FEMARA
-
letrozole tablet, film coated
Novartis Pharmaceuticals Corporation
----------
Femara®
(letrozole tablets)
2.5 mg Tablets
Rx only
Prescribing Information
DESCRIPTION
Femara® (letrozole tablets) for oral administration contains 2.5 mg of letrozole, a nonsteroidal aromatase inhibitor (inhibitor of estrogen synthesis). It is chemically described as 4,4'-(1H-1,2,4-Triazol-1-ylmethylene)dibenzonitrile, and its structural formula is
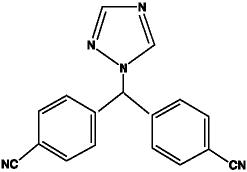
Letrozole is a white to yellowish crystalline powder, practically odorless, freely soluble in dichloromethane, slightly soluble in ethanol, and practically insoluble in water. It has a molecular weight of 285.31, empirical formula C17H11N5, and a melting range of 184°C-185°C.
Femara® (letrozole tablets) is available as 2.5 mg tablets for oral administration.
Inactive Ingredients. Colloidal silicon dioxide, ferric oxide, hydroxypropyl methylcellulose, lactose monohydrate, magnesium stearate, maize starch, microcrystalline cellulose, polyethylene glycol, sodium starch glycolate, talc, and titanium dioxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
The growth of some cancers of the breast is stimulated or maintained by estrogens. Treatment of breast cancer thought to be hormonally responsive (i.e., estrogen and/or progesterone receptor positive or receptor unknown) has included a variety of efforts to decrease estrogen levels (ovariectomy, adrenalectomy, hypophysectomy) or inhibit estrogen effects (antiestrogens and progestational agents). These interventions lead to decreased tumor mass or delayed progression of tumor growth in some women.
In postmenopausal women, estrogens are mainly derived from the action of the aromatase enzyme, which converts adrenal androgens (primarily androstenedione and testosterone) to estrone and estradiol. The suppression of estrogen biosynthesis in peripheral tissues and in the cancer tissue itself can therefore be achieved by specifically inhibiting the aromatase enzyme.
Letrozole is a nonsteroidal competitive inhibitor of the aromatase enzyme system; it inhibits the conversion of androgens to estrogens. In adult nontumor- and tumor-bearing female animals, letrozole is as effective as ovariectomy in reducing uterine weight, elevating serum LH, and causing the regression of estrogen-dependent tumors. In contrast to ovariectomy, treatment with letrozole does not lead to an increase in serum FSH. Letrozole selectively inhibits gonadal steroidogenesis but has no significant effect on adrenal mineralocorticoid or glucocorticoid synthesis.
Letrozole inhibits the aromatase enzyme by competitively binding to the heme of the cytochrome P450 subunit of the enzyme, resulting in a reduction of estrogen biosynthesis in all tissues. Treatment of women with letrozole significantly lowers serum estrone, estradiol and estrone sulfate and has not been shown to significantly affect adrenal corticosteroid synthesis, aldosterone synthesis, or synthesis of thyroid hormones.
Pharmacokinetics
Letrozole is rapidly and completely absorbed from the gastrointestinal tract and absorption is not affected by food. It is metabolized slowly to an inactive metabolite whose glucuronide conjugate is excreted renally, representing the major clearance pathway. About 90% of radiolabeled letrozole is recovered in urine. Letrozole’s terminal elimination half-life is about 2 days and steady-state plasma concentration after daily 2.5 mg dosing is reached in 2-6 weeks. Plasma concentrations at steady state are 1.5 to 2 times higher than predicted from the concentrations measured after a single dose, indicating a slight non-linearity in the pharmacokinetics of letrozole upon daily administration of 2.5 mg. These steady-state levels are maintained over extended periods, however, and continuous accumulation of letrozole does not occur. Letrozole is weakly protein bound and has a large volume of distribution (approximately 1.9 L/kg).
Metabolism and Excretion
Metabolism to a pharmacologically-inactive carbinol metabolite (4,4'-methanol-bisbenzonitrile) and renal excretion of the glucuronide conjugate of this metabolite is the major pathway of letrozole clearance. Of the radiolabel recovered in urine, at least 75% was the glucuronide of the carbinol metabolite, about 9% was two unidentified metabolites, and 6% was unchanged letrozole.
In human microsomes with specific CYP isozyme activity, CYP3A4 metabolized letrozole to the carbinol metabolite while CYP2A6 formed both this metabolite and its ketone analog. In human liver microsomes, letrozole strongly inhibited CYP2A6 and moderately inhibited CYP2C19.
Special Populations
Pediatric, Geriatric and Race
In the study populations (adults ranging in age from 35 to >80 years), no change in pharmacokinetic parameters was observed with increasing age. Differences in letrozole pharmacokinetics between adult and pediatric populations have not been studied. Differences in letrozole pharmacokinetics due to race have not been studied.
Renal Insufficiency
In a study of volunteers with varying renal function (24-hour creatinine clearance: 9-116 mL/min), no effect of renal function on the pharmacokinetics of single doses of 2.5 mg of Femara® (letrozole tablets) was found. In addition, in a study of 347 patients with advanced breast cancer, about half of whom received 2.5 mg Femara and half 0.5 mg Femara, renal impairment (calculated creatinine clearance: 20-50 mL/min) did not affect steady-state plasma letrozole concentration.
Hepatic Insufficiency
In a study of subjects with mild to moderate non-metastatic hepatic dysfunction (e.g., cirrhosis, Child-Pugh classification A and B), the mean AUC values of the volunteers with moderate hepatic impairment were 37% higher than in normal subjects, but still within the range seen in subjects without impaired function. In a pharmacokinetics study, subjects with liver cirrhosis and severe hepatic impairment (Child-Pugh classification C, which included bilirubins about 2-11 times ULN with minimal to severe ascites) had two-fold increase in exposure (AUC) and 47% reduction in systemic clearance. Breast cancer patients with severe hepatic impairment are thus expected to be exposed to higher levels of letrozole than patients with normal liver function receiving similar doses of this drug. (See DOSAGE AND ADMINISTRATION, Hepatic Impairment.)
Drug/Drug Interactions
A pharmacokinetic interaction study with cimetidine showed no clinically significant effect on letrozole pharmacokinetics. An interaction study with warfarin showed no clinically significant effect of letrozole on warfarin pharmacokinetics. In in-vitro experiments, letrozole showed no significant inhibition in the metabolism of diazepam. Similarly, no significant inhibition of letrozole metabolism by diazepam was observed.
Co-administration of Femara and tamoxifen 20 mg daily resulted in a reduction of letrozole plasma levels of 38% on average. Clinical experience in the second-line breast cancer pivotal trials indicates that the therapeutic effect of Femara therapy is not impaired if Femara is administered immediately after tamoxifen.
There is no clinical experience to date on the use of Femara in combination with other anticancer agents.
Pharmacodynamics
In postmenopausal patients with advanced breast cancer, daily doses of 0.1 mg to 5 mg Femara suppress plasma concentrations of estradiol, estrone, and estrone sulfate by 75%-95% from baseline with maximal suppression achieved within two-three days. Suppression is dose-related, with doses of 0.5 mg and higher giving many values of estrone and estrone sulfate that were below the limit of detection in the assays. Estrogen suppression was maintained throughout treatment in all patients treated at 0.5 mg or higher.
Letrozole is highly specific in inhibiting aromatase activity. There is no impairment of adrenal steroidogenesis. No clinically-relevant changes were found in the plasma concentrations of cortisol, aldosterone, 11-deoxycortisol, 17-hydroxy-progesterone, ACTH or in plasma renin activity among postmenopausal patients treated with a daily dose of Femara 0.1 mg to 5 mg. The ACTH stimulation test performed after 6 and 12 weeks of treatment with daily doses of 0.1, 0.25, 0.5, 1, 2.5, and 5 mg did not indicate any attenuation of aldosterone or cortisol production. Glucocorticoid or mineralocorticoid supplementation is, therefore, not necessary.
No changes were noted in plasma concentrations of androgens (androstenedione and testosterone) among healthy postmenopausal women after 0.1, 0.5, and 2.5 mg single doses of Femara or in plasma concentrations of androstenedione among postmenopausal patients treated with daily doses of 0.1 mg to 5 mg. This indicates that the blockade of estrogen biosynthesis does not lead to accumulation of androgenic precursors. Plasma levels of LH and FSH were not affected by letrozole in patients, nor was thyroid function as evaluated by TSH levels, T3 uptake, and T4 levels.
CLINICAL STUDIES
Adjuvant Treatment of Early Breast Cancer in Postmenopausal Women
A multicenter, double-blind study randomized over 8,000 postmenopausal women with resected, receptor-positive early breast cancer to one of the following arms:
- tamoxifen for 5 years
- Femara for 5 years
- tamoxifen for 2 years followed by Femara for 3 years
- Femara for 2 years followed by tamoxifen for 3 years
Median treatment duration was 24 months, and median follow-up duration was 26 months, 76% of the patients have been followed for more than 2 years, and 16% of patients for 5 years or longer.
Data in Table 2 reflect results from non-switching arms (arms A and B) together with data truncated 30 days after the switch in the two switching arms (arms C and D). The analysis of monotherapy vs sequencing of endocrine treatments will be conducted when the necessary number of events has been achieved. Selected baseline characteristics for the study population are shown in Table 1.
Table 1: Selected Study Population Demographics for Adjuvant Study (ITT Population)
| Baseline Status | Femara® | tamoxifen |
| N=4003 | N=4007 | |
| Age (median, years) | 61 | 61 |
| Age Range (years) | 38-89 | 39-90 |
| Hormone Receptor Status (%) | ||
| ER+ and/or PgR+ | 99.7 | 99.7 |
| Both Unknown | 0.3 | 0.3 |
| Nodal Status (%) | ||
| Node Negative | 52 | 52 |
| Node Positive | 41 | 41 |
| Nodal Status Unknown | 7 | 7 |
| Prior Adjuvant Chemotherapy (%) | 25 | 25 |
Table 2: Adjuvant Study Results
| Femara® | tamoxifen | Hazard Ratio | P-Value | |
| N=4003 | N=4007 | (95 % CI) | ||
| Disease-Free Survival1 | 296 | 369 | 0.79 (0.68, 0.92) | 0.002 |
| Node Positive | 0.71 (0.59, 0.86) | 0.0005 | ||
| Node Negative | 0.92 (0.70, 1.22) | 0.572 | ||
| Prior Adjuvant Chemotherapy | 0.70 (0.53, 0.93) | 0.013 | ||
| No Chemotherapy | 0.83 (0.69, 1.00) | 0.046 | ||
| Systemic Disease-Free Survival2 | 268 | 321 | 0.83 (0.70, 0.97) | 0.022 |
| Time to Distant Metastasis3 | 184 | 249 | 0.73 (0.60, 0.88) | 0.001 |
| Node Positive | 0.67 (0.54, 0.84) | 0.0005 | ||
| Node Negative | 0.90 (0.60, 1.34) | 0.597 | ||
| Prior Adjuvant Chemotherapy | 0.69 (0.50, 0.95) | 0.024 | ||
| No Chemotherapy | 0.75 (0.60, 0.95) | 0.018 | ||
| Contralateral Breast Cancer | 19 | 31 | 0.61 (0.35, 1.08) | 0.091 |
| Overall Survival | 166 | 192 | 0.86 (0.70, 1.06) | 0.155 |
| Node Positive | 0.81 (0.63, 1.05) | 0.113 | ||
| Node Negative | 0.88 (0.59, 1.30) | 0.507 | ||
| Prior Adjuvant Chemotherapy | 0.76 (0.51, 1.14) | 0.185 | ||
| No Chemotherapy | 0.90 (0.71, 1.15) | 0.395 | ||
| *Definition of 1 Disease-Free Survival: Time from randomization to the earliest occurrence of invasive loco-regional recurrence, distant metastases, invasive contralateral breast cancer, or death from any cause. 2Systemic Disease-Free Survival: Time from randomization to invasive regional recurrence, distant metastases, or death from any cause. 3Time to Distant Metastasis: Time from randomization to distant metastases. |
||||
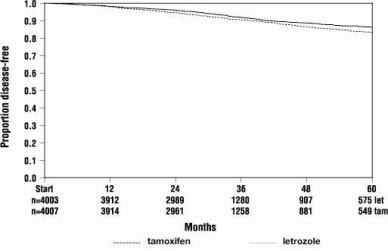
Figure 1 shows the Kaplan-Meier curves for Disease-Free Survival.
Figure 1 Disease-Free Survival (ITT Population)
Extended Adjuvant Treatment of Early Breast Cancer in Postmenopausal Women After Completion of 5 Years of Adjuvant Tamoxifen Therapy
A double-blind, randomized, placebo-controlled trial of Femara® (letrozole tablets) was performed in over 5,100 postmenopausal women with receptor-positive or unknown primary breast cancer who were disease free after 5 years of adjuvant treatment with tamoxifen. Patients had to be within 3 months of completing the 5 years of tamoxifen.
The planned duration of treatment for patients in the study was 5 years, but the trial was terminated early because of an interim analysis showing a favorable Femara effect on time without recurrence or contralateral breast cancer. At the time of unblinding, women had been followed for a median of 28 months, 30% of patients had completed 3 or more years of follow-up and less than 1% of patients had completed 5 years of follow-up.
Selected baseline characteristics for the study population are shown in Table 3.
Table 3: Selected Study Population Demographics (Modified ITT Population)
| Baseline Status | Femara® | Placebo |
| N=2582 | N=2586 | |
| Hormone Receptor Status (%) | ||
| ER+ and/or PgR+ | 98 | 98 |
| Both Unknown | 2 | 2 |
| Nodal Status (%) | ||
| Node Negative | 50 | 50 |
| Node Positive | 46 | 46 |
| Nodal Status Unknown | 4 | 4 |
| Chemotherapy | 46 | 46 |
Table 4 shows the study results. Disease-free survival was measured as the time from randomization to the earliest event of loco-regional or distant recurrence of the primary disease or development of contralateral breast cancer or death. Data were premature for an analysis of survival.
Table 4: Extended Adjuvant Study Results
| Femara®
N = 2582 | Placebo
N = 2586 | Hazard Ratio
(95% CI) | P-Value | |
| Disease Free Survival (DFS) | 122 (4.7%) | 193 (7.5%) | 0.62 (0.49, 0.78)1 | 0.00003 |
| (First event of loco-regional recurrence, distant relapse, contralateral breast cancer or death from any cause) | ||||
| Local Breast Recurrence | 9 | 22 | ||
| Local Chest Wall Recurrence | 2 | 8 | ||
| Regional Recurrence | 7 | 4 | ||
| Distant Recurrence | 55 | 92 | 0.61 (0.44 - 0.84) | 0.003 |
| Contralateral Breast Cancer | 19 | 29 | ||
| Deaths Without Recurrence or Contralateral Breast Cancer | 30 | 38 | ||
| DFS by Stratification | ||||
| Receptor Status | ||||
| - Positive | 117/2527(4.6%) | 190/2530(7.5%) | 0.60(0.48,0.76) | |
| - Unknown | 5/55(9.1%) | 3/56(5.4%) | 1.78(0.43,7.5) | |
| Nodal Status | ||||
| - Positive | 77/1184(6.5%) | 123/1187(10.4%) | 0.61(0.46,0.81) | |
| - Negative | 39/1298(3.0%) | 63/1301(4.8%) | 0.61(0.41,0.91) | |
| - Unknown | 6/100(6.0%) | 7/98(7.1%) | 0.81(0.27,2.4) | |
| Adjuvant Chemotherapy | ||||
| - Yes | 58/1197(4.8%) | 88/1199(7.3%) | 0.64(0.46,0.90) | |
| - No | 64/1385(4.6%) | 105/1387(7.6%) | 0.60(0.44,0.81) | |
| CI = confidence interval for hazard ratio. Hazard ratio of less than 1.0 indicates difference in favor of Femara (lesser risk of recurrence); hazard ratio greater than 1.0 indicates difference in favor of placebo (higher risk of recurrence with Femara). 1 Analysis stratified by receptor status, nodal status and prior adjuvant chemotherapy (stratification factors as at randomization). P-value based on stratified logrank test. |
||||
First-Line Breast Cancer
A randomized, double-blind, multinational trial compared Femara 2.5 mg with tamoxifen 20 mg in 916 postmenopausal patients with locally advanced (Stage IIIB or loco-regional recurrence not amenable to treatment with surgery or radiation) or metastatic breast cancer. Time to progression (TTP) was the primary endpoint of the trial. Selected baseline characteristics for this study are shown in Table 5.
Table 5: Selected Study Population Demographics
| Baseline Status | Femara® | tamoxifen |
| N=458 | N=458 | |
| Stage of Disease | ||
| IIIB | 6% | 7% |
| IV | 93% | 92% |
| Receptor Status | ||
| ER and PgR Positive | 38% | 41% |
| ER or PgR Positive | 26% | 26% |
| Both Unknown | 34% | 33% |
| ER- or PgR- / Other Unknown | <1% | 0 |
| Previous Antiestrogen Therapy | ||
| Adjuvant | 19% | 18% |
| None | 81% | 82% |
| Dominant Site of Disease | ||
| Soft Tissue | 25% | 25% |
| Bone | 32% | 29% |
| Viscera | 43% | 46% |
Femara was superior to tamoxifen in TTP and rate of objective tumor response (see Table 6).
Table 6 summarizes the results of the trial, with a total median follow-up of approximately 32 months. (All analyses are unadjusted and use 2-sided P-values.)
Table 6: Results
| Femara® | tamoxifen | Hazard or Odds | |
| 2.5 mg | 20 mg | Ratio (95% CI) | |
| N=453 | N=454 | P-Value (2-Sided) | |
| Median Time to Progression | 9.4 months | 6.0 months | 0.72 (0.62, 0.83)1 |
| P<0.0001 | |||
| Objective Response Rate | |||
| (CR + PR) | 145 (32%) | 95 (21%) | 1.77 (1.31, 2.39)2 |
| P=0.0002 | |||
| (CR) | 42 (9%) | 15 (3%) | 2.99 (1.63, 5.47)2 |
| P=0.0004 | |||
| Duration of Objective Response | |||
| Median | 18 months | 16 months | |
| (N=145) | (N=95) | ||
| Overall Survival | 35 months | 32 months | |
| (N=458) | (N=458) | P=0.51363 | |
| 1 Hazard ratio 2 Odds ratio 3 Overall logrank test |
|||
Figure 2 shows the Kaplan-Meier curves for TTP.
Figure 2 Kaplan-Meier Estimates of Time to Progression (Tamoxifen Study)
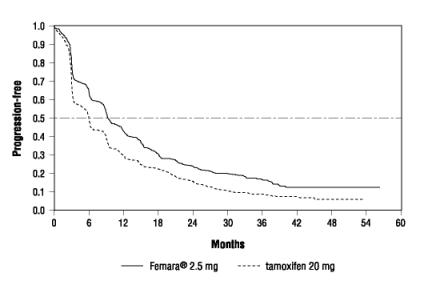
Table 7 shows results in the subgroup of women who had received prior antiestrogen adjuvant therapy, Table 8, results by disease site and Table 9, the results by receptor status.
Table 7: Efficacy in Patients Who Received Prior Antiestrogen Therapy
| Variable | Femara® | tamoxifen |
| 2.5 mg | 20 mg | |
| N=84 | N=83 | |
| Median Time to
Progression (95% CI) | 8.9 months (6.2, 12.5) | 5.9 months (3.2, 6.2) |
| Hazard Ratio for TTP (95% CI) | 0.60 (0.43, 0.84) | |
| Objective Response Rate | ||
| (CR + PR) | 22 (26%) | 7 (8%) |
|
Odds Ratio for Response (95% CI) | 3.85 (1.50, 9.60) | |
| Hazard ratio less than 1 or odds ratio greater than 1 favors Femara; hazard ratio greater than 1 or odds ratio less than 1 favors tamoxifen. | ||
Table 8: Efficacy by Disease Site
| Femara® | tamoxifen | |
| 2.5 mg | 20 mg | |
| Dominant Disease Site | ||
| Soft Tissue: | N=113 | N=115 |
| Median TTP | 12.1 months | 6.4 months |
| Objective Response Rate | 50% | 34% |
| Bone: | N=145 | N=131 |
| Median TTP | 9.5 months | 6.3 months |
| Objective Response Rate | 23% | 15% |
| Viscera: | N=195 | N=208 |
| Median TTP | 8.3 months | 4.6 months |
| Objective Response Rate | 28% | 17% |
Table 9: Efficacy by Receptor Status
| Variable | Femara® | tamoxifen |
| 2.5 mg | 20 mg | |
| Receptor Positive | N=294 | N=305 |
| Median Time to Progression (95% CI) | 9.4 months (8.9, 11.8) | 6.0 months (5.1, 8.5) |
| Hazard Ratio for TTP (95% CI) | 0.69 (0.58, 0.83) | |
| Objective Response Rate (CR+PR) | 97 (33%) | 66 (22%) |
| Odds Ratio for Response (95% CI) | 1.78 (1.20, 2.60) | |
| Receptor Unknown | N=159 | N=149 |
| Median Time to Progression (95% CI) | 9.2 months (6.1, 12.3) | 6.0 months (4.1, 6.4) |
| Hazard Ratio for TTP (95% CI) | 0.77 (0.60, 0.99) | |
| Objective Response Rate (CR+PR) | 48 (30%) | 29 (20%) |
| Odds Ratio for Response (95% CI) | 1.79 (1.10, 3.00) | |
| Hazard ratio less than 1 or odds ratio greater than 1 favors Femara; hazard ratio greater than 1 or odds ratio less than 1 favors tamoxifen. | ||
Figure 3 shows the Kaplan-Meier curves for survival.
Figure 3 Survival by Randomized Treatment Arm
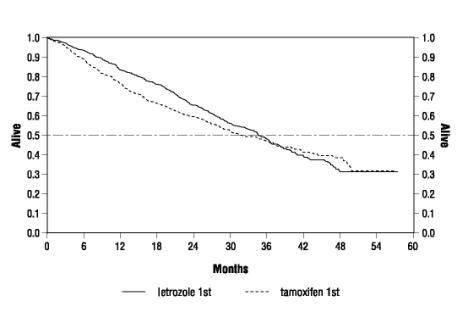
Legend: Randomized Femara: n=458, events 57%, median overall survival 35 months (95% CI 32 to 38 months)
Randomized tamoxifen: n=458, events 57%, median overall survival 32 months (95% CI 28 to 37 months)
Overall logrank P=0.5136 (i.e., there was no significant difference between treatment arms in overall survival).
The median overall survival was 35 months for the Femara group and 32 months for the tamoxifen group, with a P-value 0.5136.
Study design allowed patients to cross over upon progression to the other therapy. Approximately 50% of patients crossed over to the opposite treatment arm and almost all patients who crossed over had done so by 36 months. The median time to crossover was 17 months (Femara to tamoxifen) and 13 months (tamoxifen to Femara). In patients who did not cross over to the opposite treatment arm, median survival was 35 months with Femara (n=219, 95% Cl 29 to 43 months) vs 20 months with tamoxifen (n=229, 95% Cl 16 to 26 months).
Second-Line Breast Cancer
Femara was initially studied at doses of 0.1 mg to 5.0 mg daily in six non-comparative Phase I/II trials in 181 postmenopausal estrogen/progesterone receptor positive or unknown advanced breast cancer patients previously treated with at least antiestrogen therapy. Patients had received other hormonal therapies and also may have received cytotoxic therapy. Eight (20%) of forty patients treated with Femara 2.5 mg daily in Phase I/II trials achieved an objective tumor response (complete or partial response).
Two large randomized, controlled, multinational (predominantly European) trials were conducted in patients with advanced breast cancer who had progressed despite antiestrogen therapy. Patients were randomized to Femara 0.5 mg daily, Femara 2.5 mg daily, or a comparator (megestrol acetate 160 mg daily in one study; and aminoglutethimide 250 mg b.i.d. with corticosteroid supplementation in the other study). In each study over 60% of the patients had received therapeutic antiestrogens, and about one-fifth of these patients had had an objective response. The megestrol acetate controlled study was double-blind; the other study was open label. Selected baseline characteristics for each study are shown in Table 10.
Table 10: Selected Study Population Demographics
| Parameter | megestrol acetate | aminoglutethimide |
| study | study | |
| No. of Participants | 552 | 557 |
| Receptor Status | ||
| ER/PR Positive | 57% | 56% |
| ER/PR Unknown | 43% | 44% |
| Previous Therapy | ||
| Adjuvant Only | 33% | 38% |
| Therapeutic +/- Adj. | 66% | 62% |
| Sites of Disease | ||
| Soft Tissue | 56% | 50% |
| Bone | 50% | 55% |
| Viscera | 40% | 44% |
Confirmed objective tumor response (complete response plus partial response) was the primary endpoint of the trials. Responses were measured according to the Union Internationale Contre le Cancer (UICC) criteria and verified by independent, blinded review. All responses were confirmed by a second evaluation 4-12 weeks after the documentation of the initial response.
Table 11 shows the results for the first trial, with a minimum follow-up of 15 months, that compared Femara 0.5 mg, Femara 2.5 mg, and megestrol acetate 160 mg daily. (All analyses are unadjusted.)
Table 11: Megestrol Acetate Study Results
| Femara® | Femara® | megestrol | |
| 0.5 mg | 2.5 mg | acetate | |
| N=188 | N=174 | N=190 | |
| Objective Response (CR + PR) | 22 (11.7%) | 41 (23.6%) | 31 (16.3%) |
| Median Duration of Response | 552 days | (Not reached) | 561 days |
| Median Time to Progression | 154 days | 170 days | 168 days |
| Median Survival | 633 days | 730 days | 659 days |
| Odds Ratio for Response | Femara 2.5: Femara 0.5=2.33 | Femara 2.5: megestrol=1.58 | |
| (95% CI: 1.32, 4.17); P=0.004* | (95% CI: 0.94, 2.66); P=0.08* | ||
| Relative Risk of Progression | Femara 2.5: Femara 0.5=0.81 | Femara 2.5: megestrol=0.77 | |
| (95% CI: 0.63, 1.03); P=0.09* | (95% CI: 0.60, 0.98), P=0.03* | ||
| * two-sided P-value | |||
The Kaplan-Meier curves for progression for the megestrol acetate study is shown in Figure 4.
Figure 4 Kaplan-Meier Estimates of Time to Progression (Megestrol Acetate Study)
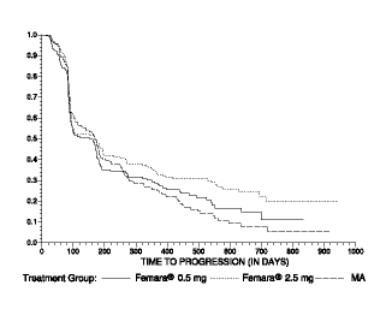
The results for the study comparing Femara to aminoglutethimide, with a minimum follow-up of 9 months, are shown in Table 12. (Unadjusted analyses are used.)
Table 12: Aminoglutethimide Study Results
| Femara® | Femara® | ||
| 0.5 mg | 2.5 mg | aminoglutethimide | |
| N=193 | N=185 | N=179 | |
| Objective Response
(CR + PR) | 34 (17.6%) | 34 (18.4%) | 22 (12.3%) |
| Median Duration of
Response | 619 days | 706 days | 450 days |
| Median Time to
Progression | 103 days | 123 days | 112 days |
| Median Survival | 636 days | 792 days | 592 days |
| Odds Ratio for
Response | Femara 2.5: | Femara 2.5: | |
| Femara 0.5=1.05 | aminoglutethimide=1.61 | ||
| (95% CI: 0.62, 1.79); P=0.85* | (95% CI: 0.90, 2.87); P=0.11* | ||
| Relative Risk of
Progression | Femara 2.5: | Femara 2.5: | |
| Femara 0.5=0.86 | aminoglutethimide=0.74 | ||
| (95% CI: 0.68, 1.11); P=0.25* | (95% CI: 0.57, 0.94); P=0.02* | ||
| *two-sided P-value | |||
The Kaplan-Meier curves for progression for the aminoglutethimide study is shown in Figure 5.
Figure 5 Kaplan-Meier Estimates of Time to Progression (Aminoglutethimide Study)
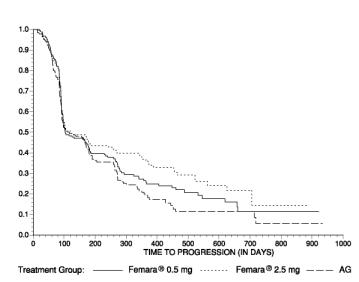
INDICATIONS AND USAGE
Femara® (letrozole tablets) is indicated for the adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer (see CLINICAL STUDIES).
The effectiveness of Femara in early breast cancer is based on an analysis of disease-free survival in patients treated for a median of 24 months and followed for a median of 26 months (see CLINICAL STUDIES). Follow up analyses will determine long-term outcomes for both safety and efficacy.
Femara is indicated for the extended adjuvant treatment of early breast cancer in postmenopausal women who have received 5 years of adjuvant tamoxifen therapy (see CLINICAL STUDIES). The effectiveness of Femara in extended adjuvant treatment of early breast cancer is based on an analysis of disease-free survival in patients treated for a median of 24 months (see CLINICAL STUDIES). Further data will be required to determine long-term outcome.
Femara is indicated for first-line treatment of postmenopausal women with hormone receptor positive or hormone receptor unknown locally advanced or metastatic breast cancer. Femara is also indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following antiestrogen therapy.
CONTRAINDICATIONS
Femara® (letrozole tablets) is contraindicated in patients with known hypersensitivity to Femara or any of its excipients.
Femara is contraindicated in women of premenopausal endocrine status (see WARNINGS, Pregnancy).
WARNINGS
Pregnancy
Femara® (letrozole tablets) may cause fetal harm when administered to pregnant women. Studies in rats at doses equal to or greater than 0.003 mg/kg (about 1/100 the daily maximum recommended human dose on a mg/m2 basis) administered during the period of organogenesis, have shown that letrozole is embryotoxic and fetotoxic, as indicated by intrauterine mortality, increased resorption, increased postimplantation loss, decreased numbers of live fetuses and fetal anomalies including absence and shortening of renal papilla, dilation of ureter, edema and incomplete ossification of frontal skull and metatarsals. Letrozole was teratogenic in rats. A 0.03 mg/kg dose (about 1/10 the daily maximum recommended human dose on a mg/m2 basis) caused fetal domed head and cervical/centrum vertebral fusion.
Letrozole is embryotoxic at doses equal to or greater than 0.002 mg/kg and fetotoxic when administered to rabbits at 0.02 mg/kg (about 1/100,000 and 1/10,000 the daily maximum recommended human dose on a mg/m2 basis, respectively). Fetal anomalies included incomplete ossification of the skull, sternebrae, and fore- and hindlegs.
There are no studies in pregnant women. Femara is indicated for postmenopausal women. If there is exposure to letrozole during pregnancy, the patient should be apprised of the potential hazard to the fetus and potential risk for loss of the pregnancy.
The physician needs to discuss the necessity of adequate contraception with women who have the potential to become pregnant including women who are perimenopausal or who recently became postmenopausal, until their postmenopausal status is fully established.
PRECAUTIONS
Since fatigue and dizziness have been observed with the use of Femara® (letrozole tablets) and somnolence was uncommonly reported, caution is advised when driving or using machinery.
Laboratory Tests
No dose-related effect of Femara on any hematologic or clinical chemistry parameter was evident. Moderate decreases in lymphocyte counts, of uncertain clinical significance, were observed in some patients receiving Femara 2.5 mg. This depression was transient in about half of those affected. Two patients on Femara developed thrombocytopenia; relationship to the study drug was unclear. Patient withdrawal due to laboratory abnormalities, whether related to study treatment or not, was infrequent.
Increases in SGOT, SGPT, and gamma GT ≥5 times the upper limit of normal (ULN) and of bilirubin ≥1.5 times the ULN were most often associated with metastatic disease in the liver. About 3% of study participants receiving Femara had abnormalities in liver chemistries not associated with documented metastases; these abnormalities may have been related to study drug therapy. In the megestrol acetate comparative study about 8% of patients treated with megestrol acetate had abnormalities in liver chemistries that were not associated with documented liver metastases; in the aminoglutethimide study about 10% of aminoglutethimide-treated patients had abnormalities in liver chemistries not associated with hepatic metastases.
In the adjuvant setting, an increase in total cholesterol (generally non-fasting) in patients who had baseline values of total serum cholesterol within the normal range, and then subsequently had an increase in total serum cholesterol of 1.5 ULN was 173/3203 (5.4%) on letrozole vs 40/3224 (1.2%) on tamoxifen. Lipid lowering medications were used by 18% of patients on letrozole and 12% on tamoxifen.
Bone Effects
In the extended adjuvant setting, preliminary results (median duration of follow-up was 20 months) from the bone sub-study (Calcium 500 mg and Vitamin D 400 IU per day mandatory; bisphosphonates not allowed) demonstrated that at 2 years the mean decrease compared to baseline in hip BMD in Femara patients was 3% vs 0.4% for placebo (P=0.048). The mean decrease from baseline BMD results for the lumbar spine at 2 years was Femara 4.6% decrease and placebo 2.2% (P=0.069). Consideration should be given to monitoring BMD.
Drug Interactions
Clinical interaction studies with cimetidine and warfarin indicated that the co-administration of Femara with these drugs does not result in clinically-significant drug interactions. (See CLINICAL PHARMACOLOGY.)
Co-administration of Femara and tamoxifen 20 mg daily resulted in a reduction of letrozole plasma levels by 38% on average. There is no clinical experience to date on the use of Femara in combination with other anticancer agents.
Hepatic Insufficiency
Subjects with cirrhosis and severe hepatic dysfunction (see CLINICAL PHARMACOLOGY, Special Populations) who were dosed with 2.5 mg of Femara experienced approximately twice the exposure to Femara as healthy volunteers with normal liver function. Therefore, a dose reduction is recommended for this patient population. The effect of hepatic impairment on Femara exposure in cancer patients with elevated bilirubin levels has not been determined. (See DOSAGE AND ADMINISTRATION.)
Drug/Laboratory Test-Interactions
None observed.
Carcinogenesis, Mutagenesis, Impairment of Fertility
A conventional carcinogenesis study in mice at doses of 0.6 to 60 mg/kg/day (about 1 to 100 times the daily maximum recommended human dose on a mg/m2 basis) administered by oral gavage for up to 2 years revealed a dose-related increase in the incidence of benign ovarian stromal tumors. The incidence of combined hepatocellular adenoma and carcinoma showed a significant trend in females when the high dose group was excluded due to low survival. In a separate study, plasma AUC0-12hr levels in mice at 60 mg/kg/day were 55 times higher than the AUC0-24hr level in breast cancer patients at the recommended dose. The carcinogenicity study in rats at oral doses of 0.1 to 10 mg/kg/day (about 0.4 to 40 times the daily maximum recommended human dose on a mg/m2 basis) for up to 2 years also produced an increase in the incidence of benign ovarian stromal tumors at 10 mg/kg/day. Ovarian hyperplasia was observed in females at doses equal to or greater than 0.1 mg/kg/day. At 10 mg/kg/day, plasma AUC0-24hr levels in rats were 80 times higher than the level in breast cancer patients at the recommended dose.
Femara was not mutagenic in in vitro tests (Ames and E. coli bacterial tests) but was observed to be a potential clastogen in in vitro assays (CHO K1 and CCL 61 Chinese hamster ovary cells). Letrozole was not clastogenic in vivo (micronucleus test in rats).
Studies to investigate the effect of letrozole on fertility have not been conducted; however, repeated dosing caused sexual inactivity in females and atrophy of the reproductive tract in males and females at doses of 0.6, 0.1 and 0.03 mg/kg in mice, rats and dogs, respectively (about one, 0.4 and 0.4 the daily maximum recommended human dose on a mg/m2 basis, respectively).
Pregnancy
Pregnancy Category D (See WARNINGS.)
Nursing Mothers
It is not known if letrozole is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when letrozole is administered to a nursing woman (see WARNINGS and PRECAUTIONS).
Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
Geriatric Use
The median age of patients in all studies of first-line and second-line treatment of metastatic breast cancer was 64-65 years. About 1/3 of the patients were ≥70 years old. In the first-line study, patients ≥70 years of age experienced longer time to tumor progression and higher response rates than patients <70.
For the extended adjuvant setting, more than 5,100 postmenopausal women were enrolled in the clinical study. In total, 41% of patients were aged 65 years or older at enrollment, while 12% were 75 or older. In the extended adjuvant setting, no overall differences in safety or efficacy were observed between these older patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
In the adjuvant setting, more than 8,000 postmenopausal women were enrolled in the clinical study. In total, 36 % of patients were aged 65 years or older at enrollment, while 12% were 75 or older. More adverse events were generally reported in elderly patients irrespective of study treatment allocation. However, in comparison to tamoxifen, no overall differences with regards to the safety and efficacy profiles were observed between elderly patients and younger patients.
ADVERSE REACTIONS
Femara® (letrozole tablets) was generally well tolerated across all studies in first-line and second-line metastatic breast cancer, adjuvant treatment, as well as extended adjuvant treatment in women who have received prior adjuvant tamoxifen treatment. Generally, the observed adverse reactions are mild or moderate in nature.
Adjuvant Treatment of Early Breast Cancer in Postmenopausal Women
The median duration of adjuvant treatment was 24 months and the median duration of follow-up for safety was 26 months for patients receiving Femara and tamoxifen.
Certain adverse events were prospectively specified for analysis, based on the known pharmacologic properties and side effect profiles of the two drugs.
Adverse events were analyzed irrespective of whether a symptom was present or absent at baseline. Most adverse events reported (82%) were Grade 1 and Grade 2 applying the Common Toxicity Criteria Version 2.0. Table 13 describes adverse events (Grades 1-4) irrespective of relationship to study treatment in the adjuvant BIG 1-98 trial (safety population, during treatment or within 30 days of stopping treatment).
Table 13: Patients with Adverse Events (CTC Grades 1-4, Irrespective of Relationship to Study Drug) in the Adjuvant Study BIG 1-98
| Grades 1-4 | Grades 3-4 | |||||||
|
Adverse Event | Femara®
N=3975 n (%) | tamoxifen
N=3988 n (%) | Femara®
N=3975 n (%) | tamoxifen
N=3988 n (%) |
||||
| Hot Flashes/Flushes | 1338 | (33.7) | 1515 | (38.0) | 0 | - | 0 | - |
| Arthralgia/Arthritis | 840 | (21.1) | 535 | (13.4) | 88 | (2.2) | 49 | (1.2) |
| Night Sweats | 561 | (14.1) | 654 | (16.4) | 0 | - | 0 | - |
| Weight Increase | 425 | (10.7) | 515 | (12.9) | 21 | (0.5) | 44 | (1.1) |
| Nausea | 378 | (9.5) | 416 | (10.4) | 6 | (0.2) | 10 | (0.3) |
| Fatigue (Lethargy, Malaise, Asthenia) | 333 | (8.4) | 345 | (8.7) | 9 | (0.2) | 9 | (0.2) |
| Edema | 286 | (7.2) | 287 | (7.2) | 5 | (0.1) | 2 | (<0.1) |
| Myalgia | 255 | (6.4) | 243 | (6.1) | 26 | (0.7) | 17 | (0.4) |
| Bone Fractures | 223 | (5.6) | 158 | (4.0) | 76 | (1.9) | 45 | (1.1) |
| Vaginal Bleeding | 177 | (4.5) | 411 | (10.3) | 2 | (<0.1) | 7 | (0.2) |
| Headache | 141 | (3.5) | 126 | (3.2) | 12 | (0.3) | 6 | (0.2) |
| Vaginal Irritation | 139 | (3.5) | 122 | (3.1) | 6 | (0.2) | 3 | (<0.1) |
| Vomiting | 109 | (2.7) | 106 | (2.7) | 6 | (0.2) | 8 | (0.2) |
| Dizziness/Light-Headedness | 96 | (2.4) | 110 | (2.8) | 1 | (<0.1) | 8 | (0.2) |
| Osteoporosis | 79 | (2.0) | 44 | (1.1) | 6 | (0.2) | 7 | (0.2) |
| Constipation | 59 | (1.5) | 95 | (2.4) | 4 | (0.1) | 1 | (<0.1) |
| Endometrial Proliferation Disorders | 10 | (0.3) | 71 | (1.8) | 1 | (<0.1) | 12 | (0.3) |
| Endometrial Cancer 1 | 7/3089 | (0.2) | 12/3157 | (0.4) | - | - | - | - |
| Other Endometrial Disorders | 3 | (<0.1) | 4 | (0.1) | 0 | - | 1 | (<0.1) |
| Myocardial Infarction | 17 | (0.4) | 14 | (0.4) | 15 | (0.4) | 11 | (0.3) |
| Cerebrovascular/TIA | 44 | (1.1) | 41 | (1.0) | 43 | (1.1) | 40 | (1.0) |
| Angina | 27 | (0.7) | 24 | (0.6) | 17 | (0.4) | 7 | (0.2) |
| Thromboembolic Event | 44 | (1.1) | 109 | (2.7) | 29 | (0.7) | 79 | (2.0) |
| Other Cardiovascular | 261 | (6.6) | 248 | (6.2) | 97 | (2.4) | 71 | (1.8) |
| Second Malignancies 2 | 76/4003 | (1.9) | 96/4007 | (2.4) | - | - | - | - |
| 1 Based on safety population excluding patients who had undergone hysterectomy; time frame is any time after randomization; no CTC grades collected (yes/no response) 2 Based on the intent-to-treat population; time frame is any time after randomization; no CTC grades collected (yes/no response) |
||||||||
When considering all grades, a higher incidence of events were seen for Femara regarding fractures (5.7% vs 4%), myocardial infarctions (0.6% vs 0.4%), and arthralgia (21.2% vs 13.5%) (Femara vs tamoxifen, respectively). A higher incidence was seen for tamoxifen regarding thromboembolic events (1.2% vs 2.8%), endometrial cancer (0.2% vs 0.4%), and endometrial proliferative disorders (0.3% vs 1.8%) (Femara vs tamoxifen, respectively).
Extended Adjuvant Treatment of Early Breast Cancer in Postmenopausal Women Who have Received 5 Years of Adjuvant Tamoxifen Therapy
The median duration of extended adjuvant treatment was 24 months and the median duration of follow-up for safety was 28 months for patients receiving Femara and placebo.
Table 14 describes the adverse events occurring at a frequency of at least 5% in any treatment group during treatment. Most adverse events reported were Grade 1 and Grade 2 based on the Common Toxicity Criteria Version 2.0. In the extended adjuvant setting, the reported drug-related adverse events that were significantly different from placebo were hot flashes, arthralgia/arthritis, and myalgia.
Table 14: Percentage of Patients with Adverse Events
| Number (%) of Patients with Grade 1-4 Adverse Event | Number (%) of Patients with Grade 3-4 Adverse Event | |||
| Femara® | Placebo | Femara® | Placebo | |
| N=2563 | N=2573 | N=2563 | N=2573 | |
| Any Adverse Event | 2232 (87.1) | 2174 (84.5) | 419 (16.3) | 389 (15.1) |
| Vascular Disorders | 1375 (53.6) | 1230 (47.8) | 59 (2.3) | 74 (2.9) |
| Flushing | 1273 (49.7) | 1114 (43.3) | 3 (0.1) | 0 - |
| General Disorders | 1154 (45.0) | 1090 (42.4) | 30 (1.2) | 28 (1.1) |
| Asthenia | 862 (33.6) | 826 (32.1) | 16 (0.6) | 7 (0.3) |
| Edema NOS | 471 (18.4) | 416 (16.2) | 4 (0.2) | 3 (0.1) |
| Musculoskeletal Disorders | 978 (38.2) | 836 (32.5) | 71 (2.8) | 50 (1.9) |
| Arthralgia | 565 (22.0) | 465 (18.1) | 25 (1.0) | 20 (0.8) |
| Arthritis NOS | 173 (6.7) | 124 (4.8) | 10 (0.4) | 5 (0.2) |
| Myalgia | 171 (6.7) | 122 (4.7) | 8 (0.3) | 6 (0.2) |
| Back Pain | 129 (5.0) | 112 (4.4) | 8 (0.3) | 7 (0.3) |
| Nervous System Disorders | 863 (33.7) | 819 (31.8) | 65 (2.5) | 58 (2.3) |
| Headache | 516 (20.1) | 508 (19.7) | 18 (0.7) | 17 (0.7) |
| Dizziness | 363 (14.2) | 342 (13.3) | 9 (0.4) | 6 (0.2) |
| Skin Disorders | 830 (32.4) | 787 (30.6) | 17 (0.7) | 16 (0.6) |
| Sweating Increased | 619 (24.2) | 577 (22.4) | 1 (<0.1) | 0 - |
| Gastrointestinal Disorders | 725 (28.3) | 731 (28.4) | 43 (1.7) | 42 (1.6) |
| Constipation | 290 (11.3) | 304 (11.8) | 6 (0.2) | 2 (<0.1) |
| Nausea | 221 (8.6) | 212 (8.2) | 3 (0.1) | 10 (0.4) |
| Diarrhea NOS | 128 (5.0) | 143 (5.6) | 12 (0.5) | 8 (0.3) |
| Metabolic Disorders | 551 (21.5) | 537 (20.9) | 24 (0.9) | 32 (1.2) |
| Hypercholesterolemia | 401 (15.6) | 398 (15.5) | 2 (<0.1) | 5 (0.2) |
| Reproductive Disorders | 303 (11.8) | 357 (13.9) | 9 (0.4) | 8 (0.3) |
| Vaginal Hemorrhage | 123 (4.8) | 171 (6.6) | 2 (<0.1) | 5 (0.2) |
| Vulvovaginal Dryness | 137 (5.3) | 127 (4.9) | 0 - | 0 - |
| Psychiatric Disorders | 320 (12.5) | 276 (10.7) | 21 (0.8) | 16 (0.6) |
| Insomnia | 149 (5.8) | 120 (4.7) | 2 (<0.1) | 2 (<0.1) |
| Respiratory Disorders | 279 (10.9) | 260 (10.1) | 30 (1.2) | 28 (1.1) |
| Dyspnea | 140 (5.5) | 137 (5.3) | 21 (0.8) | 18 (0.7) |
| Investigations | 184 (7.2) | 147 (5.7) | 13 (0.5) | 13 (0.5) |
| Infections and Infestations | 166 (6.5) | 163 (6.3) | 40 (1.6) | 33 (1.3) |
| Renal Disorders | 130 (5.1) | 100 (3.9) | 12 (0.5) | 6 (0.2) |
The duration of follow-up for both the main clinical study and the bone study were insufficient to assess fracture risk associated with long-term use of Femara. Based on a median follow-up of patients for 28 months, the incidence of clinical fractures from the core randomized study in patients who received Femara was 5.9% (152) and placebo was 5.5% (142). The incidence of self-reported osteoporosis was higher in patients who received Femara 6.9% (176) than in patients who received placebo 5.5% (141). Bisphosphonates were administered to 21.1% of the patients who received Femara and 18.7% of the patients who received placebo.
Preliminary results (median duration of follow-up was 20 months) from the bone sub-study (Calcium 500 mg and Vitamin D 400 IU per day mandatory; bisphosphonates not allowed) demonstrated that at 2 years the mean decrease compared to baseline in hip BMD in Femara patients was 3% vs 0.4% for placebo. The mean decrease from baseline BMD results for the lumbar spine at 2 years were Femara 4.6% decrease and placebo 2.2%.
The incidence of cardiovascular ischemic events from the core randomized study was comparable between patients who received Femara 6.8% (175) and placebo 6.5% (167).
Preliminary results (median duration of follow-up was 30 months) from the lipid sub-study did not show significant differences between the Femara and placebo groups. The HDL:LDL ratio decreased after the first 6 months of therapy but the decrease was similar in both groups and no statistically significant differences were detected.
A patient-reported measure that captures treatment impact on important symptoms associated with estrogen deficiency demonstrated a difference in favor of placebo for vasomotor and sexual symptom domains.
First-Line Breast Cancer
A total of 455 patients was treated for a median time of exposure of 11 months. The incidence of adverse experiences was similar for Femara and tamoxifen. The most frequently reported adverse experiences were bone pain, hot flushes, back pain, nausea, arthralgia and dyspnea. Discontinuations for adverse experiences other than progression of tumor occurred in 10/455 (2%) of patients on Femara and in 15/455 (3%) of patients on tamoxifen.
Adverse events, regardless of relationship to study drug, that were reported in at least 5% of the patients treated with Femara 2.5 mg or tamoxifen 20 mg in the first-line treatment study are shown in Table 15.
Table 15: Percentage (%) of Patients with Adverse Events
| Adverse | Femara® | tamoxifen |
| Experience | 2.5 mg | 20 mg |
| (N=455) | (N=455) | |
| % | % | |
| General Disorders | ||
| Fatigue | 13 | 13 |
| Chest Pain | 8 | 9 |
| Edema Peripheral | 5 | 6 |
| Pain NOS | 5 | 7 |
| Weakness | 6 | 4 |
| Investigations | ||
| Weight Decreased | 7 | 5 |
| Vascular Disorders | ||
| Hot Flushes | 19 | 16 |
| Hypertension | 8 | 4 |
| Gastrointestinal Disorders | ||
| Nausea | 17 | 17 |
| Constipation | 10 | 11 |
| Diarrhea | 8 | 4 |
| Vomiting | 7 | 8 |
| Infections/Infestations | ||
| Influenza | 6 | 4 |
| Urinary Tract Infection NOS | 6 | 3 |
| Injury, Poisoning and Procedural Complications | ||
| Post-Mastectomy Lymphedema | 7 | 7 |
| Metabolism and Nutrition Disorders | ||
| Anorexia | 4 | 6 |
| Musculoskeletal and Connective Tissue Disorders | ||
| Bone Pain | 22 | 21 |
| Back Pain | 18 | 19 |
| Arthralgia | 16 | 15 |
| Pain in Limb | 10 | 8 |
| Nervous System Disorders | ||
| Headache NOS | 8 | 7 |
| Psychiatric Disorders | ||
| Insomnia | 7 | 4 |
| Reproductive System and Breast Disorders | ||
| Breast Pain | 7 | 7 |
| Respiratory, Thoracic and Mediastinal Disorders | ||
| Dyspnea | 18 | 17 |
| Cough | 13 | 13 |
| Chest Wall Pain | 6 | 6 |
Other less frequent (≤2%) adverse experiences considered consequential for both treatment groups, included peripheral thromboembolic events, cardiovascular events, and cerebrovascular events. Peripheral thromboembolic events included venous thrombosis, thrombophlebitis, portal vein thrombosis and pulmonary embolism. Cardiovascular events included angina, myocardial infarction, myocardial ischemia, and coronary heart disease. Cerebrovascular events included transient ischemic attacks, thrombotic or hemorrhagic strokes and development of hemiparesis.
Second-Line Breast Cancer
Femara was generally well tolerated in two controlled clinical trials.
Study discontinuations in the megestrol acetate comparison study for adverse events other than progression of tumor 5/188 (2.7%) on Femara 0.5 mg, in 4/174 (2.3%) on Femara 2.5 mg, and in 15/190 (7.9%) on megestrol acetate. There were fewer thromboembolic events at both Femara doses than on the megestrol acetate arm (0.6% vs 4.7%). There was also less vaginal bleeding (0.3% vs 3.2%) on Femara than on megestrol acetate. In the aminoglutethimide comparison study, discontinuations for reasons other than progression occurred in 6/193 (3.1%) on 0.5 mg Femara, 7/185 (3.8%) on 2.5 mg Femara, and 7/178 (3.9%) of patients on aminoglutethimide.
Comparisons of the incidence of adverse events revealed no significant differences between the high and low dose Femara groups in either study. Most of the adverse events observed in all treatment groups were mild to moderate in severity and it was generally not possible to distinguish adverse reactions due to treatment from the consequences of the patient’s metastatic breast cancer, the effects of estrogen deprivation, or intercurrent illness.
Adverse events, regardless of relationship to study drug, that were reported in at least 5% of the patients treated with Femara 0.5 mg, Femara 2.5 mg, megestrol acetate, or aminoglutethimide in the two controlled trials are shown in Table 16.
Table 16: Percentage (%) of Patients with Adverse Events
| Adverse | Pooled | Pooled | megestrol | |
| Experience | Femara® | Femara® | acetate | aminoglutethimide |
| 2.5 mg | 0.5 mg | 160 mg | 500 mg | |
| (N=359) | (N=380) | (N=189) | (N=178) | |
| % | % | % | % | |
| Body as a Whole | ||||
| Fatigue | 8 | 6 | 11 | 3 |
| Chest Pain | 6 | 3 | 7 | 3 |
| Peripheral Edema1 | 5 | 5 | 8 | 3 |
| Asthenia | 4 | 5 | 4 | 5 |
| Weight Increase | 2 | 2 | 9 | 3 |
| Cardiovascular | ||||
| Hypertension | 5 | 7 | 5 | 6 |
| Digestive System | ||||
| Nausea | 13 | 15 | 9 | 14 |
| Vomiting | 7 | 7 | 5 | 9 |
| Constipation | 6 | 7 | 9 | 7 |
| Diarrhea | 6 | 5 | 3 | 4 |
| Pain-Abdominal | 6 | 5 | 9 | 8 |
| Anorexia | 5 | 3 | 5 | 5 |
| Dyspepsia | 3 | 4 | 6 | 5 |
| Infections/Infestations | ||||
| Viral Infection | 6 | 5 | 6 | 3 |
| Lab Abnormality | ||||
| Hypercholesterolemia | 3 | 3 | 0 | 6 |
| Musculoskeletal System | ||||
| Musculoskeletal2 | 21 | 22 | 30 | 14 |
| Arthralgia | 8 | 8 | 8 | 3 |
| Nervous System | ||||
| Headache | 9 | 12 | 9 | 7 |
| Somnolence | 3 | 2 | 2 | 9 |
| Dizziness | 3 | 5 | 7 | 3 |
| Respiratory System | ||||
| Dyspnea | 7 | 9 | 16 | 5 |
| Coughing | 6 | 5 | 7 | 5 |
| Skin and Appendages | ||||
| Hot Flushes | 6 | 5 | 4 | 3 |
| Rash3 | 5 | 4 | 3 | 12 |
| Pruritus | 1 | 2 | 5 | 3 |
| 1 Includes peripheral edema, leg edema, dependent edema, edema 2 Includes musculoskeletal pain, skeletal pain, back pain, arm pain, leg pain 3 Includes rash, erythematous rash, maculopapular rash, psoriasiform rash, vesicular rash |
||||
Other less frequent (<5%) adverse experiences considered consequential and reported in at least 3 patients treated with Femara, included hypercalcemia, fracture, depression, anxiety, pleural effusion, alopecia, increased sweating and vertigo.
First-Line and Second-Line Breast Cancer
In the combined analysis of the first- and second-line metastatic trials and post-marketing experiences other adverse events that were reported were cataract, eye irritation, palpitations, cardiac failure, tachycardia, dysesthesia (including hypoesthesia/paresthesia), arterial thrombosis, memory impairment, irritability, nervousness, urticaria, increased urinary frequency, leukopenia, stomatitis cancer pain, pyrexia, vaginal discharge, appetite increase, dryness of skin and mucosa (including dry mouth), and disturbances of taste and thirst.
Post-Marketing Experiences
Cases of blurred vision, increased hepatic enzymes, angioedema and anaphylactic reactions have been reported.
OVERDOSAGE
Isolated cases of Femara® (letrozole tablets) overdose have been reported. In these instances, the highest single dose ingested was 62.5 mg or 25 tablets. While no serious adverse events were reported in these cases, because of the limited data available, no firm recommendations for treatment can be made. However, emesis could be induced if the patient is alert. In general, supportive care and frequent monitoring of vital signs are also appropriate. In single-dose studies, the highest dose used was 30 mg, which was well tolerated; in multiple-dose trials, the largest dose of 10 mg was well tolerated.
Lethality was observed in mice and rats following single oral doses that were equal to or greater than 2,000 mg/kg (about 4,000 to 8,000 times the daily maximum recommended human dose on a mg/m2 basis); death was associated with reduced motor activity, ataxia and dyspnea. Lethality was observed in cats following single IV doses that were equal to or greater than 10 mg/kg (about 50 times the daily maximum recommended human dose on a mg/m2 basis); death was preceded by depressed blood pressure and arrhythmias.
DOSAGE AND ADMINISTRATION
Adult and Elderly Patients
The recommended dose of Femara® (letrozole tablets) is one 2.5 mg tablet administered once a day, without regard to meals. In patients with advanced disease, treatment with Femara should continue until tumor progression is evident.
In the extended adjuvant setting, the optimal treatment duration with Femara is not known. The planned duration of treatment in the study was 5 years. However, at the time of the analysis, the median treatment duration was 24 months, 25% of patients were treated for at least 3 years and less than 1% of patients were treated for the planned duration of 5 years. The median duration of follow-up was 28 months. Treatment should be discontinued at tumor relapse (see CLINICAL STUDIES).
In the adjuvant setting, the optimal duration of treatment with letrozole is unknown. The planned duration of treatment in the study is 5 years. However, at the time of analysis, the median duration of treatment was 24 months, median duration of follow-up was 26 months, and 16% of the patients have been treated for 5 years. Treatment should be discontinued at relapse (see CLINICAL STUDIES).
No dose adjustment is required for elderly patients. Patients treated with Femara do not require glucocorticoid or mineralocorticoid replacement therapy.
Renal Impairment
(See CLINICAL PHARMACOLOGY.) No dosage adjustment is required for patients with renal impairment if creatinine clearance is ≥10 mL/min.
Hepatic Impairment
No dosage adjustment is recommended for patients with mild to moderate hepatic impairment, although Femara blood concentrations were modestly increased in subjects with moderate hepatic impairment due to cirrhosis. The dose of Femara in patients with cirrhosis and severe hepatic dysfunction should be reduced by 50% (see CLINICAL PHARMACOLOGY). The recommended dose of Femara for such patients is 2.5 mg administered every other day. The effect of hepatic impairment on Femara exposure in noncirrhotic cancer patients with elevated bilirubin levels has not been determined. (See CLINICAL PHARMACOLOGY.)
HOW SUPPLIED
2.5 mg tablets - dark yellow, film-coated, round, slightly biconvex, with beveled edges (imprinted with the letters FV on one side and CG on the other side).
Packaged in HDPE bottles with a safety screw cap.
Bottles of 30 tablets NDC 0078-0249-15
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
REV: SEPTEMBER 2008 T2008-92
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
| FEMARA
letrozole tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
Revised: 12/2008Novartis Pharmaceuticals Corporation