QUALAQUIN
-
quinine sulfate capsule
AR Scientific, Inc.
----------
QUALAQUIN®quinine sulfate
CAPSULES USP, 324 mg
Rx only
DESCRIPTION
Qualaquin (quinine sulfate) is an antimalarial drug chemically described as cinchonan-9-ol, 6'-methoxy-,(8α, 9R)-, sulfate (2:1) (salt), dihydrate with a molecular formula of (C20H24N2O2)2•H2SO4•2H2O and a molecular weight of 782.96. The structural formula of quinine sulfate is:
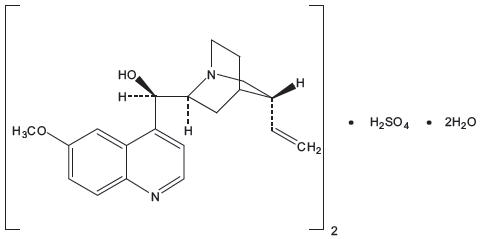
Quinine sulfate occurs as a white, crystalline powder that darkens on exposure to light. It is odorless and has a persistent very bitter taste. It is only slightly soluble in water, alcohol, chloroform, and ether.
Qualaquin is supplied for oral administration as capsules containing 324 mg of the active ingredient quinine sulfate USP, equivalent to 269 mg free base. Inactive ingredients: corn starch, magnesium stearate, and talc.
CLINICAL PHARMACOLOGY
Pharmacokinetics
Absorption
The oral bioavailability of quinine is 76 to 88% in healthy adults. Quinine exposure is higher in patients with malaria than in healthy subjects. After a single oral dose of quinine sulfate, the mean quinine Tmax was longer, and mean AUC and Cmax were higher in patients with uncomplicated P. falciparum malaria than in healthy subjects, as shown in Table 1 below.
| PHARMACOKINETIC PARAMETER | Healthy Subjects (N = 23) Mean ± SD | Uncomplicated P. falciparum Malaria Patients (N = 15) Mean ± SD |
|---|---|---|
|
||
| Dose (mg/kg)* | 8.7 | 10 |
| Tmax (h) | 2.8 ± 0.8 | 5.9 ± 4.7 |
| Cmax (mcg/mL) | 3.2 ± 0.7 | 8.4 |
| AUC0–12 (mcg*h/mL) | 28.0 | 73.0 |
Qualaquin capsules may be administered without regard to meals. When a single oral 324 mg capsule of Qualaquin was administered to healthy volunteers (N=26) with a standardized high-fat breakfast, the mean Tmax of quinine was prolonged to about 4.0 hours, but the mean Cmax and AUC0-24h were similar to those achieved when Qualaquin capsule was given under fasted conditions (See DOSAGE AND ADMINISTRATION).
Distribution
In patients with malaria, the volume of distribution (Vd/f) decreases in proportion to the severity of the infection. In published studies with healthy subjects who received a single oral 600 mg dose of quinine sulfate, the mean Vd/f ranged from 2.5 to 7.1 L/kg.
Quinine is moderately protein-bound in blood in healthy subjects, ranging from 69 to 92%. During active malarial infection, protein binding of quinine is increased to 78 to 95%, corresponding to the increase in α1-acid glycoprotein that occurs with malaria infection.
Intra-erythrocytic levels of quinine are approximately 30 to 50% of the plasma concentration. Quinine penetrates relatively poorly into the cerebrospinal fluid (CSF) in patients with cerebral malaria, with CSF concentration approximately 2 to 7% of plasma concentration.
In one study, quinine concentrations in placental cord blood and breast milk were approximately 32% and 31%, respectively, of quinine concentrations in maternal plasma. The estimated total dose of quinine secreted into breast milk was less than 2 to 3 mg per day (See Pregnancy and Nursing Mothers).
Metabolism
Quinine is metabolized almost exclusively via hepatic oxidative cytochrome P450 (CYP) pathways, resulting in four primary metabolites, 3-hydroxyquinine, 2´-quinone, O-desmethylquinine, and 10,11-dihydroxydihydroquinine. Six secondary metabolites result from further biotransformation of the primary metabolites. The major metabolite, 3-hydroxyquinine, is less active than the parent drug. In vitro studies using human liver microsomes and recombinant P450 enzymes have shown that quinine is metabolized mainly by CYP3A4. Depending on the in vitro experimental conditions, other enzymes, including CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP2E1 were shown to have some role in the metabolism of quinine. (See WARNINGS, PRECAUTIONS/Drug Interactions).
Elimination
Quinine is eliminated primarily via hepatic biotransformation. Approximately 20% of quinine is excreted unchanged in urine. Because quinine is reabsorbed when the urine is alkaline, renal excretion of the drug is twice as rapid when the urine is acidic than when it is alkaline.
In various published studies, healthy subjects who received a single oral 600 mg dose of quinine sulfate exhibited a mean plasma clearance ranging from 0.08 to 0.47 L/h/kg (median value: 0.17 L/h/kg) with a mean plasma elimination half-life of 9.7 to 12.5 hours.
In 15 patients with uncomplicated malaria who received a 10 mg/kg oral dose of quinine sulfate, the mean total clearance of quinine was slower (approximately 0.09 L/h/kg) during the acute phase of the infection, and faster (approximately 0.16 L/h/kg) during the recovery or convalescent phase.
Extracorporeal Elimination
Administration of multiple-dose activated charcoal (50 grams administered 4 hours after quinine dosing followed by 3 further doses over the next 12 hours) decreased the mean quinine elimination half-life from 8.2 to 4.6 hours, and increased the mean quinine clearance by 56% (from 11.8 L/h to 18.4 L/h) in 7 healthy adult volunteers who received a single oral 600 mg dose of quinine sulfate. Likewise, in 5 symptomatic patients with acute quinine poisoning who received multiple-dose activated charcoal (50 grams every 4 hours), the mean quinine elimination half-life was shortened to 8.1 hours in comparison to a half-life of approximately 26 hours in patients who did not receive activated charcoal (See OVERDOSAGE).
In 6 patients with quinine poisoning, forced acid diuresis did not change the half-life of quinine elimination (25.1 ± 4.6 hours vs. 26.5 ± 5.8 hours), or the amount of unchanged quinine recovered in the urine, in comparison to 8 patients not treated in this manner (See OVERDOSAGE).
Special Populations
Pediatrics
The pharmacokinetics of quinine in children (1.5 to 12 years old) with uncomplicated P. falciparum malaria appear to be similar to that seen in adults with uncomplicated malaria. Furthermore, as seen in adults, the mean total clearance and the volume of distribution of quinine were reduced in pediatric patients with malaria as compared to the healthy pediatric controls. Table 2 below provides a comparison of the mean ± SD pharmacokinetic parameters of quinine in pediatric patients vs. healthy pediatric controls.
| PHARMACOKINETIC PARAMETER | P. falciparum malaria pediatric patients (n = 15) Mean ± SD | Healthy pediatric controls (n = 5) Mean ± SD |
|---|---|---|
| Tmax (h) | 4.0 | 2.0 |
| Cmax (mcg/mL) | 7.5 ± 1.1 | 3.4 ± 1.18 |
| Half-life (h) | 12.1 ± 1.4 | 3.2 ± 0.3 |
| Total CL (L/h/kg) | 0.06 ± 0.01 | 0.30 ± 0.04 |
| Vd (L/kg) | 0.87 ± 0.12 | 1.43 ± 0.18 |
Geriatrics
Following a single oral dose of 600 mg quinine sulfate, the mean AUC was about 38% higher in 8 healthy elderly subjects (65 to 78 years old) than in 12 younger subjects (20 to 35 years old). The mean Tmax and Cmax were similar in elderly and younger subjects after a single oral dose of quinine sulfate 600 mg. The mean oral clearance of quinine was significantly decreased, and the mean elimination half-life was significantly increased in elderly subjects compared with younger subjects (0.06 vs. 0.08 L/h/kg, and 18.4 hours vs. 10.5 hours, respectively). Although there was no significant difference in the renal clearance of quinine between the two age groups, elderly subjects excreted a larger proportion of the dose in urine as unchanged drug than younger subjects (16.6% vs. 11.2%). Despite these pharmacokinetic changes, an alteration in the Qualaquin dosage regimen in elderly patients is not needed.
Hepatic impairment
In otherwise healthy subjects with moderate hepatic impairment (Child-Pugh B; N=9) who received a single oral 600 mg dose of quinine sulfate, the mean AUC increased by 55% without a significant change in mean Cmax, as compared to healthy volunteer controls (N=6). In subjects with hepatitis, the absorption of quinine was prolonged, the elimination half-life was increased, the apparent volume of distribution was higher, but there was no significant difference in weight-adjusted clearance. Therefore, in patients with mild to moderate hepatic impairment, dosage adjustment is not needed, but patients should be monitored closely for adverse effects of quinine (See DOSAGE AND ADMINISTRATION). No pharmacokinetic data are available for patients with severe hepatic impairment (Child-Pugh C).
Renal impairment
Following a single oral 600 mg dose of quinine sulfate in otherwise healthy subjects with severe chronic renal failure not receiving any form of dialysis (mean serum creatinine = 9.6 mg/dL), the median AUC was higher by 195% and the median Cmax was higher by 79% than in subjects with normal renal function (mean serum creatinine = 1 mg/dL). The mean plasma half-life in subjects with severe chronic renal impairment was prolonged to 26 hours compared to 9.7 hours in the healthy controls. Computer assisted modeling and simulation indicates that in patients with malaria and severe chronic renal failure, a dosage regimen consisting of one loading dose of 648 mg Qualaquin followed 12 hours later by a maintenance dosing regimen of 324 mg every 12 hours will provide adequate systemic exposure to quinine (See DOSAGE AND ADMINISTRATION). The effects of mild and moderate renal impairment on the pharmacokinetics and safety of quinine sulfate are not known.
Negligible to minimal amounts of circulating quinine in the blood are removed by hemodialysis or hemofiltration. In subjects with chronic renal failure (CRF) on hemodialysis, only about 6.5% of quinine is removed in 1 hour. Plasma quinine concentrations do not change during or shortly after hemofiltration in subjects with CRF (See OVERDOSAGE).
Electrocardiogram
QTc interval prolongation was evaluated in a crossover pharmacokinetic study in healthy volunteers (N=24) who received single oral doses of Qualaquin (324 mg and 648 mg). The mean ± SD maximum QTc change from baseline around the quinine Tmax was 10 ± 19 msec and 12 ± 18 msec, respectively for the 324 mg and 648 mg doses. There were no subjects who had a QTc interval greater than 500 msec, or had a maximum QTc change from baseline of greater than 60 msec (See WARNINGS).
Microbiology
Mechanism of Action
Quinine inhibits nucleic acid synthesis, protein synthesis, and glycolysis in Plasmodium falciparum and can bind with hemazoin in parasitized erythrocytes. However, the precise mechanism of the antimalarial activity of quinine sulfate is not completely understood.
Activity In Vitro and In Vivo
Quinine sulfate acts primarily on the blood schizont form of P. falciparum; it is not gametocidal and has little effect on the sporozoite or pre-erythrocytic forms.
Drug Resistance
Strains of P. falciparum with decreased susceptibility to quinine can be selected in vivo. P. falciparum malaria that is clinically resistant to quinine has been reported in some areas of South America, Southeast Asia, and Bangladesh.
INDICATIONS AND USAGE
Treatment of Malaria
Qualaquin is indicated only for treatment of uncomplicated Plasmodium falciparum malaria. Quinine sulfate has been shown to be effective in geographical regions where resistance to chloroquine has been documented (See CLINICAL STUDIES).
Qualaquin oral capsules are not approved for patients with severe or complicated P. falciparum malaria.
Qualaquin oral capsules are not approved for prevention of malaria.
Qualaquin oral capsules are not approved for the treatment or prevention of nocturnal leg cramps.
CONTRAINDICATIONS
Prolonged QT Interval
Qualaquin is contraindicated in patients with a prolonged QT interval. One case of a fatal ventricular arrhythmia was reported in an elderly patient with a prolonged QT interval at baseline, who received quinine sulfate intravenously for P. falciparum malaria (See WARNINGS).
Glucose-6-Phosphate Dehydrogenase Deficiency
Qualaquin is contraindicated in patients with glucose-6-phosphate dehydrogenase (G-6-PD) deficiency (See WARNINGS).
Myasthenia Gravis
Qualaquin is contraindicated in patients with myasthenia gravis (See WARNINGS).
Hypersensitivity
Qualaquin is contraindicated in patients with known hypersensitivity to quinine. Qualaquin is also contraindicated in patients with known hypersensitivity to mefloquine or quinidine because cross-sensitivity to quinine has been documented (See PRECAUTIONS).
Qualaquin is contraindicated in patients with a history of potential hypersensitivity reactions associated with previous quinine use. These include, but are not limited to the following:
- Thrombotic thrombocytopenic purpura (TTP) or hemolytic uremic syndrome (HUS)
- Thrombocytopenia
- Blackwater fever (acute intravascular hemolysis, hemoglobinuria, and hemoglobinemia) (See PRECAUTIONS).
Optic Neuritis
Qualaquin is contraindicated in patients with optic neuritis (See ADVERSE REACTIONS).
WARNINGS
Use of Qualaquin for Treatment or Prevention of Nocturnal Leg Cramps
Qualaquin may cause unpredictable serious and life-threatening hypersensitivity reactions, thrombocytopenia, QT prolongation, serious cardiac arrhythmias including torsades de pointes, and other serious adverse events requiring medical intervention and hospitalization. Fatalities have also been reported.
The risk associated with the use of Qualaquin in the absence of evidence of its effectiveness for treatment or prevention of nocturnal leg cramps, outweighs any potential benefit in treating and/or preventing this benign, self-limiting condition (See CONTRAINDICATIONS, PRECAUTIONS, and ADVERSE REACTIONS).
Thrombocytopenia
Quinine-induced thrombocytopenia is an immune-mediated disorder characterized by a drug-dependent antibody that is itself nonreactive, but when soluble drug is present at pharmacologic concentrations, binds tightly to specific platelet membrane glycoproteins, causing platelet destruction. Serologic testing for quinine-specific antibody is commercially available and may be useful for identifying the specific cause of thrombocytopenia in individual cases. Testing is important because a patient with quinine-dependent antibodies should not be re-exposed to quinine from any source.
A case control study found a 101-fold increased risk of severe thrombocytopenia (platelets <30,000 µL, requiring hospitalization) with quinidine (d isomer of quinine)/quinine1. The incidence of quinidine/quinine induced thrombocytopenia was 1.4 cases per 1,000 patient years of use. The incidence of less severe thrombocytopenia may be higher.
Typically, a patient with immune thrombocytopenia will have taken drug for about 1 week or intermittently over a longer period of time (possibly years) before presenting with petechiae or bruising. Systemic symptoms, such as lightheadedness, chills, fever, nausea, and vomiting, often may precede bleeding events. Thrombocytopenia may be severe. Patients being treated for malaria should have risk/benefit re-evaluated in order to continue treatment with Qualaquin. Patients using Qualaquin for any other use should have their therapy discontinued. If the drug is stopped, symptoms usually resolve within 1 or 2 days and platelet count returns to normal in less than 1 week. If quinine is not stopped, there is a risk of fatal hemorrhage. The onset of thrombocytopenia may be more rapid upon re-exposure.
QT Prolongation and Ventricular Arrhythmias
QT interval prolongation has been a consistent finding in studies which evaluated electrocardiographic changes with oral or parenteral quinine administration, regardless of age, clinical status, or severity of disease. The maximum increase in QT interval has been shown to correspond with peak quinine plasma concentration (See CLINICAL PHARMACOLOGY/Electrocardiogram). Quinine sulfate has been rarely associated with potentially fatal cardiac arrhythmias, including torsades de pointes, and ventricular fibrillation.
Qualaquin is not recommended for use with other drugs known to cause QT prolongation, including Class IA antiarrhythmic agents (e.g., quinidine, procainamide, disopyramide), and Class III antiarrhythmic agents (e.g., amiodarone, sotalol, dofetilide).
Quinine may also inhibit the metabolism of other drugs that are CYP3A4 substrates known to cause QT prolongation, such as astemizole, cisapride, terfenadine, pimozide, halofantrine and quinidine. Torsades de pointes has been reported in patients who received concomitant quinine and astemizole. Therefore, concurrent use of Qualaquin with these medications, or drugs with similar properties, should be avoided (See PRECAUTIONS/Drug Interactions).
Concomitant administration of Qualaquin with the antimalarial drugs, mefloquine or halofantrine, may result in electrocardiographic abnormalities, including QT prolongation, and increase the risk for torsades de pointes or other serious ventricular arrhythmias. Concurrent use of Qualaquin and mefloquine may also increase the risk of seizures (See PRECAUTIONS/Drug Interactions).
The use of macrolide antibiotics such as erythromycin should be avoided in patients receiving Qualaquin. Fatal torsades de pointes was reported in an elderly patient who received concomitant quinine, erythromycin, and dopamine. Although a causal relationship between a specific drug and the arrhythmia was not established in this case, erythromycin is a CYP3A4 inhibitor and could potentially increase quinine plasma levels when used concomitantly. A related macrolide antibiotic, troleandomycin, has been shown to increase quinine exposure in a pharmacokinetic study (See PRECAUTIONS/Drug Interactions).
Qualaquin should also be avoided in patients with known prolongation of QT interval (See CONTRAINDICATIONS), in elderly patients, and in patients with clinical conditions known to prolong the QT interval, such as uncorrected hypokalemia, bradycardia, and certain cardiac conditions.
Concomitant Use of Rifampin
Treatment failures may result from the concurrent use of rifampin with Qualaquin, due to decreased plasma concentrations of quinine, and concomitant use of these medications should be avoided (See PRECAUTIONS/Drug Interactions).
Glucose-6-Phosphate Dehydrogenase (G-6-PD) Deficiency
Hemolysis and hemolytic anemia can occur in patients with G-6-PD deficiency who receive quinine. Qualaquin should be stopped immediately upon the appearance of evidence of hemolysis (See CONTRAINDICATIONS).
Myasthenia Gravis
Quinine sulfate has neuromuscular blocking activity, and may exacerbate muscle weakness in patients with myasthenia gravis (See CONTRAINDICATIONS).
Neuromuscular Blocking Agents
The use of neuromuscular blocking agents should also be avoided in patients receiving Qualaquin. In one patient who received pancuronium during an operative procedure, subsequent administration of quinine resulted in respiratory depression and apnea. Although there are no clinical reports with succinylcholine or tubocurarine, quinine may also potentiate neuromuscular blockade when used with these drugs (See PRECAUTIONS/Drug Interactions).
PRECAUTIONS
Hypersensitivity
Serious hypersensitivity reactions reported with quinine sulfate include anaphylactic shock, anaphylactoid reactions, urticaria, serious skin rashes, including Stevens-Johnson syndrome and toxic epidermal necrolysis, angioedema, facial edema, bronchospasm, and pruritus (See CONTRAINDICATIONS). A number of other serious adverse reactions reported with quinine, including thrombotic thrombocytopenic purpura (TTP) and hemolytic uremic syndrome (HUS), thrombocytopenia, immune thrombocytopenic purpura (ITP), blackwater fever, disseminated intravascular coagulation, leukopenia, neutropenia, granulomatous hepatitis, and acute interstitial nephritis may also be due to hypersensitivity reactions. Qualaquin should be discontinued in case of any signs or symptoms of hypersensitivity (See CONTRAINDICATIONS).
Atrial Fibrillation and Flutter
Qualaquin should be used with caution in patients with atrial fibrillation or atrial flutter. A paradoxical increase in ventricular response rate may occur with quinine, similar to that observed with quinidine. If digoxin is used to prevent a rapid ventricular response, serum digoxin levels should be closely monitored, because digoxin levels may be increased with use of quinine (See PRECAUTIONS/Drug Interactions).
Hypoglycemia
Quinine stimulates release of insulin from the pancreas, and patients, especially pregnant women, may experience clinically significant hypoglycemia.
Information for Patients
Patients should be instructed to:
- Take all of the medication as directed.
- Take no more of the medication than the amount prescribed.
- Take with food to minimize possible gastrointestinal irritation.
If a dose is missed, patients should also be instructed not to double the next dose. If more than 4 hours has elapsed since the missed dose, the patient should wait and take the next dose as previously scheduled. (See Patient Package Insert.)
Drug Interactions
Effects of Drugs and Other Substances on Quinine Pharmacokinetics
Drugs and other substances that alter the absorption, distribution, metabolism, and excretion of quinine may increase or decrease quinine concentrations (see CLINICAL PHARMACOLOGY).
Antacids
Antacids containing aluminum and/or magnesium may delay or decrease absorption of quinine. Concomitant administration of these antacids with Qualaquin should be avoided.
Cholestyramine
In 8 healthy volunteers who received quinine sulfate 600 mg with or without 8 grams of cholestyramine resin, no significant difference in quinine pharmacokinetic parameters was seen.
Cigarette Smoking (CYP1A2 Inducer)
In healthy male heavy smokers, the mean quinine AUC following a single 600-mg dose was 44% lower, the mean Cmax was 18% lower, and the elimination half-life was shorter (7.5 hours versus 12 hours) than in their non-smoking counterparts. However, in malaria patients who received the full 7-day course of quinine therapy, cigarette smoking produced only a 25% decrease in median quinine AUC and a 16.5% decrease in median Cmax, suggesting that the already reduced clearance of quinine in acute malaria could have diminished the metabolic induction effect of smoking. Because smoking did not appear to influence the therapeutic outcome in malaria patients, it is not necessary to increase the dose of quinine in the treatment of acute malaria in heavy cigarette smokers.
Cimetidine, ranitidine (nonspecific CYP450 inhibitors)
In healthy volunteers who were given a single oral 600 mg dose of quinine sulfate after pretreatment with cimetidine (200 mg three times daily and 400 mg at bedtime for 7 days) or ranitidine (150 mg twice daily for 7 days), the apparent oral clearance of quinine decreased and the mean elimination half-life increased significantly when given with cimetidine but not with ranitidine. Compared to untreated controls, the mean AUC of quinine increased by only 20% with ranitidine and by 42% with cimetidine (p<0.05) without a significant change in mean quinine Cmax. When quinine is to be given concomitantly with a histamine H2-receptor blocker, the use of ranitidine is preferred over cimetidine. Although cimetidine may be used concomitantly with Qualaquin, patients should be monitored closely for adverse events associated with quinine.
Erythromycin (CYP3A4 inhibitor)
Erythromycin was shown to inhibit the metabolism of quinine in vitro using human liver microsomes. Therefore, concomitant administration of erythromycin with Qualaquin is likely to increase plasma quinine concentrations, and should be avoided (See WARNINGS).
Grapefruit juice (CYP3A4 inhibitor)
In a pharmacokinetic study involving 10 healthy volunteers, the administration of a single 600 mg dose of quinine sulfate with grapefruit juice (full-strength or half-strength) did not significantly alter the pharmacokinetic parameters of quinine. Qualaquin may be taken with grapefruit juice.
Histamine H2-receptor blockers (cimetidine, ranitidine)
In healthy volunteers who were given a single oral 600 mg dose of quinine sulfate after pretreatment with cimetidine (200 mg three times daily and 400 mg at bedtime for 7 days) or ranitidine (150 mg twice daily for 7 days), the apparent oral clearance of quinine decreased and the mean elimination half-life increased significantly when given with cimetidine but not with ranitidine. Compared to untreated controls, the mean AUC of quinine increased by only 20% with ranitidine and by 42% with cimetidine (p<0.05) without a significant change in mean quinine Cmax. When quinine is to be given concomitantly with a histamine H2-receptor blocker, the use of ranitidine is preferred over cimetidine. Although cimetidine may be used concomitantly with Qualaquin, patients should be monitored closely for adverse events associated with quinine.
Isoniazid
Isoniazid 300 mg/day pretreatment for 1 week did not significantly alter the pharmacokinetic parameters of quinine. Adjustment of Qualaquin dosage is not necessary when isoniazid is given concomitantly.
Ketoconazole (CYP3A4 inhibitor)
In a crossover study, healthy subjects (N=9) who received a single oral dose of quinine hydrochloride (500 mg) concomitantly with ketoconazole (100 mg twice daily for 3 days) had a mean quinine AUC that was higher by 45% and a mean oral clearance of quinine that was 31% lower than after receiving quinine alone. Although no change in the Qualaquin dosage regimen is necessary with concomitant ketoconazole, patients should be monitored closely for adverse reactions associated with quinine.
Oral contraceptives (estrogen, progestin)
In 7 healthy females who were using single-ingredient progestin or combination estrogen-containing oral contraceptives, the pharmacokinetic parameters of a single 600 mg dose of quinine sulfate were not altered in comparison to those observed in 7 age-matched female control subjects not using oral contraceptives.
Rifampin (CYP3A4 inducer)
In patients with uncomplicated P. falciparum malaria who received quinine sulfate 10 mg/kg concomitantly with rifampin 15 mg/kg/day for 7 days (N=29), the median AUC of quinine between days 3 and 7 of therapy was 75% lower as compared to those who received quinine monotherapy. In healthy volunteers (N=9) who received a single oral 600 mg dose of quinine sulfate after 2 weeks of pretreatment with rifampin 600 mg/day, the mean quinine AUC and Cmax decreased by 85% and 55%, respectively. Therefore the concomitant administration of rifampin with Qualaquin should be avoided (See WARNINGS).
Tetracycline
In 8 patients with acute uncomplicated P. falciparum malaria who were treated with oral quinine sulfate (600 mg every 8 hours for 7 days) in combination with oral tetracycline (250 mg every 6 hours for 7 days), the mean plasma quinine concentrations were about two-fold higher than in 8 patients who received quinine monotherapy. Although tetracycline may be concomitantly administered with Qualaquin, patients should be monitored closely for adverse reactions associated with quinine sulfate.
Troleandomycin (CYP3A4 inhibitor)
In a crossover study (N=10), healthy subjects who received a single oral 600 mg dose of quinine sulfate with the macrolide antibiotic, troleandomycin (500 mg every 8 hours) exhibited a 87% higher mean quinine AUC, a 45% lower mean oral clearance of quinine, and a 81% lower formation clearance of the main metabolite, 3-hydroxyquinine, than when quinine was given alone. Therefore, concomitant administration of troleandomycin with Qualaquin should be avoided (See WARNINGS).
Urinary alkalizers (acetazolamide, sodium bicarbonate)
Urinary alkalinizing agents may increase plasma quinine concentrations.
Effect of Quinine on the Pharmacokinetics of Other Drugs
Results of in vivo drug interaction studies suggest that quinine has the potential to inhibit the metabolism of drugs that are substrates of CYP3A4 and CYP2D6, as well as, inhibit the biliary excretion of drugs like digoxin.
In an in vitro induction study using human hepatocytes, quinine (5 to 30 µM) increased the metabolic activities of CYP1A2 and CYP3A4. Quinine did not significantly induce the activities of CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP2E1.
Anticonvulsants (carbamazepine, phenobarbital, and phenytoin)
A single 600 mg oral dose of quinine sulfate increased the mean plasma Cmax, and AUC0–24 of single oral doses of carbamazepine (200 mg) and phenobarbital (120 mg) but not phenytoin (200 mg) in 8 healthy subjects. The mean AUC increases of carbamazepine, phenobarbital and phenytoin were 104%, 81% and 4%, respectively; the mean increases in Cmax were 56%, 53%, and 4%, respectively. Mean urinary recoveries of the three antiepileptics over 24 hours were also profoundly increased by quinine. If concomitant administration with carbamazepine or phenobarbital cannot be avoided, frequent monitoring of anticonvulsant drug concentrations is recommended. Additionally, patients should be monitored closely for adverse reactions associated with these anticonvulsants. Carbamazepine, phenobarbital, and phenytoin are CYP3A4 inducers and may decrease quinine plasma concentrations if used concurrently with Qualaquin.
Astemizole (CYP3A4 substrate)
Elevated plasma astemizole concentrations were reported in a subject who experienced torsades de pointes after receiving three doses of quinine sulfate for nocturnal leg cramps concomitantly with chronic astemizole 10 mg/day. The concurrent use of Qualaquin with astemizole and other CYP3A4 substrates with QT prolongation potential (e.g., cisapride, terfenadine, halofantrine, pimozide and quinidine) should also be avoided (See WARNINGS).
Atorvastatin (CYP3A4 substrate)
Rhabdomyolysis with acute renal failure secondary to myoglobinuria was reported in a patient taking atorvastatin administered with a single dose of quinine. Quinine may increase plasma concentrations of atorvastatin, thereby increasing the risk of myopathy or rhabdomyolysis. Thus, clinicians considering combined therapy of Qualaquin with atorvastatin or other HMG-CoA reductase inhibitors ("statins") that are CYP3A4 substrates (e.g., simvastatin, lovastatin) should carefully weigh the potential benefits and risks of each medication. If Qualaquin is used concomitantly with any of these statins, lower starting and maintenance doses of the statin should be considered. Patients should also be monitored closely for any signs or symptoms of muscle pain, tenderness, or weakness, particularly during initial therapy. If marked creatine phosphokinase (CPK) elevation occurs or myopathy (defined as muscle aches or muscle weakness in conjunction with CPK values >10 times the upper limit of normal) is diagnosed or suspected, atorvastatin or other statin should be discontinued.
Desipramine (CYP2D6 substrate)
Quinine (750 mg/day for 2 days) decreased the metabolism of desipramine in patients who were extensive CYP2D6 metabolizers, but had no effect in patients who were poor CYP2D6 metabolizers. Lower doses (80 mg to 400 mg) of quinine did not significantly affect the pharmacokinetics of other CYP2D6 substrates, namely, debrisoquine, dextromethorphan, and methoxyphenamine. Although clinical drug interaction studies have not been performed, antimalarial doses (greater than or equal to 600 mg) of quinine may inhibit the metabolism of other drugs that are CYP2D6 substrates (e.g., flecainide, debrisoquine, dextromethorphan, metoprolol, paroxetine). Patients taking medications that are CYP2D6 substrates with Qualaquin should be monitored closely for adverse reactions associated with these medications.
Digoxin
In 4 healthy subjects who received digoxin (0.5 to 0.75 mg/day) during treatment with quinine (750 mg/day), a 33% increase in mean steady state AUC of digoxin and a 35% reduction in the steady-state biliary clearance of digoxin were observed compared to digoxin alone. Thus, if Qualaquin is administered to patients receiving digoxin, plasma digoxin concentrations should be closely monitored, and the digoxin dose adjusted, as necessary (See PRECAUTIONS).
Halofantrine
Although not studied clinically, quinine was shown to inhibit the metabolism of halofantrine in vitro using human liver microsomes. Therefore, concomitant administration of Qualaquin is likely to increase plasma halofantrine concentrations (See WARNINGS).
Mefloquine
In 7 healthy subjects who received mefloquine (750 mg) at 24 hours before an oral 600 mg dose of quinine sulfate, the AUC of mefloquine was increased by 22% compared to mefloquine alone. In this study, the QTc interval was significantly prolonged in the subjects who received mefloquine and quinine sulfate 24 hours apart. The concomitant administration of mefloquine and Qualaquin may produce electrocardiographic abnormalities (including QTc prolongation) and may increase the risk of seizures (See WARNINGS).
Neuromuscular blocking agents (pancuronium, succinylcholine, tubocurarine)
In one report, quinine potentiated neuromuscular blockade in a patient who received pancuronium during an operative procedure, and subsequently (3 hours after receiving pancuronium) received quinine 1800 mg daily. Quinine may also enhance the neuromuscular blocking effects of succinylcholine and tubocurarine (See WARNINGS).
Theophylline or aminophylline (CYP1A2 substrate)
Although not studied clinically, quinine has been shown to induce the activity of CYP1A2 in vitro using human hepatocytes. Therefore, concomitant administration of Qualaquin and theophylline or aminophylline is likely to decrease the plasma theophylline concentrations, possibly reducing the effect of theophylline or aminophylline. Plasma theophylline concentrations should be monitored frequently during concurrent therapy with theophylline or aminophylline and Qualaquin.
Warfarin and oral anticoagulants
Cinchona alkaloids, including quinine, may have the potential to depress hepatic enzyme synthesis of vitamin K-dependent coagulation pathway proteins and may enhance the action of warfarin and other oral anticoagulants. Quinine may also interfere with the anticoagulant effect of heparin. Thus, in patients receiving these anticoagulants, the prothrombin time (PT), partial thromboplastin time (PTT), or international normalization ratio (INR) should be closely monitored as appropriate, during concurrent therapy with Qualaquin.
Drug/Laboratory Interactions
Quinine may produce an elevated value for urinary 17-ketogenic steroids when the Zimmerman method is used.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies of quinine have not been conducted.
Genotoxicity studies of quinine were positive in the Ames bacterial mutation assay with metabolic activation and in the sister chromatid exchange assay in mice. There were non-positive genotoxicity findings in the sex-linked recessive lethal test performed in Drosophila, in the in vivo mouse micronucleus assay, and in the chromosomal aberration assay in mice and Chinese hamsters.
Studies to evaluate the effect of quinine upon fertility in animals or in humans have not been conducted.
Pregnancy
Category C
There are no adequate and well-controlled studies in pregnant women.
Hypoglycemia, due to increased pancreatic secretion of insulin, has been associated with quinine use, particularly in pregnant women.
Quinine crosses the placenta and gives measurable blood concentrations in the fetus. In 8 women who delivered live infants 1 to 6 days after starting quinine therapy, placental cord plasma quinine concentrations were between 1.0 and 4.6 mg/L (mean 2.4 mg/L) and the mean (±SD) ratio of cord plasma to maternal plasma quinine concentrations was 0.32 ± 0.14 (See CLINICAL PHARMACOLOGY).
Quinine levels in the fetus may not be therapeutic. If congenital malaria is suspected after delivery, the infant should be evaluated and treated appropriately.
Rare and isolated case reports describe deafness and optic nerve hypoplasia in children exposed in utero due to maternal ingestion of high doses of quinine.
A study from Thailand (1999) of women with P. falciparum malaria who were treated with oral quinine sulfate 10 mg/kg 3 times daily for 7 days at anytime in pregnancy reported no significant difference in the rate of stillbirths at >28 weeks of gestation in women treated with quinine (10 of 633 women [1.6%]) as compared with a control group without malaria or exposure to antimalarial drugs during pregnancy (40 of 2201 women [1.8%]). The overall rate of congenital malformations (9 of 633 offspring [1.4%]) was not different for women who were treated with quinine sulfate compared with the control group (38 of 2201 offspring [1.7%]). The spontaneous abortion rate was higher in the control group (10.9%) than in women treated with quinine sulfate (3.5%) [OR = 3.1; 95% CI 2.1–4.7].
In an epidemiologic survey that included 104 mother-child pairs exposed to quinine during the first 4 months of pregnancy, no increased risk of structural birth defects was seen (2 fetal malformations [1.9%]).
P. falciparum malaria carries a higher risk of morbidity and mortality in pregnant women than in the general population. Pregnant women with P. falciparum malaria have an increased incidence of fetal loss (including spontaneous abortion and stillbirth), preterm labor and delivery, intrauterine growth retardation, low birth weight, and maternal death. Therefore, treatment of malaria in pregnancy is important. Qualaquin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. The risks and benefits of alternative treatments should be considered. If Qualaquin is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazards to the fetus.
Teratogenic effects have been demonstrated in some animal species but not in others when quinine was given by the subcutaneous or intramuscular route at dose levels in the same range as the maximum recommended human dose. Teratogenic effects were observed in rabbits (death in utero, degenerated auditory nerve and spiral ganglion, and CNS anomalies such as anencephaly and microcephaly), dogs (death in utero), guinea pigs (hemorrhage and mitochondrial change in cochlea), and chinchillas (death and growth suppression in utero and CNS anomalies, such as anencephaly and microcephaly). There were no teratogenic findings in mice, rats, and monkeys.
Labor and Delivery
There is no evidence that quinine causes uterine contractions at the doses recommended for the treatment of malaria. In doses several-times higher than those used to treat malaria, quinine may stimulate the pregnant uterus.
Nursing Mothers
There is limited information on the safety of quinine in breastfed infants. No toxicity was reported in infants in a single study where oral quinine sulfate (10 mg/kg every 8 hours for 1 to 10 days) was administered to 25 lactating women. It is estimated from this study that breastfed infants would receive less than 2 to 3 mg per day of quinine base (< 0.4% of the maternal dose) via breast milk (See CLINICAL PHARMACOLOGY).
Although quinine is generally considered compatible with breastfeeding, the risks and benefits to infant and mother should be assessed.
If malaria is suspected in the infant, appropriate evaluation and treatment should be provided. Plasma quinine levels may not be therapeutic in infants of nursing mothers receiving Qualaquin.
Pediatric Use
The safety and efficacy of Qualaquin in pediatric patients under the age of 16 has not been established.
Geriatric Use
Clinical studies of quinine sulfate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
ADVERSE REACTIONS
Quinine can adversely affect almost every body system. The most common adverse events associated with quinine use are a cluster of symptoms called "cinchonism", which occurs to some degree in almost all patients taking quinine. Symptoms of mild cinchonism include headache, vasodilation and sweating, nausea, tinnitus, hearing impairment, vertigo or dizziness, blurred vision, and disturbance in color perception. More severe symptoms of cinchonism are vomiting, diarrhea, abdominal pain, deafness, blindness, and disturbances in cardiac rhythm or conduction. Most symptoms of cinchonism are reversible and resolve with discontinuation of quinine.
The following ADVERSE REACTIONS have been reported with quinine sulfate. Most of these reactions are thought to be uncommon, but the actual incidence is unknown:
General: fever, chills, sweating, flushing, asthenia, lupus-like syndrome, and hypersensitivity reactions (See WARNINGS and PRECAUTIONS).
Hematologic: agranulocytosis, hypoprothrombinemia, thrombocytopenia, disseminated intravascular coagulation, hemolytic anemia; hemolytic uremic syndrome, thrombotic thrombocytopenic purpura, idiopathic thrombocytopenic purpura, petechiae, ecchymosis, hemorrhage, coagulopathy, blackwater fever, leukopenia, neutropenia, pancytopenia, aplastic anemia, and lupus anticoagulant.
Neuropsychiatric: headache, diplopia, confusion, altered mental status, seizures, coma, disorientation, tremors, restlessness, ataxia, acute dystonic reaction, aphasia, and suicide.
Dermatologic: cutaneous rashes, including urticarial, papular, or scarlatinal rashes, pruritus, bullous dermatitis, exfoliative dermatitis, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, fixed drug eruption, photosensitivity reactions, allergic contact dermatitis, acral necrosis, and cutaneous vasculitis.
Respiratory: asthma, dyspnea, pulmonary edema.
Cardiovascular: chest pain, vasodilatation, hypotension, postural hypotension, tachycardia, bradycardia, palpitations, syncope, atrioventricular block, atrial fibrillation, irregular rhythm, unifocal premature ventricular contractions, nodal escape beats, U waves, QT prolongation, ventricular fibrillation, ventricular tachycardia, torsades de pointes, and cardiac arrest (See WARNINGS).
Gastrointestinal: nausea, vomiting, diarrhea, abdominal pain, gastric irritation, and esophagitis.
Hepatobiliary: granulomatous hepatitis, hepatitis, jaundice, and abnormal liver function tests.
Metabolic: hypoglycemia and anorexia.
Musculoskeletal: myalgias and muscle weakness.
Renal: hemoglobinuria, renal failure, renal impairment, and acute interstitial nephritis.
Special Senses: visual disturbances, including blurred vision with scotomata, sudden loss of vision, photophobia, diplopia, night blindness, diminished visual fields, fixed pupillary dilatation, disturbed color vision, optic neuritis, blindness, vertigo, tinnitus, hearing impairment, and deafness.
DRUG ABUSE AND DEPENDENCE
Tolerance, abuse, or dependence with quinine sulfate has not been reported.
OVERDOSAGE
Quinine overdose can be associated with serious complications, including visual impairment, hypoglycemia, cardiac arrhythmias, and death. Visual impairment can range from blurred vision and defective color perception, to visual field constriction and permanent blindness. Cinchonism occurs in virtually all patients with quinine overdose. Symptoms range from headache, nausea, vomiting, abdominal pain, diarrhea, tinnitus, vertigo, hearing impairment, sweating, flushing, and blurred vision, to deafness, blindness, serious cardiac arrhythmias, hypotension, and circulatory collapse. Central nervous system toxicity (drowsiness, disturbances of consciousness, ataxia, convulsions, respiratory depression and coma) has also been reported with quinine overdose, as well as pulmonary edema and adult respiratory distress syndrome.
Most toxic reactions are dose-related; however some reactions may be idiosyncratic because of the variable sensitivity of patients to the toxic effects of quinine. A lethal dose of quinine has not been clearly defined, but fatalities have been reported after the ingestion of 2 to 8 grams in adults.
Quinine, like quinidine, has Class I antiarrhythmic properties. The cardiotoxicity of quinine is due to its negative inotropic action, and to its effect on cardiac conduction, resulting in decreased rates of depolarization and conduction, and increased action potential and effective refractory period. ECG changes observed with quinine overdose include sinus tachycardia, PR prolongation, T wave inversion, bundle branch block, an increased QT interval, and a widening of the QRS complex. Quinine's alpha-blocking properties may result in hypotension and further exacerbate myocardial depression by decreasing coronary perfusion. Quinine overdose has been also associated with hypotension, cardiogenic shock, and circulatory collapse, ventricular arrhythmias, including ventricular tachycardia, ventricular fibrillation, idioventricular rhythm, and torsades de pointes, as well as bradycardia, and atrioventricular block (See WARNINGS, PRECAUTIONS, and ADVERSE EVENTS).
Quinine is rapidly absorbed, and attempts to remove residual quinine sulfate from the stomach by gastric lavage may not be effective. Multiple-dose activated charcoal has been shown to decrease plasma quinine concentrations (See CLINICAL PHARMACOLOGY/Extracorporeal elimination).
Forced acid diuresis, hemodialysis, charcoal column hemoperfusion, and plasma exchange were not found to be effective in significantly increasing quinine elimination in a series of 16 patients.
DOSAGE AND ADMINISTRATION
(SEE INDICATIONS AND USAGE)
For treatment of uncomplicated P. falciparum malaria in adults, the Qualaquin dosage is 648 mg (two capsules) every 8 hours for 7 days (See CLINICAL STUDIES).
Qualaquin should be taken with food to minimize gastric upset (See CLINICAL PHARMACOLOGY).
For patients with hepatic impairment
In otherwise healthy subjects with Child-Pugh B hepatic impairment, the AUC of quinine increased by 55% compared to subjects with normal liver function. In patients with mild to moderate hepatic impairment (Child-Pugh A and Child-Pugh B, respectively), dosage reduction is not warranted but patients should be monitored closely for adverse reactions associated with quinine (See CLINICAL PHARMACOLOGY/Special Populations). The effects of severe hepatic impairment (Child-Pugh C) on the safety and pharmacokinetics of quinine sulfate are not known.
For patients with renal impairment
In otherwise healthy subjects with severe chronic renal failure not receiving any form of dialysis (mean serum creatinine = 9.6 mg/dL), the median plasma quinine exposure (AUC) increased by 195% compared to subjects with normal renal function. In patients with acute uncomplicated malaria and severe chronic renal failure, the following modified dosage regimen is recommended: one loading dose of 648 mg Qualaquin followed 12 hours later by maintenance doses of 324 mg every 12 hours (See CLINICAL PHARMACOLOGY/Special Populations). The effects of mild and moderate renal impairment on the pharmacokinetics and safety of quinine sulfate are not known.
HOW SUPPLIED
Qualaquin capsules USP, 324 mg are available as clear/clear capsules imprinted AR 102:
| Bottles of 30 | NDC 13310-153-07 |
| Bottles of 100 | NDC 13310-153-01 |
| Bottles of 500 | NDC 13310-153-05 |
| Bottles of 1000 | NDC 13310-153-10 |
Store at 25– 30°C (77–86°F).
Dispense in a tight container as defined in the USP.
CLINICAL STUDIES
Quinine has been used worldwide for hundreds of years in the treatment of malaria. Thorough searches of the published literature identified over 1300 references to the treatment of malaria with quinine, and from these, 21 randomized, active-controlled studies were identified which evaluated oral quinine monotherapy or combination therapy for treatment of uncomplicated P. falciparum malaria. Over 2900 patients from malaria-endemic areas were enrolled in these studies, and more than 1400 patients received oral quinine. The following conclusions were drawn from review of these studies:
In areas where multi-drug resistance of P. falciparum is increasing, such as Southeast Asia, cure rates with 7 days of oral quinine monotherapy were at least 80%; while cure rates for 7 days of oral quinine combined with an antimicrobial agent (tetracycline or clindamycin) were greater than 90%. In areas where multi-drug resistance of the parasite was not as widespread, cure rates with 7 days of quinine monotherapy ranged from 86 to 100%. Cure was defined as initial clearing of parasitemia within 7 days without recrudescence by day 28 after treatment initiation. P. falciparum malaria that is clinically resistant to quinine has been reported in some areas of South America, Southeast Asia, and Bangladesh, and quinine may not be as effective in those areas.
Completion of a 7-day oral quinine treatment regimen may be limited by drug intolerance, and shorter courses (3 days) of quinine combination therapy have been used. However, the published data from randomized, controlled clinical trials for shorter regimens of oral quinine in conjunction with tetracycline, doxycycline, or clindamycin for treatment of uncomplicated P. falciparum malaria is limited, and these shorter course combination regimens may not be as effective as the longer regimens.
REFERENCES
- Kaufman DW, Kelly JP, Johannes CB, et al. Acute thrombocytopenic purpura in relation to the use of drugs, Blood, 1993;82: 2714–2718
Manufactured for:
AR SCIENTIFIC, INC.
Philadelphia, PA 19124 USA
by:
MUTUAL PHARMACEUTICAL COMPANY, INC.
Philadelphia, PA 19124 USA
Rev 04, June 2009
Patient Information
QUALAQUIN®
quinine sulfate
CAPSULES USP, 324 mg
This leaflet contains a summary of the most important information about QUALAQUIN® capsules and should be read completely before starting your treatment. This leaflet does not replace talking to your doctor or health care provider about your treatment or medical condition. If you have any questions about your treatment or medical condition, ask your doctor. Only your doctor or other health care provider can prescribe QUALAQUIN® and determine if it is right for you.
Malaria is a serious infection, and if not treated, can be life-threatening. Quinine Sulfate has been used for many years as an effective treatment for uncomplicated malaria caused by the parasite Plasmodium falciparum.
What is the most important information I should know about QUALAQUIN®?
QUALAQUIN® may cause serious side effects or even death if QUALAQUIN® levels are too high in your body. Keep QUALAQUIN® out of the reach of children.
- Quinine may cause problems with the heart rhythm. This can be made worse when QUALAQUIN® is taken with some other medicines. Tell your doctor if you are taking any other medications or if you have any heart rhythm problems.
- Quinine, especially when taken for the non-approved condition of leg cramps, has been associated with bleeding. If you notice easy bruising, bleeding, or a non-fading pinpoint type rash (which may be due to small bleeds under the skin), call your doctor immediately. If you are taking QUALAQUIN® for malaria do not stop unless your doctor tells you to stop. If you are taking QUALAQUIN® for any other reason, stop taking it immediately.
Tell your healthcare provider about all your medical conditions, including if you have a known sensitivity to quinine. Your dose of QUALAQUIN® may need to be changed or discontinued.
Certain medicines when taken with QUALAQUIN® can cause the levels of QUALAQUIN® to be too high in your body. It is important for you to tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. QUALAQUIN® and other medicines may affect each other causing serious side effects or even death. Do not start taking a new medicine without telling your healthcare provider or pharmacist.
Even medications that you might take for a short period of time, such as antibiotics, can interact with QUALAQUIN® and cause serious side effects or death.
Especially tell your healthcare provider if you take:
- Antacids containing aluminum and/or magnesium
- Cimetidine (Tagamet®)
- Erythromycin
- Ketoconazole (Nizoral®)
- Itraconazole (Sporanox®)
- Rifampin (Rifadin®)
- Tetracycline
- Troleandomycin (Tao®)
- Urinary alkalizers (acetazolamide, sodium bicarbonate)
- Anticonvulsants (carbamazepine, Phenobarbital, and phenytoin)
- Astemizole
- Atorvastatin (Lipitor®)
- Desipramine (Norpramin®)
- Digoxin
- Halofantrine (Halfan®)
- Mefloquine
- Neuromuscular blocking agents (pancuronium, succinylcholine, tubocurarine)
- Theophylline or aminophylline
- Warfarin and oral anticoagulants
This is not a complete list of all the medicines that can interact with QUALAQUIN®. Talk to your healthcare provider or pharmacist to find out if taking QUALAQUIN® with the other medicines you are taking could be dangerous.
Talk to your healthcare provider before taking any new medicine.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
What is QUALAQUIN®?
QUALAQUIN® is a prescription medication used in the treatment of uncomplicated malaria caused by the parasite Plasmodium falciparum.
QUALAQUIN® is NOT approved for the prevention of malaria or for the prevention or treatment of night-time leg cramps.
Who should not take QUALAQUIN®?
Do not take QUALAQUIN® if you:
- Had previous allergic reactions to quinine, quinidine, or mefloquine (Lariam®).
- Had previous serious side effects to quinine, such as decreased platelets, which are components of blood necessary for clotting.
- Have low levels of an enzyme called Glucose-6-phosphate dehydrogenase (G-6-PD).
- Have myasthenia gravis.
- Have optic neuritis, which is an inflammation of the nerve important for vision.
- Have certain heart rhythm problems or certain inherited abnormalities on your electrocardiogram (ECG). Your doctor will tell you whether your ECG has these abnormalities.
What should I tell my healthcare provider before starting QUALAQUIN®?
Tell your doctor or health care provider:
- About all your medical conditions, including any heart, kidney, or liver problems.
- About all the prescription and non-prescription medications you are taking, including vitamins and herbal medications.
- If you are pregnant or could be pregnant. Treatment of malaria is important because it can be a serious disease for a pregnant woman and her unborn baby. Your doctor can tell you more about the benefits and risks of taking this medication during pregnancy for uncomplicated malaria. You and your doctor can decide if QUALAQUIN® is right for you.
- If you are breast-feeding. Small amounts of QUALAQUIN® can pass into the breast milk, but no problems with this medicine have been reported in nursing babies. Discuss with your doctor whether you should breastfeed while taking QUALAQUIN®.
How should I take QUALAQUIN®?
- Take QUALAQUIN® exactly as prescribed.
- QUALAQUIN® is a clear capsule that is taken by mouth.
- Unless directed otherwise by your doctor, the usual dose is 648 mg (two 324 mg capsules) of QUALAQUIN® every 8 hours by mouth at the same time every day for 7 days.
- To lower the chance of stomach upset, take this medication WITH FOOD.
- Finish all the QUALAQUIN® that is prescribed even if you feel better. Do not stop taking the medication without talking to your doctor.
- Do not take more than the amount prescribed. Do not take more than 2 capsules at one time or more than 3 doses in one day. If you take more than the prescribed dose, call your doctor right away.
What should I do if I miss a dose?
If you forget to take QUALAQUIN®, do NOT double the next dose. If it has been more than 4 hours since the missed dose, WAIT and take the regular dose at the next scheduled time. Call your doctor if you are not sure what to do.
What are the possible side effects of QUALAQUIN®?
If you experience bruising or bleeding from the nose or gums, discontinue QUALAQUIN® and notify your doctor immediately.
The most common side effects that you may have when taking QUALAQUIN® are not usually serious, and will usually get better when QUALAQUIN® is stopped. Common side effects with QUALAQUIN® include:
- Headache
- Sweating
- Ringing in your ears
- Dizziness
- Change in color vision
- Nausea
- Flushing
- Mild hearing loss
- Blurred vision
Occasionally, more severe symptoms such as vomiting, diarrhea, and abdominal pain may occur. Rarely, rapid or irregular heartbeat, severe hearing loss, or blindness, may occur. If you experience any severe side effects, call your doctor.
Some patients may experience low blood sugar (hypoglycemia) while taking QUALAQUIN®. Symptoms of low blood sugar include lightheadedness, dizziness, sweating, confusion, shakiness, anxiety, and weakness. If you experience symptoms of low blood sugar, drink some fruit juice or eat a snack, and call your doctor.
Elderly patients may be more sensitive to the side effects of QUALAQUIN® than younger patients, and should quickly report any side effects to their doctor.
QUALAQUIN® has other less common side effects that are not listed here. For a complete list of side effects, ask your doctor. If you notice any side effects not mentioned in this leaflet, or if you have any concerns about a side effect you are having, talk to your doctor.
QUALAQUIN® is NOT approved for the treatment of leg cramps because quinine has not been proven to work for this condition, and may cause serious or life-threatening side effects. Some of the more serious side effects of quinine are blindness, deafness, and abnormal heart rhythm. Your doctor can tell you additional information about serious side effects reported with quinine.
Call your doctor or health care provider right away if:
- You feel worse; or if you do not start feeling better within a day or two of taking QUALAQUIN®.
- If your fevers come back after completing treatment with QUALAQUIN®, call your doctor to make sure that the malaria has not returned.
- You experience serious problems such as:
- Serious allergic reactions: rash, hives, severe itching, severe flushing, trouble breathing.
- Eyesight problems: blurred vision, double vision, blindness.
- Heart problems: chest pain, rapid heartbeats, abnormal heart rhythm.
- Other reactions: dizziness, confusion, lightheadedness, fainting, seizure.
- Other problems: abnormal bleeding, (such as severe nosebleed, and blood in the urine, or stool), severe bruising, or the appearance of unusual purple-brown or red spots on your skin.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of QUALAQUIN®. For more information, ask your healthcare provider or pharmacist.
How do I store QUALAQUIN®?
Keep Qualaquin out of reach of children. Keep the capsules in a tightly closed container. Do not refrigerate or freeze. Store at 25– 30°C (77–86°F).
General advice about QUALAQUIN®:
Do not use QUALAQUIN® for a condition for which it was not prescribed. Do NOT give QUALAQUIN® to other people, even if they have the same symptoms, because it may be harmful.
This leaflet highlights the most important information about QUALAQUIN®. For more information, you should talk with your doctor or health care provider.
Active Ingredients: Quinine Sulfate, USP
Inactive Ingredients: Corn starch, magnesium stearate, talc
Rev 04, June 2009
PRINCIPAL DISPLAY PANEL - 30 Capsule Bottle
30 CAPSULES
NDC 13310-153-07
QUALAQUIN®
quinine sulfate
capsules USP
324 mg
AR
SCIENTIFIC
Rx only

| QUALAQUIN
quinine sulfate capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA021799 | 07/10/2006 | |
| Labeler - AR Scientific, Inc. (606563521) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Mutual Pharmaceutical Company Inc. | 121735955 | MANUFACTURE | |