ISOFLURANE- isoflurane liquid
Hospira, Inc.
----------
DESCRIPTION
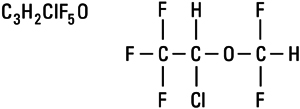
Isoflurane, USP, a nonflammable liquid administered by vaporizing, is a general inhalation anesthetic drug. It is 1-chloro-2,2,2-trifluoroethyl difluoromethyl ether, and its structural formula is:
| Some physical constants are: | ||
|
Molecular weight |
184.5 |
|
|
Boiling point at 760 mm Hg |
48.5°C (uncorr.) |
|
|
Refractive index n20D |
1.2990-1.3005 |
|
|
Specific gravity 25°/25°C |
1.496 |
|
|
Vapor pressure in mm Hg** |
20°C |
238 |
|
25°C |
295 |
|
|
30°C |
367 |
|
|
35°C |
450 |
|
| **Equation for vapor pressure calculation: | ||
|
log10Pvap = A + B/T | where: | A = 8.056 B = -1664.58 T = °C + 273.16 (Kelvin) |
| Partition coefficients at 37°C | |
|
Water/gas |
0.61 |
|
Blood/gas |
1.43 |
|
Oil/gas |
90.8 |
| Partition coefficients at 25°C - rubber and plastic | |
| Conductive rubber/gas | 62.0 |
| Butyl rubber/gas | 75.0 |
| Polyvinyl chloride/gas | 110.0 |
| Polyethylene/gas | ~2.0 |
| Polyurethane/gas | ~1.4 |
| Polyolefin/gas | ~1.1 |
| Butyl acetate/gas | ~2.5 |
| Purity by gas chromatography | >99.9% |
|
Lower limit of flammability in oxygen or nitrous oxide at 9 joules/sec. and 23°C |
None |
|
Lower limit of flammability in oxygen or nitrous oxide at 900 joules/sec. and 23°C |
Greater than useful concentration in anesthesia. |
Isoflurane is a clear, colorless, stable liquid containing no additives or chemical stabilizers. Isoflurane has a mildly pungent, musty, ethereal odor. Samples stored in stability chambers in amber glass for five years, as well as samples directly exposed for 7 days to accelerated light conditions were unchanged in composition as determined by gas chromatography. Isoflurane in one normal sodium methoxide-methanol solution, a strong base, for over six months consumed essentially no alkali, indicative of strong base stability. Isoflurane does not decompose in the presence of soda lime (at normal operating temperatures), and does not attack aluminum, tin, brass, iron or copper.
CLINICAL PHARMACOLOGY
Isoflurane, USP is an inhalation anesthetic. The MAC (minimum alveolar concentration) in man is as follows:
| Age | 100% Oxygen | 70% N2O |
|
26 ± 4 |
1.28 |
0.56 |
|
44 ± 7 |
1.15 |
0.50 |
|
64 ± 5 |
1.05 |
0.37 |
Induction of and recovery from isoflurane anesthesia are rapid. Isoflurane has a mild pungency which limits the rate of induction, although excessive salivation or tracheobronchial secretions do not appear to be stimulated. Pharyngeal and laryngeal reflexes are readily obtunded. The level of anesthesia may be changed rapidly with isoflurane. Isoflurane is a profound respiratory depressant. RESPIRATION MUST BE MONITORED CLOSELY AND SUPPORTED WHEN NECESSARY. As anesthetic dose is increased, tidal volume decreases and respiratory rate is unchanged. This depression is partially reversed by surgical stimulation, even at deeper levels of anesthesia. Isoflurane evokes a sigh response reminiscent of that seen with diethyl ether and enflurane, although the frequency is less than with enflurane.
Blood pressure decreases with induction of anesthesia but returns toward normal with surgical stimulation. Progressive increases in depth of anesthesia produce corresponding decreases in blood pressure. Nitrous oxide diminishes the inspiratory concentration of isoflurane required to reach a desired level of anesthesia and may reduce the arterial hypotension seen with isoflurane alone. Heart rhythm is remarkably stable. With controlled ventilation and normal PaCO2, cardiac output is maintained despite increasing depth of anesthesia primarily through an increase in heart rate which compensates for a reduction in stroke volume. The hypercapnia which attends spontaneous ventilation during isoflurane anesthesia further increases heart rate and raises cardiac output above awake levels. Isoflurane does not sensitize the myocardium to exogenously administered epinephrine in the dog. Limited data indicate that subcutaneous injection of 0.25 mg of epinephrine (50 mL of 1:200,000 solution) does not produce an increase in ventricular arrhythmias in patients anesthetized with isoflurane.
Muscle relaxation is often adequate for intra-abdominal operations at normal levels of anesthesia. Complete muscle paralysis can be attained with small doses of muscle relaxants. ALL COMMONLY USED MUSCLE RELAXANTS ARE MARKEDLY POTENTIATED WITH ISOFLURANE, THE EFFECT BEING MOST PROFOUND WITH THE NONDEPOLARIZING TYPE. Neostigmine reverses the effect of nondepolarizing muscle relaxants in the presence of isoflurane. All commonly used muscle relaxants are compatible with isoflurane.
Isoflurane can produce coronary vasodilation at the arteriolar level in selected animal models1,2; the drug is probably also a coronary dilator in humans. Isoflurane, like some other coronary arteriolar dilators, has been shown to divert blood from collateral dependent myocardium to normally perfused areas in an animal model (“coronary steal”)3. Clinical studies to date evaluating myocardial ischemia, infarction and death as outcome parameters have not established that the coronary arteriolar dilation property of isoflurane is associated with coronary steal or myocardial ischemia in patients with coronary artery disease4,5,6,7.
Pharmacokinetics: Isoflurane undergoes minimal biotransformation in man. In the postanesthesia period, only 0.17% of the isoflurane taken up can be recovered as urinary metabolites.
INDICATIONS AND USAGE
Isoflurane, USP may be used for induction and maintenance of general anesthesia. Adequate data have not been developed to establish its application in obstetrical anesthesia.
CONTRAINDICATIONS
Known sensitivity to Isoflurane, USP or to other halogenated agents.
Known or suspected genetic susceptibility to malignant hyperthermia.
WARNINGS
Perioperative Hyperkalemia: Use of inhaled anesthetic agents has been associated with rare increases in serum potassium levels that have resulted in cardiac arrhythmias and death in pediatric patients during the postoperative period. Patients with latent as well as overt neuromuscular disease, particularly Duchenne muscular dystrophy, appear to be most vulnerable. Concomitant used of Succinylcholine has been associated with most, but not all, of these cases. These patients also experienced significant elevations in serum creatinine kinase levels and, in some cases, changes in urine consistent with myoglobinuria. Despite the similarity in presentation to malignant hyperthermia, none of these patients exhibited signs or symptoms of muscle rigidity or hypermetabolic state. Early and aggressive intervention to treat hyperkalemia and resistant arrhythmias is recommended, as is subsequent evaluation for latent neuromuscular disease.
Malignant Hyperthermia: In susceptible individuals, isoflurane anesthesia may trigger a skeletal muscle hypermetabolic state leading to high oxygen demand and the clinical syndrome known as malignant hyperthermia. The syndrome includes nonspecific features such as muscle rigidity, tachycardia, tachypnea, cyanosis, arrhythmias, and unstable blood pressure. (It should also be noted that many of these nonspecific signs may appear with light anesthesia, acute hypoxia, etc.) An increase in overall metabolism may be reflected in an elevated temperature, (which may rise rapidly early or late in the case, but usually is not the first sign of augmented metabolism) and an increased usage of the CO2 absorption system (hot canister). PaO2 and pH may decrease, and hyperkalemia and a base deficit may appear. Treatment includes discontinuance of triggering agents (e.g., isoflurane), administration of intravenous dantrolene sodium, and application of supportive therapy. Such therapy includes vigorous efforts to restore body temperature to normal, respiratory and circulatory support as indicated, and management of electrolyte-fluid-acid-base derangements. (Consult prescribing information for dantrolene sodium intravenous for additional information on patient management). Renal failure may appear later, and urine flow should be sustained if possible.
Since levels of anesthesia may be altered easily and rapidly, only vaporizers producing predictable concentrations should be used. Hypotension and respiratory depression increase as anesthesia is deepened.
Increased blood loss comparable to that seen with halothane has been observed in patients undergoing abortions.
Isoflurane, USP markedly increases cerebral blood flow at deeper levels of anesthesia. There may be a transient rise in cerebral spinal fluid pressure which is fully reversible with hyperventilation.
PRECAUTIONS
General: As with any potent general anesthetic, Isoflurane, USP should only be administered in an adequately equipped anesthetizing environment by those who are familiar with the pharmacology of the drug and qualified by training and experience to manage the anesthetized patient.
Regardless of the anesthetics employed, maintenance of normal hemodynamics is important to the avoidance of myocardial ischemia in patients with coronary artery disease4,5,6,7.
Isoflurane, like some other inhalational anesthetics, can react with desiccated carbon dioxide (CO2) absorbents to produce carbon monoxide which may result in elevated levels of carboxyhemoglobin in some patients. Case reports suggest that barium hydroxide lime and soda lime become desiccated when fresh gases are passed through the CO2 absorber canister at high flow rates over many hours or days. When a clinician suspects that CO2 absorbent may be desiccated, it should be replaced before the administration of isoflurane.
As with other halogenated anesthetic agents, isoflurane may cause sensitivity hepatitis in patients who have been sensitized by previous exposure to halogenated anesthetics (see CONTRAINDICATIONS).
Information for Patients: Isoflurane, as well as other general anesthetics, may cause a slight decrease in intellectual function for 2 or 3 days following anesthesia. As with other anesthetics, small changes in moods and symptoms may persist for up to 6 days after administration.
Laboratory Tests: Transient increases in BSP retention, blood glucose and serum creatinine with decrease in BUN, serum cholesterol and alkaline phosphatase have been observed.
Drug Interactions: Isoflurane potentiates the muscle relaxant effect of all muscle relaxants, most notably nondepolarizing muscle relaxants, and MAC (minimum alveolar concentration) is reduced by concomitant administration of N2O. See CLINICAL PHARMACOLOGY.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Swiss ICR mice were given isoflurane to determine whether such exposure might induce neoplasia. Isoflurane was given at 1/2, 1/8 and 1/32 MAC for four in-utero exposures and for 24 exposures to the pups during the first nine weeks of life. The mice were killed at 15 months of age. The incidence of tumors in these mice was the same as in untreated control mice, which were given the same background gases, but not the anesthetic.
Pregnancy Category C: Isoflurane has been shown to have a possible anesthetic-related fetotoxic effect in mice when given in doses 6 times the human dose. There are no adequate and well-controlled studies in pregnant women. Isoflurane should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers: It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when isoflurane is administered to a nursing woman.
ADVERSE REACTIONS
Adverse reactions encountered in the administration of Isoflurane, USP are in general dose dependent extensions of pharmacophysiologic effects and include respiratory depression, hypotension and arrhythmias.
Shivering, nausea, vomiting and ileus have been observed in the postoperative period.
As with all other general anesthetics, transient elevations in white blood count have been observed even in the absence of surgical stress. See WARNINGS for information regarding malignant hyperthermia and elevated carboxyhemoglobin levels.
During marketing, there have been rare reports of mild, moderate and severe (some fatal) post-operative hepatic dysfunction and hepatitis.
Isoflurane, USP has also been associated with perioperative hyperkalemia (see WARNINGS).
There have been rare post-marketing reports of hepatic failure and hepatic necrosis associated with the use of potent volatile anesthetic agents, including Isoflurane, USP. Due to the spontaneous nature of these reports, the actual incidence and relationship of Isoflurane, USP to these events cannot be established with certainty.
OVERDOSAGE
In the event of overdosage, or what may appear to be overdosage, the following action should be taken:
Stop drug administration, establish a clear airway and initiate assisted or controlled ventilation with pure oxygen.
DOSAGE AND ADMINISTRATION
Premedication: Premedication should be selected according to the need of the individual patient, taking into account that secretions are weakly stimulated by Isoflurane, USP and the heart rate tends to be increased. The use of anticholinergic drugs is a matter of choice.
Inspired Concentration: The concentration of isoflurane being delivered from a vaporizer during anesthesia should be known. This may be accomplished by using:
a) vaporizers calibrated specifically for isoflurane;
b) vaporizers from which delivered flows can be calculated, such as vaporizers delivering a saturated vapor which is then diluted. The delivered concentration from such a vaporizer may be calculated using the formula:
|
% isoflurane = |
100 PV FV FT (PA — PV) |
| where: |
PA = Pressure of atmosphere |
Isoflurane contains no stabilizer. Nothing in the agent alters calibration or operation of these vaporizers.
Induction: Induction with isoflurane in oxygen or in combination with oxygen-nitrous oxide mixtures may produce coughing, breath holding, or laryngospasm. These difficulties may be avoided by the use of a hypnotic dose of an ultra-short-acting barbiturate. Inspired concentrations of 1.5 to 3% isoflurane usually produce surgical anesthesia in 7 to 10 minutes.
Maintenance: Surgical levels of anesthesia may be sustained with a 1 to 2.5% concentration when nitrous oxide is used concomitantly. An additional 0.5 to 1% may be required when isoflurane is given using oxygen alone. If added relaxation is required, supplemental doses of muscle relaxant may be used.
The level of blood pressure during maintenance is an inverse function of isoflurane concentration in the absence of other complicating problems. Excessive decreases may be due to depth of anesthesia and in such instances may be corrected by lightening anesthesia.
HOW SUPPLIED
Isoflurane, USP, is supplied in 100 mL and 250 mL amber-colored bottles (NDC 0409-3292-01 and NDC 0409-3292-02).
Safety and Handling: Occupational Caution: There is no specific work exposure limit established for Isoflurane, USP. However, the National Institute for Occupational Safety and Health Administration (NIOSH) recommends that no worker should be exposed at ceiling concentrations greater than 2 ppm of any halogenated anesthetic agent over a sampling period not to exceed one hour.
The predicted effects of acute overexposure by inhalation of Isoflurane, USP include headache, dizziness or (in extreme cases) unconsciousness. There are no documented adverse effects of chronic exposure to halogenated anesthetic vapors (Waste Anesthetic Gases or WAGs) in the workplace. Although results of some epidemiological studies suggest a link between exposure to halogenated anesthetics and increased health problems (particularly spontaneous abortion), the relationship is not conclusive. Since exposure to WAGs is one possible factor in the findings for these studies, operating room personnel, and pregnant women in particular, should minimize exposure. Precautions include adequate general ventilation in the operating room, the use of well-designed and well-maintained scavenging system, work practices to minimize leaks and spills while the anesthetic agent is in use, and routine equipment maintenance to minimize leaks.
Storage: Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.] Isoflurane contains no additives and has been demonstrated to be stable at room temperature for periods in excess of five years.
References
-
JC Sill, et al, Anesthesiology 66:273-279, 1987
-
RF Hickey, et al, Anesthesiology 68:21-30, 1988
-
CW Buffington, et al, Anesthesiology 66:280-292, 1987
-
S Reiz, et al, Anesthesiology 59:91-97, 1983
-
S Slogoff and AS Keats, Anesthesiology 70:179-188, 1989
-
KJ Tuman, et al, Anesthesiology 70:189-198, 1989
-
DT Mangano, Editorial Views, Anesthesiology 70:175-178, 1989
Product of United Kingdom
Product inquiries should be directed to
Hospira, Inc., Lake Forest, IL 60045 USA
Revised: July, 2007
Printed in USA EN-1566
Hospira, Inc., Lake Forest, IL 60045 USA
| ISOFLURANE
isoflurane liquid |
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |