PROPOXYPHENE AND ACETAMINOPHEN
-
acetaminophen and
propoxyphene napsylate tablet, film coated
Qualitest Pharmaceuticals
----------
PROPOXYPHENE NAPSYLATE AND ACETAMINOPHEN TABLETS, USPCIV
Rx only
DESCRIPTION:
Propoxyphene Napsylate, USP is an odorless, white, crystalline solid with a bitter taste. It is very slightly soluble in water and soluble in methanol, ethanol, chloroform, and acetone. Chemically, it is (αS,1R)-α-[2-(Dimethylamino)-1-methylethyl]-α-phenylphenethyl propionate compound with 2-napthalenesulfonic acid (1:1) monohydrate, which can be represented by the accompanying structural formula:
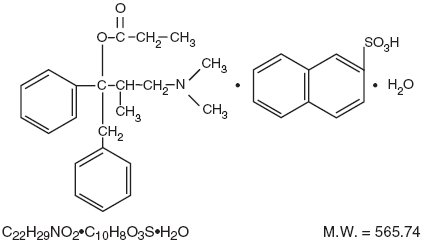
Propoxyphene napsylate differs from propoxyphene hydrochloride in that it allows more stable liquid dosage forms and tablet formulations. Because of differences in molecular weight, a dose of 100 mg (176.8 mcmol) of propoxyphene napsylate is required to supply an amount of propoxyphene equivalent to that present in 65 mg (172.9 mcmol) of propoxyphene hydrochloride.
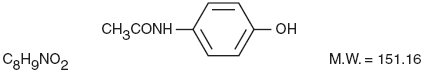
The acetaminophen component is 4'-Hydroxyacetanilide, a white, odorless, crystalline powder possessing a slightly bitter taste, and is represented by the following structural formula:
Propoxyphene napsylate and acetaminophen is supplied in tablet form for oral administration.
Propoxyphene Napsylate and Acetaminophen Tablets, USP 50 mg/325 mg
Each tablet contains:
Propoxyphene Napsylate ............................50 mg
Acetaminophen .........................................325 mg
In addition each tablet contains the following inactive ingredients: carnauba wax, D&C Yellow #10 Aluminum Lake, FD&C Red #40 Aluminum Lake, hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, polysorbate 80, sodium starch glycolate and titanium dioxide.
Propoxyphene Napsylate and Acetaminophen Tablets, USP 100 mg/650 mg
Each tablet contains:
Propoxyphene Napsylate ...........................100 mg
Acetaminophen ..........................................650 mg
In addition each tablet contains the following inactive ingredients: carnauba wax, hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, polysorbate 80, sodium starch glycolate and titanium dioxide. The orange tablets also contain D&C Yellow #10 Aluminum Lake and FD&C Red #40 Aluminum Lake. The pink tablets also contain FD&C Red #40 Aluminum Lake.
CLINICAL PHARMACOLOGY:
Propoxyphene is a centrally acting narcotic analgesic agent. Equimolar doses of propoxyphene hydrochloride or napsylate provide similar plasma concentrations. Following administration of 65, 130, or 195 mg of propoxyphene hydrochloride, the bioavailability of propoxyphene is equivalent to that of 100, 200, or 300 mg respectively of propoxyphene napsylate. Peak plasma concentrations of propoxyphene are reached in 2 to 2 ½ hours. After a 100 mg oral dose of propoxyphene napsylate, peak plasma levels of 0.05 to 0.1 mcg/mL are achieved. As shown in Figure 1, the napsylate salt tends to be absorbed more slowly than the hydrochloride. At or near therapeutic doses, this absorption difference is small when compared with that among subjects and among doses.
Because of this several hundredfold difference in solubility, the absorption rate of very large doses of the napsylate salt is significantly lower than that of equimolar doses of the hydrochloride.
Repeated doses of propoxyphene at 6 hour intervals lead to increasing plasma concentrations, with a plateau after the ninth dose at 48 hours.
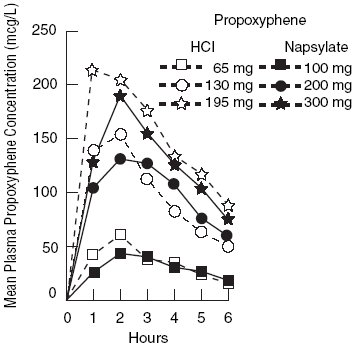
Figure 1. Mean plasma concentrations of propopxyphene in 8 human subjects following oral administration of 65 and 130 mg of the hydrochloride salt and 100 mg and 200 mg of the napsylate salt and in 7 given 195 mg of the hydrochloride and 300 mg of the napyslate salt.
Propoxyphene is metabolized in the liver to yield norpropoxyphene. Propoxyphene has a half-life of 6 to 12 hours, whereas that of norpropoxyphene is 30 to 36 hours.
Norpropoxyphene has substantially less central nervous system depressant effect than propoxyphene but a greater local anesthetic effect, which is similar to that of amitriptyline and antiarrhythmic agents such as lidocaine and quinidine.
In animal studies in which propoxyphene and norpropoxyphene were continuously infused in large amounts, intracardiac conduction time (PR and QRS intervals) was prolonged. Any intracardiac conduction delay attributable to high concentrations of norpropoxyphene may be of relatively long duration.
ACTIONS:
Propoxyphene is a mild narcotic analgesic structurally related to methadone. The potency of propoxyphene napsylate is from two-thirds to equal that of codeine.
Propoxyphene napsylate and acetaminophen tablets provide the analgesic activity of propoxyphene napsylate and the antipyretic-analgesic activity of acetaminophen.
The combination of propoxyphene and acetaminophen produces greater analgesia than that produced by either propoxyphene or acetaminophen administered alone.
INDICATIONS AND USAGE:
Propoxyphene napsylate and acetaminophen tablets are indicated for the relief of mild to moderate pain, either when pain is present alone or when it is accompanied by fever.
CONTRAINDICATIONS:
Hypersensitivity to propoxyphene or to acetaminophen.
WARNINGS:
- Do not prescribe propoxyphene for patients who are suicidal or addiction-prone.
- Prescribe propoxyphene with caution for patients taking tranquilizers or antidepressant drugs and patients who use alcohol in excess.
- Tell your patients not to exceed the recommended dose and to limit their intake of alcohol.
Propoxyphene products in excessive doses, either alone or in combination with other CNS depressants, including alcohol, are a major cause of drug-related deaths. Fatalities within the first hour of overdosage are not uncommon. In a survey of deaths due to overdosage conducted in 1975, in approximately 20% of the fatal cases, death occurred within the first hour (5% occurred within 15 minutes). Propoxyphene should not be taken in doses higher than those recommended by the physician. The judicious prescribing of propoxyphene is essential to the safe use of this drug. With patients who are depressed or suicidal, consideration should be given to the use of non-narcotic analgesics. Patients should be cautioned about the concomitant use of propoxyphene products and alcohol because of potentially serious CNS-additive effects of these agents. Because of its added depressant effects, propoxyphene should be prescribed with caution for those patients whose medical condition requires the concomitant administration of sedatives, tranquilizers, muscle relaxants, antidepressants, or other CNS-depressant drugs. Patients should be advised of the additive depressant effects of these combinations.
Many of the propoxyphene-related deaths have occurred in patients with previous histories of emotional disturbances or suicidal ideation or attempts as well as histories of misuse of tranquilizers, alcohol, and other CNS-active drugs. Some deaths have occurred as a consequence of the accidental ingestion of excessive quantities of propoxyphene alone or in combination with other drugs. Patients taking propoxyphene should be warned not to exceed the dosage recommended by the physician.
Drug Dependence
Propoxyphene, when taken in higher-than-recommended doses over long periods of time, can produce drug dependence characterized by psychic dependence and less frequently, physical dependence and tolerance. Propoxyphene will only partially suppress the withdrawal syndrome in individuals physically dependent on morphine or other narcotics. The abuse liability of propoxyphene is qualitatively similar to that of codeine although quantitatively less, and propoxyphene should be prescribed with the same degree of caution appropriate to the use of codeine.
Usage in Ambulatory Patients
Propoxyphene may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a car or operating machinery. The patient should be cautioned accordingly.
PRECAUTIONS:
General
Propoxyphene should be administered with caution to patients with hepatic or renal impairment since higher serum concentrations or delayed elimination may occur.
Information for Patients
A Patient Information Sheet is available for this product. See text below.
Drug Interactions
The CNS-depressant effect of propoxyphene is additive with that of other CNS depressants, including alcohol.
As is the case with many medicinal agents, propoxyphene may slow the metabolism of a concomitantly administered drug. Should this occur, the higher serum concentrations of that drug may result in increased pharmacologic or adverse effects of that drug. Such occurrences have been reported when propoxyphene was administered to patients on antidepressants, anticonvulsants, or warfarin-like drugs. Severe neurologic signs, including coma, have occurred with concurrent use of carbamazepine.
Pregnancy
Safe use in pregnancy has not been established relative to possible adverse effects on fetal development. Instances of withdrawal symptoms in the neonate have been reported following usage during pregnancy. Therefore, propoxyphene should not be used in pregnant women unless, in the judgement of the physician, the potential benefits outweigh the possible hazards.
Nursing Mothers
Low levels of propoxyphene have been detected in human milk. In postpartum studies involving nursing mothers who were given propoxyphene, no adverse effects were noted in infants receiving mother's milk. Caution should be exercised when propoxyphene napsylate and acetaminophen tablets are administered to a nursing woman.
Pediatric Use
Propoxyphene is not recommended for use in pediatric patients because documented clinical experience has been insufficient to establish safety and a suitable dosage regimen in the pediatric age group.
Geriatric Use
The rate of propoxyphene metabolism may be reduced in some patients. Increased dosing interval should be considered.
ADVERSE REACTIONS:
In a survey conducted in hospitalized patients, less than 1% of patients taking propoxyphene hydrochloride at recommended doses experienced side effects. The most frequently reported were dizziness, sedation, nausea, and vomiting. Some of these adverse reactions may be alleviated if the patient lies down. Other adverse reactions include constipation, abdominal pain, skin rashes, lightheadedness, headache, weakness, euphoria, dysphoria, hallucinations, and minor visual disturbances.
Liver dysfunction has been reported in association with both active components of propoxyphene napsylate and acetaminophen tablets. Propoxyphene therapy has been associated with abnormal liver function tests and, more rarely, with instances of reversible jaundice (including cholestatic jaundice). Hepatic necrosis may result from acute overdose of acetaminophen (see OVERDOSAGE). In chronic ethanol abusers, this has been reported rarely with short-term use of acetaminophen doses of 2.5 to 10 g/day. Fatalities have occurred.
Renal papillary necrosis may result from chronic acetaminophen use, particularly when the dosage is greater than recommended and when combined with aspirin. Subacute painful myopathy has occurred following chronic propoxyphene overdosage.
OVERDOSAGE:
In all cases of suspected overdosage, call your regional Poison Control Center to obtain the most up-to-date information about the treatment of overdose. This recommendation is made because, in general, information regarding the treatment of overdosage may change more rapidly than do package inserts. Initial consideration should be given to the management of the CNS effects of propoxyphene overdosage. Resuscitative measures should be initiated promptly.
Symptoms of Propoxyphene Overdosage
The manifestations of acute overdosage with propoxyphene are those of narcotic overdosage. The patient is usually somnolent but may be stuporous or comatose and convulsing. Respiratory depression is characteristic. The ventilatory rate and/or tidal volume is decreased, which results in cyanosis and hypoxia. Pupils, initially pinpoint, may become dilated as hypoxia increases. Cheyne-Stokes respiration and apnea may occur. Blood pressure and heart rate are usually normal initially, but blood pressure falls and cardiac performance deteriorates, which ultimately results in pulmonary edema and circulatory collapse, unless the respiratory depression is corrected and adequate ventilation is restored promptly. Cardiac arrhythmias and conduction delay may be present. A combined respiratory-metabolic acidosis occurs owing to retained CO2 (hypercapnia) and to lactic acid formed during anaerobic glycolysis. Acidosis may be severe if large amounts of salicylates have also been ingested. Death may occur.
Treatment of Propoxyphene Overdosage
Attention should be directed first to establishing a patent airway and to restoring ventilation. Mechanically assisted ventilation, with or without oxygen, may be required, and positive pressure respiration may be desirable if pulmonary edema is present. The narcotic antagonist naloxone will markedly reduce the degree of respiratory depression, and 0.4 to 2 mg should be administered promptly, preferably intravenously. If the desired degree of counteraction with improvement in respiratory functions is not obtained, naloxone should be repeated at 2 to 3 minute intervals. The duration of action of the antagonist may be brief. If no response is observed after 10 mg of naloxone have been administered, the diagnosis of propoxyphene toxicity should be questioned. Naloxone may also be administered by continuous intravenous infusion.
Treatment of Propoxyphene Overdosage in Children
The usual initial dose of naloxone in children is 0.01 mg/kg body weight given intravenously. If this dose does not result in the desired degree of clinical improvement, a subsequent increased dose of 0.1 mg/kg body weight may be administered. If an IV route of administration is not available, naloxone may be administered IM or subcutaneously in divided doses. If necessary, naloxone can be diluted with Sterile Water for injection.
Blood gases, pH, and electrolytes should be monitored in order that acidosis and any electrolyte disturbance present may be corrected promptly. Acidosis, hypoxia, and generalized CNS depression predispose to the development of cardiac arrhythmias. Ventricular fibrillation or cardiac arrest may occur and necessitate the full complement of cardiopulmonary resuscitation (CPR) measures. Respiratory acidosis rapidly subsides as ventilation is restored and hypercapnia eliminated, but lactic acidosis may require intravenous bicarbonate for prompt correction.
Electrocardiographic monitoring is essential. Prompt correction of hypoxia, acidosis, and electrolyte disturbance (when present) will help prevent these cardiac complications and will increase the effectiveness of agents administered to restore normal cardiac function.
In addition to the use of a narcotic antagonist, the patient may require careful titration with an anticonvulsant to control convulsions. Analeptic drugs (for example, caffeine or amphetamine) should not be used because of their tendency to precipitate convulsions.
General supportive measures, in addition to oxygen, include, when necessary, intravenous fluids, vasopressor-inotropic compounds, and when infection is likely, anti-infective agents. Gastric lavage may be useful, and activated charcoal can adsorb a significant amount of ingested propoxyphene. Dialysis is of little value in poisoning due to propoxyphene. Efforts should be made to determine whether other agents, such as alcohol, barbiturates, tranquilizers or other CNS depressants, were also ingested, since these increase CNS depression as well as cause specific toxic effects.
Symptoms of Acetaminophen Overdosage
Shortly after oral ingestion of an overdosage of acetaminophen and for the next 24 hours, anorexia, nausea, vomiting, diaphoresis, general malaise, and abdominal pain have been noted. The patient may then present no symptoms, but evidence of liver dysfunction may become apparent up to 72 hours after ingestion, with elevated serum transaminase and lactic dehydrogenase levels, an increase in serum bilirubin concentrations, and a prolonged prothrombin time. Death from hepatic failure may result 3 to 7 days after overdosage.
Acute renal failure may accompany the hepatic dysfunction and has been noted in patients who do not exhibit signs of fulminant hepatic failure. Typically, renal impairment is more apparent 6 to 9 days after ingestion of the overdose.
Treatment of Acetaminophen Overdosage
Acetaminophen in massive overdosage may cause hepatic toxicity in some patients. In all cases of suspected overdose, immediately call your regional poison center or the Rocky Mountain Poison Center's toll-free number (800-525-6115) for assistance in diagnosis and for directions in the use of N-acetylcysteine as an antidote.
In adults, hepatic toxicity has rarely been reported with acute overdoses of less than 10 g and fatalities with less than 15 g. Importantly, young children seem to be more resistant than adults to the hepatotoxic effect of an acetaminophen overdose. Despite this, the measures outlined below should be initiated in any adult or child suspected of having ingested an acetaminophen overdose.
Because clinical and laboratory evidence of hepatic toxicity may not be apparent until 48 to 72 hours postingestion, liver function studies should be obtained initially and repeated at 24-hour intervals.
Consider emptying the stomach promptly by lavage or by induction of emesis with syrup of ipecac. Patients' estimates of the quantity of a drug ingested are notoriously unreliable. Therefore, if an acetaminophen overdose is suspected, a serum acetaminophen assay should be obtained as early as possible, but no sooner than 4 hours following ingestion. The antidote, N-acetylcysteine, should be administered as early as possible, preferably within 16 hours of the overdose ingestion for optimal results. Following recovery, there are no residual, structural, or functional hepatic abnormalities.
DOSAGE AND ADMINISTRATION:
This products is given orally. The usual dosage is 100 mg propoxyphene napsylate and 500 mg acetaminophen every 4 hours as needed for pain. The maximum recommended dose of propoxyphene napsylate is 600 mg per day.
Consideration should be given to a reduced total daily dosage in patients with hepatic or renal impairment.
HOW SUPPLIED:
Propoxyphene napsylate and acetaminophen tablets contain 50 mg propoxyphene napsylate and 325 mg acetaminophen. They are supplied as orange film coated, unscored, capsule shaped tablets, debossed "5111" and "V" in containers of 10, 100, 500 and 1000 and unit dose packages of 100's.
Propoxyphene napsylate and acetaminophen tablets contain 100 mg propoxyphene napsylate and 650 mg acetaminophen. They are supplied as orange film coated, unscored, capsule shaped tablets, debossed "5112" and "V", in white film coated, unscored capsule shaped tablets, debossed "5113" and "V" and in pink film coated, unscored capsule shaped tablets, debossed "5114" and "V" in containers of 20, 30, 50, 60, 90, 100, 120, 180, 270, 500 and 1000.
Dispense in a tight, light-resistant container as defined in the USP/NF with a child-resistant closure.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
ANIMAL TOXICOLOGY
The acute lethal doses of the hydrochloride and napsylate salts of propoxyphene were determined in 4 species. The results shown in Figure 2 indicate that, on a molar basis, the napsylate salt is less toxic than the hydrochloride. This may be due to the relative insolubility and retarded absorption of propoxyphene napsylate.
| ACUTE ORAL TOXICITY OF PROPOXYPHENE | ||
| LD50(mg/kg)± SE
LD50 (mmol/kg) |
||
| Species | Propoxyphene Hydrochloride | Propoxyphene Napsylate |
| Mouse | 282 ± 39 | 915 ± 163 |
| 0.75 | 1.62 | |
| Rat | 230 ± 44 | 647 ± 95 |
| 0.61 | 1.14 | |
| Rabbit | ca 82 | >183 |
| 0.22 | >0.32 | |
| Dog | ca 100 | >183 |
| 0.27 | >0.32 | |
Some indication of the relative insolubility and retarded absorption of propoxyphene napsylate was obtained by measuring plasma propoxyphene levels in 2 groups of 4 dogs following oral administration of equimolar doses of the 2 salts.
As shown in Figure 3, the peak plasma concentration observed with propoxyphene hydrochloride was much higher than that obtained after administration of the napsylate salt.
Although none of the animals in this experiment died, 3 of the 4 dogs given propoxyphene hydrochloride exhibited convulsive seizures during the time interval corresponding to the peak plasma levels. The 4 animals receiving the napsylate salt were ataxic but not acutely ill.
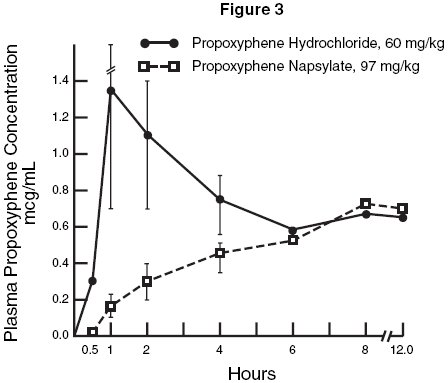
Figure 3. Plasma propoxyphene concentrations in dogs following large doses of the hydrochloride and napsylate salts.
The following information includes the maximum daily dosage and is available to patients receiving propoxyphene napsylate and acetaminophen tablets.
Patient Information Sheet
YOUR PRESCRIPTION FOR A PROPOXYPHENE PRODUCT SUMMARY
Products containing Propoxyphene are used to relieve pain.
LIMIT YOUR INTAKE OF ALCOHOL WHILE TAKING THIS DRUG. Make sure your doctor knows if you are taking tranquilizers, sleep aids, antidepressants, antihistamines, or any other drugs that make you sleepy. Combining propoxyphene with alcohol or these drugs in excessive doses is dangerous.
Use care while driving a car or using machines until you see how the drug affects you because propoxyphene can make you sleepy. Do not take more of the drug than your doctor prescribed. Dependence has occurred when patients have taken propoxyphene for a long period of time at doses greater than recommended.
The rest of this leaflet gives you more information about propoxyphene. Please read it and keep it for future use.
Uses for Propoxyphene
Products containing propoxyphene are used for the relief of mild to moderate pain. Products that contain propoxyphene plus acetaminophen are prescribed for the relief of pain or pain associated with fever.
Before Taking Propoxyphene
Make sure your doctor knows if you have ever had an allergic reaction to propoxyphene or acetaminophen.
The effect of propoxyphene in children under 12 has not been studied; therefore, use of the drug in this age group is not recommended.
How to Take Propoxyphene
Follow your doctor's directions exactly. Do not increase the amount you take without your doctor's approval. If you miss a dose of the drug, do not take twice as much the next time.
Pregnancy
Do not take propoxyphene during pregnancy unless your doctor knows you are pregnant and specifically recommends its use. Cases of temporary dependence in the newborn have occurred when the mother has taken propoxyphene consistently in the weeks before delivery. As a general principle, no drug should be taken during pregnancy unless it is clearly necessary.
General Cautions
Heavy use of alcohol with propoxyphene is hazardous and may lead to overdosage symptoms (see "Overdosage" below). THEREFORE, LIMIT YOUR INTAKE OF ALCOHOL WHILE TAKING PROPOXYPHENE.
Combinations of excessive doses of propoxyphene, alcohol, and tranquilizers are dangerous. Make sure your doctor knows if you are taking tranquilizers, sleep aids, antidepressant drugs, antihistamines, or any other drugs that make you sleepy. The use of these drugs with propoxyphene increases their sedative effects and may lead to overdosage symptoms, including death (see "Overdosage" below). Propoxyphene may cause drowsiness or impair your mental and/or physical abilities; therefore, use caution when driving a vehicle or operating dangerous machinery. DO NOT perform any hazardous task until you have seen your response to this drug.
Propoxyphene may increase the concentration in the body of medications such as anticoagulants ("blood thinners"), antidepressants, or drugs used for epilepsy. The result may be excessive or adverse effects of these medications. Make sure your doctor knows if you are taking any of these medications.
Dependence
You can become dependent on propoxyphene if you take it in higher than recommended doses over a long period of time. Dependence is a feeling of need for the drug and a feeling that you cannot perform normally without it.
Overdosage
An overdose of propoxyphene alone or in combination with other drugs, including alcohol, may cause weakness, difficulty in breathing, confusion, anxiety, and more severe drowsiness and dizziness. Extreme overdosage may lead to unconsciousness and death.
This product contains acetaminophen. Acetaminophen overdosage symptoms include nausea, vomiting, lack of appetite, and abdominal pain. Liver damage may occur even after symptoms disappear. Death can occur days later.
In any suspected overdosage situation, contact your doctor or nearest hospital emergency room. GET EMERGENCY HELP IMMEDIATELY. KEEP THIS DRUG AND ALL DRUGS OUT OF THE REACH OF CHILDREN.
Possible Side Effects
When propoxyphene is taken as directed, side effects are infrequent. Among those reported are drowsiness, dizziness, nausea, and vomiting. If these effects occur, it may help if you lie down and rest.
Less frequently reported side effects are constipation, abdominal pain, skin rashes, lightheadedness, headache, weakness, hallucinations, minor visual disturbances, and feelings of elation or discomfort.
If side effects occur and concern you, contact your doctor.
Other information
The safe and effective use of propoxyphene depends on you taking it exactly as directed. This drug has been prescribed specifically for you and your present condition. Do not give this drug to others who may have similar symptoms. Do not use it for any other reason.
If you would like more information about propoxyphene, ask your doctor or pharmacist. They have a more technical leaflet (professional labeling) you may read.
Manufactured for:
QUALITEST PHARMACEUTICALS
Huntsville, AL 35811
8181438
R11/07-R3
PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL

| PROPOXYPHENE AND ACETAMINOPHEN
propoxyphene napsylate and acetaminophen tablet, film coated |
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA074843 | 02/12/1997 | |
| PROPOXYPHENE AND ACETAMINOPHEN
propoxyphene napsylate and acetaminophen tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA074843 | 02/12/1997 | |
| PROPOXYPHENE AND ACETAMINOPHEN
propoxyphene napsylate and acetaminophen tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA074843 | 02/12/1997 | |
| PROPOXYPHENE AND ACETAMINOPHEN
propoxyphene napsylate and acetaminophen tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA074843 | 02/12/1997 | |
| Labeler - Qualitest Pharmaceuticals (011103059) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Vintage Pharmaceuticals-Charlotte | 151228897 | MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Vintage Pharmaceuticals-Huntsville | 958430845 | MANUFACTURE | |