2 |
0.5* |
-0.1 |
|
PAPm (mm Hg) |
51 |
49 |
-5* |
1 |
|
RAPm (mm Hg) |
13 |
11 |
-1* |
1 |
|
PVR (Wood U) |
14 |
11 |
-5* |
1 |
|
SAPm (mm Hg) |
93 |
89 |
-8* |
-1 |
* Denotes statistically significant difference between FLOLAN and conventional therapy groups (N is the number of patients with hemodynamic data).
CI = cardiac index, PAPm = mean pulmonary arterial pressure, RAPm = mean right arterial pressure, PVR = pulmonary vascular resistance, SAPm = mean systemic arterial pressure.
Clinical Effects
Statistically significant improvement was observed in exercise capacity, as measured by the 6-minute walk, in patients receiving continuous intravenous FLOLAN plus conventional therapy for 12 weeks compared to those receiving conventional therapy alone. Improvements were apparent in some patients at the end of the first week of therapy. Increases in exercise capacity were accompanied by statistically significant improvements in dyspnea and fatigue, as measured by the Borg Dyspnea Index and Dyspnea Fatigue Index. At week 12, NYHA functional class improved in 21 of 51 (41%) patients treated with FLOLAN compared to none of the 48 patients treated with conventional therapy alone. However, more patients in both treatment groups (28/51 [55%] with FLOLAN and 35/48 [73%] with conventional therapy alone) showed no change in functional class, and 2/51 (4%) with FLOLAN and 13/48 (27%) with conventional therapy alone worsened. Of the patients randomized, NYHA functional class data at 12 weeks were not available for 5 patients treated with FLOLAN and 7 patients treated with conventional therapy alone.
No statistical difference in survival over 12 weeks was observed in PH/SSD patients treated with FLOLAN as compared to those receiving conventional therapy alone. At the end of the treatment period, 4 of 56 (7%) patients receiving FLOLAN died, whereas 5 of 55 (9%) patients receiving conventional therapy alone died.
No controlled clinical trials with FLOLAN have been performed in patients with pulmonary hypertension associated with other diseases.
INDICATIONS AND USAGE
FLOLAN is indicated for the long-term intravenous treatment of primary pulmonary hypertension and pulmonary hypertension associated with the scleroderma spectrum of disease in NYHA Class III and Class IV patients who do not respond adequately to conventional therapy (see CLINICAL TRIALS IN PULMONARY HYPERTENSION).
CONTRAINDICATIONS
A large study evaluating the effect of FLOLAN on survival in NYHA Class III and IV patients with congestive heart failure due to severe left ventricular systolic dysfunction was terminated after an interim analysis of 471 patients revealed a higher mortality in patients receiving FLOLAN plus conventional therapy than in those receiving conventional therapy alone. The chronic use of FLOLAN in patients with congestive heart failure due to severe left ventricular systolic dysfunction is therefore contraindicated.
Some patients with pulmonary hypertension have developed pulmonary edema during dose initiation, which may be associated with pulmonary veno-occlusive disease. FLOLAN should not be used chronically in patients who develop pulmonary edema during dose initiation.
FLOLAN is also contraindicated in patients with known hypersensitivity to the drug or to structurally related compounds.
WARNINGS
FLOLAN must be reconstituted only as directed using STERILE DILUENT for FLOLAN. FLOLAN must not be reconstituted or mixed with any other parenteral medications or solutions prior to or during administration.
Abrupt Withdrawal
Abrupt withdrawal (including interruptions in drug delivery) or sudden large reductions in dosage of FLOLAN may result in symptoms associated with rebound pulmonary hypertension, including dyspnea, dizziness, and asthenia. In clinical trials, one Class III PPH patient's death was judged attributable to the interruption of FLOLAN. Abrupt withdrawal should be avoided.
Sepsis
See ADVERSE REACTIONS: Adverse Events Attributable to the Drug Delivery System.
PRECAUTIONS
General
FLOLAN should be used only by clinicians experienced in the diagnosis and treatment of pulmonary hypertension. The diagnosis of PPH or PH/SSD should be carefully established.
FLOLAN is a potent pulmonary and systemic vasodilator. Dose initiation with FLOLAN must be performed in a setting with adequate personnel and equipment for physiologic monitoring and emergency care. Dose initiation in controlled PPH clinical trials was performed during right heart catheterization. In uncontrolled PPH and controlled PH/SSD clinical trials, dose initiation was performed without cardiac catheterization. The risk of cardiac catheterization in patients with pulmonary hypertension should be carefully weighed against the potential benefits. During dose initiation, asymptomatic increases in pulmonary artery pressure coincident with increases in cardiac output occurred rarely. In such cases, dose reduction should be considered, but such an increase does not imply that chronic treatment is contraindicated.
FLOLAN is a potent inhibitor of platelet aggregation. Therefore, an increased risk for hemorrhagic complications should be considered, particularly for patients with other risk factors for bleeding (see PRECAUTIONS: Drug Interactions).
During chronic use, FLOLAN is delivered continuously on an ambulatory basis through a permanent indwelling central venous catheter. Unless contraindicated, anticoagulant therapy should be administered to PPH and PH/SSD patients receiving FLOLAN to reduce the risk of pulmonary thromboembolism or systemic embolism through a patent foramen ovale. In order to reduce the risk of infection, aseptic technique must be used in the reconstitution and administration of FLOLAN as well as in routine catheter care. Because FLOLAN is metabolized rapidly, even brief interruptions in the delivery of FLOLAN may result in symptoms associated with rebound pulmonary hypertension including dyspnea, dizziness, and asthenia. The decision to initiate therapy with FLOLAN should be based upon the understanding that there is a high likelihood that intravenous therapy with FLOLAN will be needed for prolonged periods, possibly years, and the patient's ability to accept and care for a permanent intravenous catheter and infusion pump should be carefully considered.
Based on clinical trials, the acute hemodynamic response to FLOLAN did not correlate well with improvement in exercise tolerance or survival during chronic use of FLOLAN. Dosage of FLOLAN during chronic use should be adjusted at the first sign of recurrence or worsening of symptoms attributable to pulmonary hypertension or the occurrence of adverse events associated with FLOLAN (see DOSAGE AND ADMINISTRATION). Following dosage adjustments, standing and supine blood pressure and heart rate should be monitored closely for several hours.
Information for Patients
Patients receiving FLOLAN should receive the following information. FLOLAN must be reconstituted only with STERILE DILUENT for FLOLAN. FLOLAN is infused continuously through a permanent indwelling central venous catheter via a small, portable infusion pump. Thus, therapy with FLOLAN requires commitment by the patient to drug reconstitution, drug administration, and care of the permanent central venous catheter. Sterile technique must be adhered to in preparing the drug and in the care of the catheter, and even brief interruptions in the delivery of FLOLAN may result in rapid symptomatic deterioration. A patient’s decision to receive FLOLAN should be based upon the understanding that there is a high likelihood that therapy with FLOLAN will be needed for prolonged periods, possibly years. The patient's ability to accept and care for a permanent intravenous catheter and infusion pump should also be carefully considered.
Drug Interactions
Additional reductions in blood pressure may occur when FLOLAN is administered with diuretics, antihypertensive agents, or other vasodilators. When other antiplatelet agents or anticoagulants are used concomitantly, there is the potential for FLOLAN to increase the risk of bleeding. However, patients receiving infusions of FLOLAN in clinical trials were maintained on anticoagulants without evidence of increased bleeding. In clinical trials, FLOLAN was used with digoxin, diuretics, anticoagulants, oral vasodilators, and supplemental oxygen.
In a pharmacokinetic substudy in patients with congestive heart failure receiving furosemide or digoxin in whom therapy with FLOLAN was initiated, apparent oral clearance values for furosemide (n = 23) and digoxin (n = 30) were decreased by 13% and 15%, respectively, on the second day of therapy and had returned to baseline values by day 87. The change in furosemide clearance value is not likely to be clinically significant. However, patients on digoxin may show elevations of digoxin concentrations after initiation of therapy with FLOLAN, which may be clinically significant in patients prone to digoxin toxicity.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential. A micronucleus test in rats revealed no evidence of mutagenicity. The Ames test and DNA elution tests were also negative, although the instability of epoprostenol makes the significance of these tests uncertain. Fertility was not impaired in rats given FLOLAN by subcutaneous injection at doses up to 100 mcg/kg/day (600 mcg/m2/day, 2.5 times the recommended human dose [4.6 ng/kg/min or 245.1 mcg/m2/day, IV] based on body surface area).
Pregnancy
Pregnancy Category B. Reproductive studies have been performed in pregnant rats and rabbits at doses up to 100 mcg/kg/day (600 mcg/m2/day in rats, 2.5 times the recommended human dose, and 1,180 mcg/m2/day in rabbits, 4.8 times the recommended human dose based on body surface area) and have revealed no evidence of impaired fertility or harm to the fetus due to FLOLAN. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Labor and Delivery
The use of FLOLAN during labor, vaginal delivery, or cesarean section has not been adequately studied in humans.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when FLOLAN is administered to a nursing woman.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatric Use
Clinical studies of FLOLAN in pulmonary hypertension did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
ADVERSE REACTIONS
During clinical trials, adverse events were classified as follows: (1) adverse events during dose initiation and escalation, (2) adverse events during chronic dosing, and (3) adverse events associated with the drug delivery system.
Adverse Events During Dose Initiation and Escalation
During early clinical trials, FLOLAN was increased in 2-ng/kg/min increments until the patients developed symptomatic intolerance. The most common adverse events and the adverse events that limited further increases in dose were generally related to vasodilation, the major pharmacologic effect of FLOLAN. The most common dose-limiting adverse events (occurring in ≥1% of patients) were nausea, vomiting, headache, hypotension, and flushing, but also include chest pain, anxiety, dizziness, bradycardia, dyspnea, abdominal pain, musculoskeletal pain, and tachycardia. Table 3 lists the adverse events reported during dose initiation and escalation in decreasing order of frequency.
|
Adverse Events Occurring in≥1% of Patients |
FLOLAN (n = 391) |
|
Flushing |
58% |
|
Headache |
49% |
|
Nausea/vomiting |
32% |
|
Hypotension |
16% |
|
Anxiety, nervousness, agitation |
11% |
|
Chest pain |
11% |
|
Dizziness |
8% |
|
Bradycardia |
5% |
|
Abdominal pain |
5% |
|
Musculoskeletal pain |
3% |
|
Dyspnea |
2% |
|
Back pain |
2% |
|
Sweating |
1% |
|
Dyspepsia |
1% |
|
Hypesthesia/paresthesia |
1% |
|
Tachycardia |
1% |
Adverse Events During Chronic Administration
Interpretation of adverse events is complicated by the clinical features of PPH and PH/SSD, which are similar to some of the pharmacologic effects of FLOLAN (e.g., dizziness, syncope). Adverse events probably related to the underlying disease include dyspnea, fatigue, chest pain, edema, hypoxia, right ventricular failure, and pallor. Several adverse events, on the other hand, can clearly be attributed to FLOLAN. These include headache, jaw pain, flushing, diarrhea, nausea and vomiting, flu-like symptoms, and anxiety/nervousness.
Adverse Events During Chronic Administration for PPH
In an effort to separate the adverse effects of the drug from the adverse effects of the underlying disease, Table 4 lists adverse events that occurred at a rate at least 10% different in the 2 groups in controlled trials for PPH.
|
Adverse Event |
FLOLAN (n = 52) |
Conventional Therapy (n = 54) |
|
Occurrence More Common With FLOLAN |
||
|
General | ||
|
Chills/fever/sepsis/flu-like symptoms |
25% |
11% |
|
Cardiovascular | ||
|
Tachycardia |
35% |
24% |
|
Flushing |
42% |
2% |
|
Gastrointestinal | ||
|
Diarrhea |
37% |
6% |
|
Nausea/vomiting |
67% |
48% |
|
Musculoskeletal | ||
|
Jaw pain |
54% |
0% |
|
Myalgia |
44% |
31% |
|
Nonspecific musculoskeletal pain |
35% |
15% |
|
Neurological | ||
|
Anxiety/nervousness/tremor |
21% |
9% |
|
Dizziness |
83% |
70% |
|
Headache |
83% |
33% |
|
Hypesthesia, hyperesthesia, paresthesia |
12% |
2% |
|
Occurrence More Common With Standard Therapy |
||
|
Cardiovascular | ||
|
Heart failure |
31% |
52% |
|
Syncope |
13% |
24% |
|
Shock |
0% |
13% |
|
Respiratory | ||
|
Hypoxia |
25% |
37% |
Thrombocytopenia has been reported during uncontrolled clinical trials in patients receiving FLOLAN.
Table 5 lists additional adverse events reported in PPH patients receiving FLOLAN plus conventional therapy or conventional therapy alone during controlled clinical trials.
|
Adverse Event |
FLOLAN (n = 52) |
Conventional Therapy (n = 54) |
|
General | ||
|
Asthenia |
87% |
81% |
|
Cardiovascular | ||
|
Angina pectoris |
19% |
20% |
|
Arrhythmia |
27% |
20% |
|
Bradycardia |
15% |
9% |
|
Supraventricular tachycardia |
8% |
0% |
|
Pallor |
21% |
30% |
|
Cyanosis |
31% |
39% |
|
Palpitation |
63% |
61% |
|
Cerebrovascular accident |
4% |
0% |
|
Hemorrhage |
19% |
11% |
|
Hypotension |
27% |
31% |
|
Myocardial ischemia |
2% |
6% |
|
Gastrointestinal | ||
|
Abdominal pain |
27% |
31% |
|
Anorexia |
25% |
30% |
|
Ascites |
12% |
17% |
|
Constipation |
6% |
2% |
|
Metabolic | ||
|
Edema |
60% |
63% |
|
Hypokalemia |
6% |
4% |
|
Weight reduction |
27% |
24% |
|
Weight gain |
6% |
4% |
|
Musculoskeletal | ||
|
Arthralgia |
6% |
0% |
|
Bone pain |
0% |
4% |
|
Chest pain |
67% |
65% |
|
Neurological | ||
|
Confusion |
6% |
11% |
|
Convulsion |
4% |
0% |
|
Depression |
37% |
44% |
|
Insomnia |
4% |
4% |
|
Respiratory | ||
|
Cough increase |
38% |
46% |
|
Dyspnea |
90% |
85% |
|
Epistaxis |
4% |
2% |
|
Pleural effusion |
4% |
2% |
|
Skin and Appendages | ||
|
Pruritus |
4% |
0% |
|
Rash |
10% |
13% |
|
Sweating |
15% |
20% |
|
Special Senses | ||
|
Amblyopia |
8% |
4% |
|
Vision abnormality |
4% |
0% |
Adverse Events During Chronic Administration for PH/SSD
In an effort to separate the adverse effects of the drug from the adverse effects of the underlying disease, Table 6 lists adverse events that occurred at a rate at least 10% different in the 2 groups in the controlled trial for patients with PH/SSD.
|
Adverse Event |
FLOLAN (n = 56) |
Conventional Therapy (n = 55) |
|
Occurrence More Common With FLOLAN |
||
|
Cardiovascular | ||
|
Flushing |
23% |
0% |
|
Hypotension |
13% |
0% |
|
Gastrointestinal | ||
|
Anorexia |
66% |
47% |
|
Nausea/vomiting |
41% |
16% |
|
Diarrhea |
50% |
5% |
|
Musculoskeletal | ||
|
Jaw pain |
75% |
0% |
|
Pain/neck pain/arthralgia |
84% |
65% |
|
Neurological | ||
|
Headache |
46% |
5% |
|
Skin and Appendages | ||
|
Skin ulcer |
39% |
24% |
|
Eczema/rash/urticaria |
25% |
4% |
|
Occurrence More Common With Conventional Therapy |
||
|
Cardiovascular | ||
|
Cyanosis |
54% |
80% |
|
Pallor |
32% |
53% |
|
Syncope |
7% |
20% |
|
Gastrointestinal | ||
|
Ascites |
23% |
33% |
|
Esophageal reflux/gastritis |
61% |
73% |
|
Metabolic | ||
|
Weight decrease |
45% |
56% |
|
Neurological | ||
|
Dizziness |
59% |
76% |
|
Respiratory | ||
|
Hypoxia |
55% |
65% |
Table 7 lists additional adverse events reported in PH/SSD patients receiving FLOLAN plus conventional therapy or conventional therapy alone during controlled clinical trials.
|
Adverse Event* |
FLOLAN (n = 56) |
Conventional Therapy (n = 55) |
|
General | ||
|
Asthenia |
100% |
98% |
|
Hemorrhage/hemorrhage injection site/hemorrhage rectal |
11% |
2% |
|
Infection/rhinitis |
21% |
20% |
|
Chills/fever/sepsis/flu-like symptoms |
13% |
11% |
|
Blood and Lymphatic | ||
|
Thrombocytopenia |
4% |
0% |
|
Cardiovascular | ||
|
Heart failure/heart failure right |
11% |
13% |
|
Myocardial Infarction |
4% |
0% |
|
Palpitation |
63% |
71% |
|
Shock |
5% |
5% |
|
Tachycardia |
43% |
42% |
|
Vascular disorder peripheral |
96% |
100% |
|
Vascular disorder |
95% |
89% |
|
Gastrointestinal | ||
|
Abdominal enlargement |
4% |
0% |
|
Abdominal pain |
14% |
7% |
|
Constipation |
4% |
2% |
|
Flatulence |
5% |
4% |
|
Metabolic | ||
|
Edema/edema peripheral/edema genital |
79% |
87% |
|
Hypercalcemia |
48% |
51% |
|
Hyperkalemia |
4% |
0% |
|
Thirst |
0% |
4% |
|
Musculoskeletal | ||
|
Arthritis |
52% |
45% |
|
Back pain |
13% |
5% |
|
Chest pain |
52% |
45% |
|
Cramps leg |
5% |
7% |
|
Respiratory | ||
|
Cough increase |
82% |
82% |
|
Dyspnea |
100% |
100% |
|
Epistaxis |
9% |
7% |
|
Pharyngitis |
5% |
2% |
|
Pleural effusion |
7% |
0% |
|
Pneumonia |
5% |
0% |
|
Pneumothorax |
4% |
0% |
|
Pulmonary edema |
4% |
2% |
|
Respiratory disorder |
7% |
4% |
|
Sinusitis |
4% |
4% |
|
Neurological | ||
|
Anxiety/hyperkinesia/nervousness/tremor |
7% |
5% |
|
Depression/depression psychotic |
13% |
4% |
|
Hyperesthesia/hypesthesia/paresthesia |
5% |
0% |
|
Insomnia |
9% |
0% |
|
Somnolence |
4% |
2% |
|
Skin and Appendages | ||
|
Collagen disease |
82% |
84% |
|
Pruritus |
4% |
2% |
|
Sweat |
41% |
36% |
|
Urogenital | ||
|
Hematuria |
5% |
0% |
|
Urinary tract infection |
7% |
0% |
*Adverse events that occurred in at least 2 patients in either treatment group.
Although the relationship to FLOLAN administration has not been established, pulmonary embolism has been reported in several patients taking FLOLAN and there have been reports of hepatic failure.
Adverse Events Attributable to the Drug Delivery System
Chronic infusions of FLOLAN are delivered using a small, portable infusion pump through an indwelling central venous catheter. During controlled PPH trials of up to 12 weeks’ duration, up to 21% of patients reported a local infection and up to 13% of patients reported pain at the injection site. During a controlled PH/SSD trial of 12 weeks’ duration, 14% of patients reported a local infection and 9% of patients reported pain at the injection site. During long-term follow-up in the clinical trial of PPH, sepsis was reported at least once in 14% of patients and occurred at a rate of 0.32 infections/patient per year in patients treated with FLOLAN. This rate was higher than reported in patients using chronic indwelling central venous catheters to administer parenteral nutrition, but lower than reported in oncology patients using these catheters. Malfunctions in the delivery system resulting in an inadvertent bolus of or a reduction in FLOLAN were associated with symptoms related to excess or insufficient FLOLAN, respectively (see ADVERSE REACTIONS: Adverse Events During Chronic Administration).
Observed During Clinical Practice
In addition to adverse reactions reported from clinical trials, the following events have been identified during post-approval use of FLOLAN. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to FLOLAN.
Blood and Lymphatic
Anemia, hypersplenism, pancytopenia, splenomegaly.
Endocrine and Metabolic
Hyperthyroidism.
OVERDOSAGE
Signs and symptoms of excessive doses of FLOLAN during clinical trials are the expected dose-limiting pharmacologic effects of FLOLAN, including flushing, headache, hypotension, tachycardia, nausea, vomiting, and diarrhea. Treatment will ordinarily require dose reduction of FLOLAN.
One patient with secondary pulmonary hypertension accidentally received 50 mL of an unspecified concentration of FLOLAN. The patient vomited and became unconscious with an initially unrecordable blood pressure. FLOLAN was discontinued and the patient regained consciousness within seconds. In clinical practice, fatal occurrences of hypoxemia, hypotension, and respiratory arrest have been reported following overdosage of FLOLAN.
Single intravenous doses of FLOLAN at 10 and 50 mg/kg (2,703 and 27,027 times the recommended acute phase human dose based on body surface area) were lethal to mice and rats, respectively. Symptoms of acute toxicity were hypoactivity, ataxia, loss of righting reflex, deep slow breathing, and hypothermia.
DOSAGE AND ADMINISTRATION
Important Note
FLOLAN must be reconstituted only with STERILE DILUENT for FLOLAN. Reconstituted solutions of FLOLAN must not be diluted or administered with other parenteral solutions or medications (see WARNINGS).
Dosage
Continuous chronic infusion of FLOLAN should be administered through a central venous catheter. Temporary peripheral intravenous infusion may be used until central access is established. Chronic infusion of FLOLAN should be initiated at 2 ng/kg/min and increased in increments of 2 ng/kg/min every 15 minutes or longer until dose-limiting pharmacologic effects are elicited or until a tolerance limit to the drug is established and further increases in the infusion rate are not clinically warranted (see Dosage Adjustments). If dose-limiting pharmacologic effects occur, then the infusion rate should be decreased to an appropriate chronic infusion rate whereby the pharmacologic effects of FLOLAN are tolerated. In clinical trials, the most common dose-limiting adverse events were nausea, vomiting, hypotension, sepsis, headache, abdominal pain, or respiratory disorder (most treatment-limiting adverse events were not serious). If the initial infusion rate of 2 ng/kg/min is not tolerated, a lower dose that is tolerated by the patient should be identified.
In the controlled 12-week trial in PH/SSD, for example, the dose increased from a mean starting dose of 2.2 ng/kg/min. During the first 7 days of treatment, the dose was increased daily to a mean dose of 4.1 ng/kg/min on day 7 of treatment. At the end of week 12, the mean dose was 11.2 ng/kg/min. The mean incremental increase was 2 to 3 ng/kg/min every 3 weeks.
Dosage Adjustments
Changes in the chronic infusion rate should be based on persistence, recurrence, or worsening of the patient's symptoms of pulmonary hypertension and the occurrence of adverse events due to excessive doses of FLOLAN. In general, increases in dose from the initial chronic dose should be expected.
Increments in dose should be considered if symptoms of pulmonary hypertension persist or recur after improving. The infusion should be increased by 1- to 2-ng/kg/min increments at intervals sufficient to allow assessment of clinical response; these intervals should be at least 15 minutes. In clinical trials, incremental increases in dose occurred at intervals of 24 to 48 hours or longer. Following establishment of a new chronic infusion rate, the patient should be observed, and standing and supine blood pressure and heart rate monitored for several hours to ensure that the new dose is tolerated.
During chronic infusion, the occurrence of dose-limiting pharmacological events may necessitate a decrease in infusion rate, but the adverse event may occasionally resolve without dosage adjustment. Dosage decreases should be made gradually in 2-ng/kg/min decrements every 15 minutes or longer until the dose-limiting effects resolve. Abrupt withdrawal of FLOLAN or sudden large reductions in infusion rates should be avoided. Except in life-threatening situations (e.g., unconsciousness, collapse, etc.), infusion rates of FLOLAN should be adjusted only under the direction of a physician.
In patients receiving lung transplants, doses of FLOLAN were tapered after the initiation of cardiopulmonary bypass.
Administration
FLOLAN is administered by continuous intravenous infusion via a central venous catheter using an ambulatory infusion pump. During initiation of treatment, FLOLAN may be administered peripherally.
The ambulatory infusion pump used to administer FLOLAN should: (1) be small and lightweight, (2) be able to adjust infusion rates in 2-ng/kg/min increments, (3) have occlusion, end-of-infusion, and low-battery alarms, (4) be accurate to ±6% of the programmed rate, and (5) be positive pressure-driven (continuous or pulsatile) with intervals between pulses not exceeding 3 minutes at infusion rates used to deliver FLOLAN. The reservoir should be made of polyvinyl chloride, polypropylene, or glass. The infusion pump used in the most recent clinical trials was the CADD-1 HFX 5100 (SIMS Deltec). A 60-inch microbore non-DEHP extension set with proximal antisyphon valve, low priming volume (0.9 mL), and in-line 0.22 micron filter was used during clinical trials.
To avoid potential interruptions in drug delivery, the patient should have access to a backup infusion pump and intravenous infusion sets. A multi-lumen catheter should be considered if other intravenous therapies are routinely administered.
To facilitate extended use at ambient temperatures exceeding 25°C (77°F), a cold pouch with frozen gel packs was used in clinical trials (see DOSAGE AND ADMINISTRATION: Storage and Stability). The cold pouches and gel packs used in clinical trials were obtained from Palco Labs, Palo Alto, California. Any cold pouch used must be capable of maintaining the temperature of reconstituted FLOLAN between 2° and 8°C for 12 hours.
Reconstitution
FLOLAN is stable only when reconstituted with STERILE DILUENT for FLOLAN. FLOLAN must not be reconstituted or mixed with any other parenteral medications or solutions prior to or during administration.
A concentration for the solution of FLOLAN should be selected that is compatible with the infusion pump being used with respect to minimum and maximum flow rates, reservoir capacity, and the infusion pump criteria listed above. FLOLAN, when administered chronically, should be prepared in a drug delivery reservoir appropriate for the infusion pump with a total reservoir volume of at least 100 mL. FLOLAN should be prepared using 2 vials of STERILE DILUENT for FLOLAN for use during a 24-hour period. Table 8 gives directions for preparing several different concentrations of FLOLAN.
|
To make 100 mL of solution with Final Concentration (ng/mL) of: |
Directions: |
|
3,000 ng/mL |
Dissolve contents of one 0.5-mg vial with 5 mL of STERILE DILUENT for FLOLAN. Withdraw 3 mL and add to sufficient STERILE DILUENT for FLOLAN to make a total of 100 mL. |
|
5,000 ng/mL |
Dissolve contents of one 0.5-mg vial with 5 mL of STERILE DILUENT for FLOLAN. Withdraw entire vial contents and add sufficient STERILE DILUENT for FLOLAN to make a total of 100 mL. |
|
10,000 ng/mL |
Dissolve contents of two 0.5-mg vials each with 5 mL of STERILE DILUENT for FLOLAN. Withdraw entire vial contents and add sufficient STERILE DILUENT for FLOLAN to make a total of 100 mL. |
|
15,000 ng/mL* |
Dissolve contents of one 1.5-mg vial with 5 mL of STERILE DILUENT for FLOLAN. Withdraw entire vial contents and add sufficient STERILE DILUENT for FLOLAN to make a total of 100 mL. |
* Higher concentrations may be required for patients who receive FLOLAN long-term.
Generally, 3,000 ng/mL and 10,000 ng/mL are satisfactory concentrations to deliver between 2 to 16 ng/kg/min in adults. Infusion rates may be calculated using the following formula:
Infusion Rate (mL/hr) = [Dose (ng/kg/min) x Weight (kg) x 60 min/hr]
Final Concentration (ng/mL)
Tables 9 through 12 provide infusion delivery rates for doses up to 16 ng/kg/min based upon patient weight, drug delivery rate, and concentration of the solution of FLOLAN to be used. These tables may be used to select the most appropriate concentration of FLOLAN that will result in an infusion rate between the minimum and maximum flow rates of the infusion pump and that will allow the desired duration of infusion from a given reservoir volume. Higher infusion rates, and therefore, more concentrated solutions may be necessary with long-term administration of FLOLAN.
|
Patient |
Dose or Drug Delivery Rate (ng/kg/min) |
|||||||
|
Weight (kg) |
2 |
4 |
6 |
8 |
10 |
12 |
14 |
16 |
|
Infusion Delivery Rate (mL/h) |
||||||||
|
10 |
--- |
--- |
1.2 |
1.6 |
2.0 |
2.4 |
2.8 |
3.2 |
|
20 |
--- |
1.6 |
2.4 |
3.2 |
4.0 |
4.8 |
5.6 |
6.4 |
|
30 |
1.2 |
2.4 |
3.6 |
4.8 |
6.0 |
7.2 |
8.4 |
9.6 |
|
40 |
1.6 |
3.2 |
4.8 |
6.4 |
8.0 |
9.6 |
11.2 |
12.8 |
|
50 |
2.0 |
4.0 |
6.0 |
8.0 |
10.0 |
12.0 |
14.0 |
16.0 |
|
60 |
2.4 |
4.8 |
7.2 |
9.6 |
12.0 |
14.4 |
16.8 |
19.2 |
|
70 |
2.8 |
5.6 |
8.4 |
11.2 |
14.0 |
16.8 |
19.6 |
22.4 |
|
80 |
3.2 |
6.4 |
9.6 |
12.8 |
16.0 |
19.2 |
22.4 |
25.6 |
|
90 |
3.6 |
7.2 |
10.8 |
14.4 |
18.0 |
21.6 |
25.2 |
28.8 |
|
100 |
4.0 |
8.0 |
12.0 |
16.0 |
20.0 |
24.0 |
28.0 |
32.0 |
|
Patient |
Dose or Drug Delivery Rate (ng/kg/min) |
|||||||
|
Weight (kg) |
2 |
4 |
6 |
8 |
10 |
12 |
14 |
16 |
|
Infusion Delivery Rate (mL/h) |
||||||||
|
10 |
--- |
--- |
--- |
1.0 |
1.2 |
1.4 |
1.7 |
1.9 |
|
20 |
--- |
1.0 |
1.4 |
1.9 |
2.4 |
2.9 |
3.4 |
3.8 |
|
30 |
--- |
1.4 |
2.2 |
2.9 |
3.6 |
4.3 |
5.0 |
5.8 |
|
40 |
1.0 |
1.9 |
2.9 |
3.8 |
4.8 |
5.8 |
6.7 |
7.7 |
|
50 |
1.2 |
2.4 |
3.6 |
4.8 |
6.0 |
7.2 |
8.4 |
9.6 |
|
60 |
1.4 |
2.9 |
4.3 |
5.8 |
7.2 |
8.6 |
10.1 |
11.5 |
|
70 |
1.7 |
3.4 |
5.0 |
6.7 |
8.4 |
10.1 |
11.8 |
13.4 |
|
80 |
1.9 |
3.8 |
5.8 |
7.7 |
9.6 |
11.5 |
13.4 |
15.4 |
|
90 |
2.2 |
4.3 |
6.5 |
8.6 |
10.8 |
13.0 |
15.1 |
17.3 |
|
100 |
2.4 |
4.8 |
7.2 |
9.6 |
12.0 |
14.4 |
16.8 |
19.2 |
|
Patient |
Dose or Drug Delivery Rate (ng/kg/min) |
||||||
|
Weight (kg) |
4 |
6 |
8 |
10 |
12 |
14 |
16 |
|
Infusion Delivery Rate (mL/h) |
|||||||
|
20 |
--- |
--- |
1.0 |
1.2 |
1.4 |
1.7 |
1.9 |
|
30 |
--- |
1.1 |
1.4 |
1.8 |
2.2 |
2.5 |
2.9 |
|
40 |
1.0 |
1.4 |
1.9 |
2.4 |
2.9 |
3.4 |
3.8 |
|
50 |
1.2 |
1.8 |
2.4 |
3.0 |
3.6 |
4.2 |
4.8 |
|
60 |
1.4 |
2.2 |
2.9 |
3.6 |
4.3 |
5.0 |
5.8 |
|
70 |
1.7 |
2.5 |
3.4 |
4.2 |
5.0 |
5.9 |
6.7 |
|
80 |
1.9 |
2.9 |
3.8 |
4.8 |
5.8 |
6.7 |
7.7 |
|
90 |
2.2 |
3.2 |
4.3 |
5.4 |
6.5 |
7.6 |
8.6 |
|
100 |
2.4 |
3.6 |
4.8 |
6.0 |
7.2 |
8.4 |
9.6 |
|
Patient |
Dose or Drug Delivery Rate (ng/kg/min) |
||||||
|
Weight (kg) |
4 |
6 |
8 |
10 |
12 |
14 |
16 |
|
Infusion Delivery Rate (mL/h) |
|||||||
|
30 |
--- |
--- |
1.0 |
1.2 |
1.4 |
1.7 |
1.9 |
|
40 |
--- |
1.0 |
1.3 |
1.6 |
1.9 |
2.2 |
2.6 |
|
50 |
--- |
1.2 |
1.6 |
2.0 |
2.4 |
2.8 |
3.2 |
|
60 |
1.0 |
1.4 |
1.9 |
2.4 |
2.9 |
3.4 |
3.8 |
|
70 |
1.1 |
1.7 |
2.2 |
2.8 |
3.4 |
3.9 |
4.5 |
|
80 |
1.3 |
1.9 |
2.6 |
3.2 |
3.8 |
4.5 |
5.1 |
|
90 |
1.4 |
2.2 |
2.9 |
3.6 |
4.3 |
5.0 |
5.8 |
|
100 |
1.6 |
2.4 |
3.2 |
4.0 |
4.8 |
5.6 |
6.4 |
Storage and Stability
Unopened vials of FLOLAN are stable until the date indicated on the package when stored at 15° to 25°C (59° to 77°F) and protected from light in the carton. Unopened vials of STERILE DILUENT for FLOLAN are stable until the date indicated on the package when stored at 15° to 25°C (59° to 77°F).
Prior to use, reconstituted solutions of FLOLAN must be protected from light and must be refrigerated at 2° to 8°C (36° to 46°F) if not used immediately. Do not freeze reconstituted solutions of FLOLAN. Discard any reconstituted solution that has been frozen.Discard any reconstituted solution if it has been refrigerated for more than 48 hours.
During use, a single reservoir of reconstituted solution of FLOLAN can be administered at room temperature for a total duration of 8 hours, or it can be used with a cold pouch and administered up to 24 hours with the use of 2 frozen 6-oz gel packs in a cold pouch. When stored or in use, reconstituted FLOLAN must be insulated from temperatures greater than 25°C (77°F) and less than 0°C (32°F), and must not be exposed to direct sunlight.
Use at Room Temperature
Prior to use at room temperature, 15° to 25°C (59° to 77°F), reconstituted solutions of FLOLAN may be stored refrigerated at 2° to 8°C (36° to 46°F) for no longer than 40 hours. When administered at room temperature, reconstituted solutions may be used for no longer than 8 hours. This 48-hour period allows the patient to reconstitute a 2-day supply (200 mL) of FLOLAN. Each 100-mL daily supply may be divided into 3 equal portions. Two of the portions are stored refrigerated at 2° to 8°C (36° to 46°F) until they are used.
Use with a Cold Pouch
Prior to infusion with the use of a cold pouch, solutions may be stored refrigerated at 2° to 8°C (36° to 46°F) for up to 24 hours. When a cold pouch is employed during the infusion, reconstituted solutions of FLOLAN may be used for no longer than 24 hours. The gel packs should be changed every 12 hours. Reconstituted solutions may be kept at 2° to 8°C (36° to 46°F), either in refrigerated storage or in a cold pouch or a combination of the two, for no more than 48 hours.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. If either occurs, FLOLAN should not be administered.
HOW SUPPLIED
FLOLAN for Injection is supplied as a sterile freeze-dried powder in 17-mL flint glass vials with gray butyl rubber closures, individually packaged in a carton.
17-mL vial containing epoprostenol sodium equivalent to 0.5 mg (500,000 ng), carton of 1 (NDC 0173-0517-00).
17-mL vial containing epoprostenol sodium equivalent to 1.5 mg (1,500,000 ng), carton of 1 (NDC 0173-0519-00).
Store the vials of FLOLAN at 15° to 25°C (59° to 77°F). Protect from light.
The STERILE DILUENT for FLOLAN is supplied in flint glass vials containing 50-mL diluent with fluororesin-faced butyl rubber closures.
50-mL of STERILE DILUENT for FLOLAN, tray of 2 vials (NDC 0173-0518-01).
Store the vials of STERILE DILUENT for FLOLAN at 15° to 25°C (59° to 77°F). DO NOT FREEZE.
GlaxoSmithKline
Research Triangle Park, NC 27709
©2008, GlaxoSmithKline. All rights reserved.
January 2008 FLL:1PI
Principal Display Panel
NDC 0173-0517-00
FLOLAN®
(epoprostenol sodium)
for Injection
0.5 mg (500,000 ng)
Rx only
Each vial contains epoprostenol sodium equivalent to 0.5 mg (500,000 ng) epoprostenol, 3.76 mg glycine, 2.93 mg sodium chloride, and 50 mg mannitol. Sodium hydroxide may have been added to adjust pH.
For intravenous infusion only.
Not for mixture with other drugs.
Use only STERILE DILUENT for FLOLAN® for reconstitution.
See package insert for Dosage and Administration.
Store at 15o and 25o C (59o to 77oF).
Protect from light.
GlaxoSmithKline
Research Triangle Park, NC 27709
Made in Italy
10000000025562
Rev. 2/06

NDC 0173-0518-01
STERILE DILUENT for FLOLAN®
50 mL
Rx only
Contains drug diluent for use only with FLOLAN® (epoprostenol sodium) for Injection.
Each vial contains 94 mg glycine, ,73.3 mg sodium chloride, sodium hydroxide (added to adjust pH), and water for Injection, USP.
For reconstitution information see package insert for FLOLAN® (epoprostenol sodium) for Injection.
Store at 15o to 25oC (59o to 77oF).
DO NOT FREEZE.
GlaxoSmithKline
Research Triangle Park, NC 27709
Made in England
10000000023966 Rev. 1/06

| FLOLAN
epoprostenol sodium injection, powder, lyophilized, for solution |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020444 | 12/08/1995 | |
| FLOLAN
epoprostenol sodium injection, powder, lyophilized, for solution |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020444 | 12/08/1995 | |
| DILUENT
water solution |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020444 | 05/06/2003 | |
| Labeler - SmithKline Beecham Corporation (167380711) |